Advanced Photocatalytic Olefin Difunctionalization for Scalable Pharmaceutical Intermediate Production
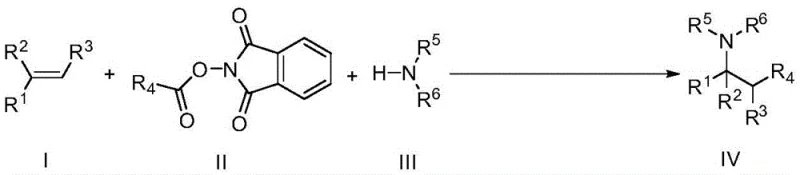
The chemical industry is constantly evolving towards more sustainable and efficient synthetic methodologies, particularly in the realm of complex molecule construction. Patent CN108707081B introduces a groundbreaking approach to olefin 1,2-difunctionalization, a critical transformation in the synthesis of high-value pharmaceutical intermediates and fine chemicals. This technology leverages visible-light photoredox catalysis to achieve bond formation under exceptionally mild conditions, specifically at room temperature, which stands in stark contrast to traditional thermal methods requiring harsh energy inputs. By utilizing readily available starting materials such as vinyl compounds, N-substituted phthalimides, and amines, this process opens new avenues for constructing nitrogen-containing scaffolds that are ubiquitous in bioactive molecules. The strategic integration of a photocatalyst and a specific reaction assistant allows for precise control over reactivity, minimizing side reactions and improving overall process safety. For R&D teams seeking robust pathways to complex amine derivatives, this patent represents a significant leap forward in synthetic capability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of olefins has relied heavily on transition metal catalysis involving precious metals such as Palladium, Rhodium, or Iron, often necessitating elevated temperatures and stringent anhydrous conditions. These conventional protocols frequently suffer from limited substrate tolerance, where sensitive functional groups may decompose under the rigorous thermal stress required to drive the reaction to completion. Furthermore, the reliance on stoichiometric oxidants or harsh reagents can generate substantial quantities of hazardous waste, complicating downstream processing and increasing the environmental footprint of the manufacturing process. The removal of trace heavy metal residues from the final product is another critical bottleneck, often requiring additional purification steps that erode overall yield and increase production costs. Consequently, there is a pressing industrial need for methodologies that can bypass these thermal and toxicological constraints while maintaining high levels of chemical efficiency and selectivity.
The Novel Approach
The methodology disclosed in CN108707081B circumvents these traditional limitations by employing a visible-light-driven photoredox catalytic system that operates effectively at ambient temperature. This novel approach utilizes the energy from photons to excite the photocatalyst, generating reactive radical species that facilitate the difunctionalization of the olefin double bond without the need for external heating. The use of mild conditions not only preserves the integrity of sensitive functional groups present on the substrate but also significantly reduces the energy consumption associated with the reaction process. Additionally, the protocol employs a specific Lewis acid assistant, tris(pentafluorophenyl)boron, which synergistically enhances the reactivity of the system, allowing for high conversion rates even with sterically hindered substrates. This shift from thermal to photochemical activation represents a paradigm change in how chemists approach C-N and C-C bond formation, offering a cleaner and more sustainable alternative for industrial applications.
Mechanistic Insights into Photoredox-Catalyzed Olefin Difunctionalization
The core of this transformative chemistry lies in the intricate interplay between the photocatalyst, typically Ru(bpy)3Cl2, and the reaction components under illumination. Upon absorption of visible light, the ruthenium complex enters an excited state capable of engaging in single-electron transfer processes with the N-substituted phthalimide derivative. This electron transfer event generates a nitrogen-centered radical species, which subsequently adds across the carbon-carbon double bond of the vinyl compound to form a new carbon-centered radical intermediate. The presence of the amine nucleophile then traps this intermediate, completing the 1,2-difunctionalization sequence to yield the target product. The careful selection of the solvent, with dimethyl sulfoxide (DMSO) identified as optimal, plays a crucial role in stabilizing these charged or radical intermediates, ensuring smooth progression of the catalytic cycle. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for specific substrate classes.
Impurity control is inherently superior in this photocatalytic system due to the high selectivity of the radical generation step and the mild reaction environment. Unlike thermal methods that might promote non-selective polymerization or decomposition of the olefin, the photochemical pathway directs reactivity specifically towards the desired difunctionalization. The use of an inert atmosphere, such as argon or nitrogen, further suppresses oxidative side reactions that could lead to byproduct formation. Moreover, the ability to monitor the reaction progress via thin-layer chromatography (TLC) or GC-MS allows for precise endpoint determination, preventing over-reaction or degradation of the product. The post-processing involves straightforward concentration and silica gel column chromatography, which effectively separates the target molecule from any unreacted starting materials or minor byproducts, ensuring high purity specifications suitable for pharmaceutical applications.
How to Synthesize Olefin Difunctionalized Products Efficiently
Implementing this synthesis route requires attention to detail regarding the light source and atmospheric conditions to ensure reproducibility and high yields. The patent outlines a standardized procedure where reactants are combined in a Schlenk sealed tube reactor, ensuring an oxygen-free environment that is critical for the stability of the radical intermediates. While the general protocol is robust, slight adjustments in the molar ratios of the photocatalyst and the boron assistant can fine-tune the reaction kinetics for different substrate combinations. The following guide summarizes the critical operational steps derived from the patent data to assist technical teams in replicating this efficient synthesis.
- Charge a Schlenk sealed tube reactor with the vinyl compound, N-substituted phthalimide compound, amine, photocatalyst such as Ru(bpy)3Cl2, reaction assistant like B(C6F5)3, and organic solvent such as DMSO.
- Stir the reaction mixture at room temperature under an inert atmosphere while illuminating with a light source, such as a 5W blue LED or energy-saving lamp, for approximately 24 hours.
- Upon completion monitored by TLC, concentrate the mixture under reduced pressure and purify the residue via silica gel column chromatography using a hexane and ethyl acetate mixture to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this photocatalytic technology offers compelling advantages that directly address key pain points in chemical procurement and supply chain management. The elimination of expensive precious metal catalysts like Palladium significantly lowers the raw material costs, while the ability to recover and reuse the photocatalyst further enhances the economic efficiency of the process. The mild reaction conditions reduce the demand for specialized high-temperature or high-pressure equipment, allowing for production in standard glass-lined reactors which are more readily available and cheaper to maintain. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The substitution of traditional thermal catalysts with a recyclable photocatalytic system drastically reduces the cost of goods sold by eliminating the need for expensive metal scavenging processes. Since the reaction proceeds at room temperature, energy costs associated with heating and cooling are substantially minimized, leading to significant operational expenditure savings. Furthermore, the use of commercially available and inexpensive starting materials ensures that the input costs remain stable and predictable, shielding the manufacturing process from volatile market fluctuations associated with rare earth metals.
- Enhanced Supply Chain Reliability: The reliance on broadly available reagents such as styrenes, phthalimides, and common amines mitigates the risk of supply chain disruptions often caused by the scarcity of specialized catalysts. The simplicity of the reaction setup means that production can be easily scaled or shifted between different manufacturing sites without requiring extensive requalification of equipment. This flexibility ensures consistent delivery timelines and reduces the lead time for high-purity pharmaceutical intermediates, allowing downstream customers to maintain lean inventory levels with confidence.
- Scalability and Environmental Compliance: The environmental profile of this method is exceptionally favorable, as it avoids the generation of heavy metal waste streams that require costly disposal and treatment. The straightforward workup procedure involving concentration and chromatography simplifies the purification train, making the process highly amenable to commercial scale-up from kilogram to multi-ton scales. Compliance with increasingly stringent environmental regulations is easier to achieve, reducing the regulatory burden and potential liabilities associated with hazardous waste management in large-scale chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this photocatalytic technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of this photocatalytic method over traditional transition metal catalysis?
A: The primary advantage is the use of mild reaction conditions at room temperature without the need for expensive and toxic heavy metal catalysts like Palladium or Rhodium, significantly reducing environmental impact and purification costs.
Q: Can the photocatalyst be recovered and reused in this process?
A: Yes, the patent specifies that the photocatalyst Ru(bpy)3Cl2 can be recovered and recycled through a simple filtration method, enhancing the economic viability of the process.
Q: What is the substrate scope for this olefin difunctionalization reaction?
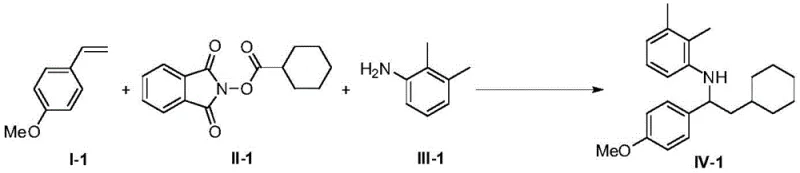
A: The method demonstrates a wide application range, accommodating various substituted vinyl compounds, N-substituted phthalimides, and diverse amines including cyclic and acyclic structures, making it highly versatile for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olefin Difunctionalization Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in modern drug synthesis and are committed to delivering these advanced solutions to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of olefin difunctionalized intermediates meets the highest quality standards required by the pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this innovative method can optimize your supply chain and reduce overall manufacturing costs.
