Advanced Semi-Synthetic Routes for Scalable Paclitaxel and Docetaxel Production
Advanced Semi-Synthetic Routes for Scalable Paclitaxel and Docetaxel Production
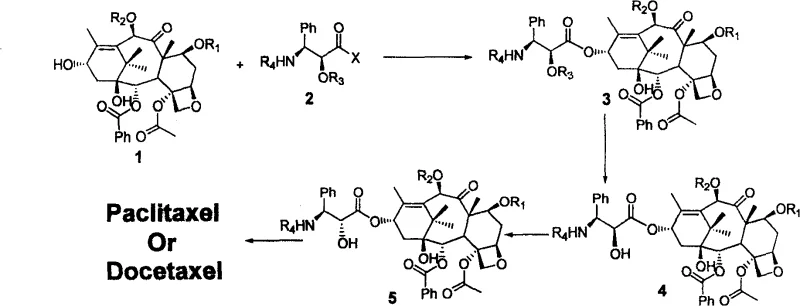
The global demand for potent antineoplastic agents continues to surge, placing immense pressure on the supply chains of critical oncology drugs like Paclitaxel and Docetaxel. Traditional extraction from Taxus species is environmentally unsustainable and yields are notoriously low, driving the industry toward robust semi-synthetic alternatives. Patent CN100586940C introduces a transformative semi-synthetic methodology that addresses these bottlenecks by leveraging a novel chiral side chain docking strategy. This technology bypasses the arduous direct synthesis of the native cis-configured side chain, opting instead for a more accessible trans-isomer followed by a precise stereochemical inversion. For R&D directors and procurement specialists, this represents a pivotal shift towards cost-effective, high-purity API intermediate manufacturing that aligns with modern green chemistry principles while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthesis of taxanes has been plagued by the extreme difficulty and high cost associated with preparing the C-13 side chain in its correct natural stereochemistry. Conventional routes often require complex asymmetric synthesis or resolution steps to establish the (2'R, 3'S) configuration directly, which drastically inflates raw material costs and extends lead times. Furthermore, direct coupling reactions using these sensitive cis-side chains often suffer from epimerization risks and lower yields due to steric hindrance and the delicate nature of the intermediates. These inefficiencies create significant vulnerabilities in the supply chain, making the final API price volatile and dependent on the availability of highly specialized, low-volume chiral building blocks. The reliance on cryogenic conditions or exotic catalysts in older methods further complicates commercial scale-up, limiting the ability of manufacturers to respond rapidly to market spikes in demand for these life-saving medications.
The Novel Approach
The methodology disclosed in CN100586940C fundamentally reengineers this workflow by utilizing a readily available trans-chiral side chain, which is synthetically much simpler to produce in bulk quantities. The core innovation lies in the post-coupling stereochemical inversion, where the configuration at the 2'-position is flipped after the side chain is already attached to the baccatin core. This approach allows for the use of robust condensation conditions, typically employing dicyclohexylcarbodiimide (DCC) and DMAP in toluene at moderate temperatures of 30 to 40°C. By decoupling the complexity of chirality establishment from the coupling event, the process achieves exceptional yields, with experimental data showing step yields exceeding 86% for the initial condensation and maintaining high efficiency through the inversion steps. This streamlined pathway not only simplifies the operational protocol but also significantly enhances the overall throughput of the manufacturing line.
Mechanistic Insights into Stereochemical Inversion Strategies
The heart of this patented process is the precise control over stereochemistry during the inversion phase, which differs slightly between the synthesis of Paclitaxel and Docetaxel to accommodate their specific structural requirements. For Paclitaxel production, the mechanism involves the formation of an oxazoline intermediate. After the initial condensation and deprotection of the side chain hydroxyl group, the intermediate is treated with Vilsmeier reagent in anhydrous pyridine. This generates a cyclic oxazoline species that locks the conformation, which is subsequently hydrolyzed under acidic reflux conditions to release the desired (2'R, 3'S) configuration with high optical purity. This intramolecular participation ensures that the inversion is clean and minimizes the formation of diastereomeric impurities that are notoriously difficult to separate in taxane chemistry.
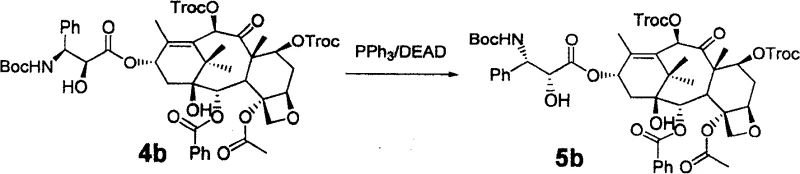
In the case of Docetaxel, the patent leverages the classic Mitsunobu reaction to achieve the necessary stereochemical flip. Following the removal of the silyl protecting groups (such as TBS) using HF/Pyridine, the exposed 2'-hydroxyl group undergoes inversion using triphenylphosphine (PPh3) and diethyl azodicarboxylate (DEAD). This reaction proceeds via an SN2-type mechanism where the alcohol is activated as a leaving group and displaced by a carboxylate nucleophile (often from the protecting group itself or an added acid), effectively inverting the stereocenter. The beauty of this mechanistic approach lies in its reliability; the Mitsunobu reaction is well-understood and highly predictable, allowing process chemists to tightly control impurity profiles. The subsequent global deprotection using Zinc powder in acetic acid/methanol cleanly removes the remaining Troc and acetyl groups without affecting the sensitive oxetane ring or the newly established ester linkage.
How to Synthesize Paclitaxel and Docetaxel Efficiently
Implementing this semi-synthetic route requires careful attention to reagent stoichiometry and purification protocols to maximize the benefits of the high-yield design. The process begins with the activation of the trans-chiral side chain acid, followed by coupling with the protected 10-deacetylbaccatin III (10-DAB) nucleus. Critical control points include the maintenance of anhydrous conditions during the coupling and inversion steps to prevent hydrolysis of the activated intermediates. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures validated in the patent examples, are provided below to guide your process development team in replicating these results.
- Condense protected 10-deacetylbaccatin III (10-DAB) with a novel trans-chiral side chain using DCC and DMAP in toluene at 30-40°C to form the ester linkage.
- Remove the hydroxyl protecting group (e.g., TBS or Troc) from the side chain using acidic conditions (HCl/MeOH) or fluoride sources (HF/Pyridine) to expose the 2'-hydroxyl group.
- Invert the stereochemistry at the 2'-position: use Vilsmeier reagent followed by hydrolysis for Paclitaxel, or Mitsunobu reaction (PPh3/DEAD) for Docetaxel.
- Perform final global deprotection of the baccatin core (7-OH and 10-OH) using Zinc powder in acetic acid/methanol to yield the final active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this semi-synthetic methodology offers profound strategic advantages that extend far beyond simple yield improvements. By shifting the synthetic burden to a more accessible trans-side chain precursor, the dependency on scarce, high-cost chiral starting materials is drastically reduced. This structural change in the supply base means that manufacturers are no longer held hostage by the limited capacity of specialized chiral synthesis vendors, thereby stabilizing the cost of goods sold (COGS) and insulating the project from raw material price volatility. The use of commodity reagents such as DCC, Zinc powder, and common organic solvents like toluene and methanol further ensures that the production process remains economically viable even at fluctuating market rates, as these inputs are widely available from multiple global suppliers.
- Cost Reduction in Manufacturing: The elimination of complex asymmetric synthesis steps for the side chain translates directly into substantial cost savings. Since the trans-side chain is easier to prepare, the upstream manufacturing costs are significantly lower compared to traditional cis-side chain routes. Additionally, the high yields reported in the patent examples (consistently above 70-80% per step) mean that less raw material is wasted, and the throughput per batch is maximized. The avoidance of expensive transition metal catalysts or cryogenic equipment also reduces capital expenditure (CAPEX) and operational expenditure (OPEX), making the process highly attractive for large-scale commercial production where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The robustness of this chemical route significantly de-risks the supply chain. The reaction conditions are mild, typically ranging from room temperature to moderate reflux, which reduces the likelihood of batch failures due to thermal runaway or equipment malfunction. Because the intermediates are stable and the reagents are non-hazardous commodities, sourcing is straightforward and lead times for raw materials are minimized. This reliability ensures that production schedules can be met consistently, preventing stock-outs of the final API and allowing pharmaceutical partners to maintain steady inventory levels for their finished dosage forms without the need for excessive safety stocks.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is designed for industrial feasibility. The solvents used are standard and easily recoverable, facilitating efficient recycling and waste management protocols that align with strict environmental regulations. The final deprotection step using Zinc in acetic acid is a clean reduction that generates manageable waste streams compared to heavy metal hydrogenation methods. This ease of waste treatment, combined with the high atom economy of the coupling and inversion steps, supports a greener manufacturing profile. Consequently, scaling from pilot plant to multi-ton commercial production is seamless, requiring no fundamental changes to the chemistry, which accelerates time-to-market for generic or biosimilar versions of these critical cancer therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthetic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply partnership.
Q: Why is the trans-chiral side chain preferred over the cis-side chain in this synthesis?
A: The trans-chiral side chain is significantly easier and less expensive to synthesize compared to the naturally occurring cis-configuration. This patent utilizes a strategic stereochemical inversion step post-coupling to convert the easily accessible trans-form into the required cis-configuration, thereby reducing overall raw material costs and synthetic complexity.
Q: What are the key reagents used for the stereochemical inversion step?
A: The method employs two distinct inversion strategies depending on the target molecule. For Paclitaxel, an oxazoline intermediate is formed using Vilsmeier reagent followed by acid hydrolysis. For Docetaxel, a classic Mitsunobu reaction utilizing triphenylphosphine (PPh3) and diethyl azodicarboxylate (DEAD) is used to invert the 2'-hydroxyl stereocenter with high fidelity.
Q: How does this process improve supply chain reliability for taxane APIs?
A: By decoupling the difficult synthesis of the chiral side chain from the coupling step, manufacturers can stockpile the simpler trans-side chain intermediates. Furthermore, the use of robust, non-cryogenic reaction conditions (30-40°C condensation, room temperature inversion) allows for easier scale-up in standard stainless steel reactors, minimizing batch failures and ensuring consistent supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel and Docetaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing muscle. Our facility is equipped to handle the complexities of taxane semi-synthesis, leveraging advanced process engineering to optimize the condensation and inversion steps described in CN100586940C. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and potency of every batch, guaranteeing that our API intermediates meet the highest global regulatory standards.
We invite you to collaborate with us to unlock the full potential of this efficient semi-synthetic route for your oncology portfolio. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive down your costs while securing a reliable, long-term supply of high-purity Paclitaxel and Docetaxel intermediates.
