Advanced Manufacturing of Ofloxacin: A High-Yield Route for Global Supply Chains
Introduction to Advanced Ofloxacin Manufacturing
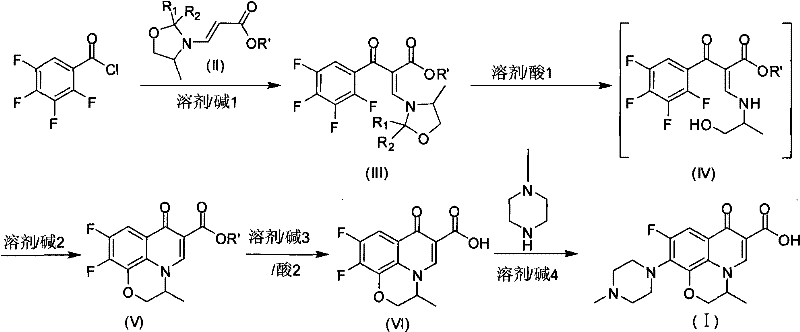
The global demand for high-quality fluoroquinolone antibiotics necessitates manufacturing processes that balance efficiency with rigorous purity standards. Patent CN101648960B introduces a transformative methodology for the preparation of Ofloxacin, addressing critical bottlenecks found in legacy production techniques. This innovation centers on a novel condensation strategy using tetrafluorobenzoyl chloride as the primary starting material, bypassing the cumbersome multi-step sequences typical of earlier generations. By integrating a specialized oxazolidinyl acrylate intermediate, the process achieves a dramatic reduction in production cycles while maintaining exceptional raw material utilization rates. For pharmaceutical manufacturers, this represents a pivotal shift towards more sustainable and economically viable API production, ensuring a stable supply of this essential antimicrobial agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ofloxacin has been plagued by inefficiencies that drive up costs and complicate supply chain logistics. Traditional routes, such as those described in US4777253, rely on 2,3,4,5-tetrafluorobenzoic acid and involve lengthy sequences including condensation decarboxylation and triethyl orthoformate reactions, resulting in a dismal total recovery rate of approximately 21%. Alternative pathways utilizing trifluoronitrobenzene derivatives, referenced in patents like US4382892, fare even worse with total yields hovering around 14% and generating substantial quantities of hazardous waste. These legacy methods not only consume excessive amounts of raw materials but also produce intermediates that are often difficult to purify, typically appearing as yellow powders that require energy-intensive refining processes to meet pharmacopeial standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN101648960B streamlines the synthesis by directly reacting tetrafluorobenzoyl chloride with 3-(2-R1-2-R2-4-methyl oxazolidinyl) acrylate. This strategic modification eliminates several intermediate isolation steps, allowing for direct hydrolysis and cyclization during post-processing. The result is a robust process where the yield for the critical cyclization step reaches an impressive 85-90%, vastly outperforming the 50-70% yields seen in comparable prior art. Furthermore, the intermediates generated, specifically Compounds V and VI, precipitate as high-purity white solids, effectively removing the need for complex purification protocols associated with colored impurities. This streamlined approach not only accelerates production timelines but also significantly enhances the overall economic feasibility of large-scale manufacturing.
Mechanistic Insights into Tetrafluorobenzoyl Chloride Condensation
The core of this technological advancement lies in the precise mechanistic interaction between the electrophilic tetrafluorobenzoyl chloride and the nucleophilic enamine system of the oxazolidinyl acrylate. Unlike traditional condensations that might require harsh conditions or unstable intermediates, this reaction proceeds efficiently in the presence of organic or inorganic bases at moderate temperatures ranging from 50-90°C. The oxazolidine ring serves a dual purpose: it stabilizes the acrylic moiety during the initial coupling and facilitates the subsequent intramolecular cyclization required to form the pyridobenzoxazine core. This controlled reactivity minimizes side reactions and polymerization, which are common pitfalls in quinolone synthesis, thereby ensuring a cleaner reaction profile and higher selectivity for the desired regioisomer.
Impurity control is inherently built into the chemical design of this pathway. The use of specific solvents like acetonitrile or chlorobenzene, which can be recovered and reused, helps maintain a consistent reaction environment that discourages the formation of by-products. Moreover, the hydrolysis step is carefully managed to convert the ester intermediate directly into the cyclized acid without isolating unstable species. The resulting carboxylic acid intermediate (Compound VI) exhibits an HPLC content greater than 99%, with single impurities maintained below 0.2%. This high level of chemical fidelity is crucial for downstream processing, as it ensures that the final substitution with methylpiperazine proceeds without the interference of residual contaminants that could compromise the safety profile of the final API.
How to Synthesize Ofloxacin Efficiently
The synthesis of Ofloxacin via this patented route involves a sequence of highly optimized steps designed for reproducibility and scale. The process begins with the preparation of the key acrylate intermediate, followed by the critical condensation with the fluorinated benzoyl chloride. Subsequent hydrolysis and cyclization steps are telescoped to maximize throughput, culminating in the final amination to yield the active pharmaceutical ingredient. The detailed operational parameters, including specific temperature ramps, stoichiometric ratios, and workup procedures, are essential for achieving the reported high yields and purity levels. For a comprehensive breakdown of the standardized synthesis protocol, please refer to the technical guide below.
- Condense tetrafluorobenzoyl chloride with 3-(2-R1-2-R2-4-methyl oxazolidinyl) acrylate in the presence of an organic base at 50-90°C to form the intermediate acrylate.
- Hydrolyze the intermediate using acid treatment followed by base-catalyzed cyclization to form the benzoxazine carboxylic ester (Compound V).
- Perform saponification to obtain the carboxylic acid (Compound VI) and react with methylpiperazine to finalize the Ofloxacin structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits that extend beyond simple yield improvements. The reduction in reaction steps directly correlates to lower operational expenditures, as fewer unit operations mean reduced labor, energy, and equipment usage. Additionally, the ability to recover and reuse solvents throughout the process significantly diminishes raw material costs and waste disposal fees. The generation of white solid intermediates rather than yellow powders simplifies logistics, as these materials are easier to handle, filter, and transport without the risk of degradation or contamination that often accompanies less stable forms. This operational simplicity translates into a more resilient supply chain capable of meeting fluctuating market demands with greater agility.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in methylpiperazine usage through optimized alkaline conditions lead to substantial cost savings. By avoiding the need for extensive refining of colored intermediates, manufacturers can significantly lower their utility consumption and solvent purchase requirements. The high atom economy of the condensation reaction ensures that a greater proportion of raw materials are converted into valuable product, minimizing waste and maximizing return on investment for every batch produced.
- Enhanced Supply Chain Reliability: The robustness of this chemical pathway ensures consistent output quality, reducing the risk of batch failures that can disrupt supply schedules. The use of readily available starting materials like tetrafluorobenzoyl chloride and common organic solvents mitigates the risk of raw material shortages. Furthermore, the shortened production cycle allows for faster turnaround times, enabling suppliers to respond more rapidly to urgent orders and maintain healthy inventory levels without tying up excessive working capital in long-duration production runs.
- Scalability and Environmental Compliance: This process is inherently designed for commercial scale-up, with reaction conditions that are easily manageable in large reactors without compromising safety or efficiency. The significant reduction in "three wastes" (waste water, gas, and residue) aligns with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing facilities. The ability to operate with high concentrations and efficient solvent recovery systems further enhances the sustainability profile of the manufacturing site, making it a preferred partner for environmentally conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ofloxacin synthesis method. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams assessing supplier qualifications.
Q: How does this new synthesis route improve yield compared to traditional methods?
A: Traditional methods utilizing trifluoronitrobenzene or multi-step condensation often suffer from total yields as low as 14% to 21%. This patented process achieves a yield of 85-90% for the critical cyclization step by utilizing a direct condensation with oxazolidinyl acrylate, significantly minimizing material loss.
Q: What are the purity advantages of the intermediates produced in this process?
A: Unlike conventional additive methods that produce yellow powders requiring extensive refining, this process generates white solid intermediates (Compound V and VI) with HPLC content exceeding 99%. This inherent high purity reduces the need for aggressive recrystallization steps.
Q: Does this method offer environmental benefits for large-scale production?
A: Yes, the process utilizes recoverable organic solvents such as acetonitrile or chlorobenzene throughout the reaction sequence rather than switching solvents frequently. Additionally, the reduction in reaction steps and the optimization of methylpiperazine usage contribute to a significant decrease in chemical waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ofloxacin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable manufacturing processes in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of advanced patents like CN101648960B are fully realized in practical application. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Ofloxacin or its intermediates meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of fluoroquinolone synthesis with precision, delivering products that support the global fight against bacterial infections.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this high-yield methodology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of our high-quality intermediates into your manufacturing operations.
