Advanced Liquid-Phase Synthesis of Fenoxaprop-p-ethyl for Commercial Scale Production
The global demand for high-efficiency, low-toxicity herbicides continues to drive innovation in agrochemical intermediate manufacturing. A significant breakthrough in this sector is detailed in patent CN111732553B, which discloses a novel method for synthesizing fenoxaprop-p-ethyl, a critical active ingredient for controlling gramineous weeds in cereal crops. This patent addresses long-standing industrial challenges by transitioning from traditional solid-liquid phase reactions to a more efficient liquid-phase process. The core innovation lies in the strategic use of an acetone-water solvent system coupled with in-situ solvent evaporation, which not only enhances reaction kinetics but also facilitates superior solvent recovery. For R&D directors and process engineers, this represents a pivotal shift towards greener chemistry, offering a pathway to achieve product purity exceeding 99.9% optical rotation content while drastically simplifying downstream processing. As a leading manufacturer, understanding these mechanistic nuances is essential for optimizing supply chains and ensuring the consistent delivery of high-purity fenoxaprop-p-ethyl to the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of fenoxaprop-p-ethyl has been plagued by the inefficiencies inherent in solid-liquid phase reactions. Traditional methodologies often require vast quantities of organic solvents to maintain suspension and facilitate mass transfer, leading to inflated operational expenditures and complex solvent recovery systems. Furthermore, these legacy processes frequently generate substantial volumes of nitrogen-containing wastewater, posing severe environmental compliance challenges and necessitating costly treatment protocols before discharge. The heterogeneity of solid-liquid systems also introduces variability in reaction rates, often resulting in incomplete conversions and the formation of difficult-to-remove impurities that compromise the final optical purity of the herbicide. For procurement managers, these factors translate into unpredictable lead times and higher overall costs per kilogram of active ingredient, creating bottlenecks in the commercial scale-up of complex agrochemical intermediates.
The Novel Approach
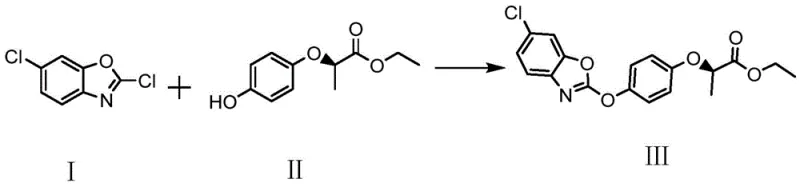
In stark contrast, the methodology outlined in patent CN111732553B leverages a homogeneous liquid-phase reaction environment that fundamentally alters the production landscape. By dissolving 2,6-dichlorobenzoxazole in acetone and reacting it with the chiral phenol substrate in an aqueous base system, the process ensures intimate contact between reactants, thereby accelerating the nucleophilic substitution. A defining feature of this approach is the simultaneous evaporation of acetone during the reaction at 70-75°C, which drives the equilibrium forward and prevents the hydrolysis of the sensitive benzoxazole ring. This dynamic solvent management eliminates the need for large-scale post-reaction solvent separation, as the acetone is removed continuously and can be recovered via membrane technology. Consequently, this novel route offers a streamlined workflow that significantly reduces energy consumption and waste generation, positioning it as a superior choice for cost reduction in herbicide manufacturing.
Mechanistic Insights into Phase Transfer Catalyzed Nucleophilic Substitution
The chemical transformation at the heart of this synthesis is a nucleophilic aromatic substitution where the phenoxide anion attacks the C-2 position of the 2,6-dichlorobenzoxazole ring. The process initiates with the deprotonation of R-(+)-2-(4-hydroxyphenoxy) ethyl propionate by an inorganic base, such as sodium carbonate, generating a highly reactive phenoxide species in the aqueous phase. To bridge the immiscibility gap between the aqueous base and the organic benzoxazole, a phase transfer catalyst (PTC), preferably benzyltriethylammonium chloride, is employed. The PTC facilitates the transport of the phenoxide anion into the organic micro-environment or the interface where the electrophilic benzoxazole resides. This catalytic cycle ensures that the reaction proceeds rapidly at moderate temperatures (70-75°C) without requiring harsh conditions that could degrade the chiral center or the ester functionality.
Impurity control is meticulously managed through the precise regulation of reaction parameters and stoichiometry. The patent specifies a slight molar excess of 2,6-dichlorobenzoxazole (1:1.01 to 1.05 ratio) to ensure complete consumption of the valuable chiral phenol, preventing unreacted starting material from contaminating the final product. Additionally, the choice of sodium carbonate over stronger alkalis like sodium hydroxide minimizes the risk of ester hydrolysis, a common side reaction that generates carboxylic acid impurities. The continuous removal of acetone not only drives the reaction to completion but also limits the time the intermediate spends in the reactive medium, further suppressing byproduct formation. This rigorous control mechanism guarantees that the final fenoxaprop-p-ethyl product maintains an effective optical rotation content of greater than 99.9%, meeting the stringent quality standards required for reliable agrochemical intermediate supplier certifications.
How to Synthesize Fenoxaprop-p-ethyl Efficiently
Implementing this synthesis route requires careful attention to temperature gradients and addition rates to maximize yield and safety. The process begins with the preparation of two distinct streams: an acetone solution of the benzoxazole and an aqueous slurry containing the chiral phenol, base, and catalyst. The key operational step involves heating the aqueous mixture to 70-75°C and slowly introducing the acetone solution while simultaneously distilling off the acetone. This concurrent addition and evaporation strategy maintains a constant reaction volume and temperature, preventing thermal runaway and ensuring consistent product quality. Following the reaction, the mixture is allowed to settle, allowing for a clean phase separation where the organic layer containing the crude product is isolated.
- Dissolve 2,6-dichlorobenzoxazole in acetone at 20-25°C and prepare a mixed solution of the chiral phenol substrate, inorganic base, catalyst, and water at 60-65°C.
- Heat the aqueous mixture to 70-75°C and dropwise add the acetone solution while simultaneously evaporating acetone to drive the reaction and prevent hydrolysis.
- After reaction completion, separate the organic phase, wash with hot water, remove residual solvents under vacuum, and recrystallize from ethanol to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of complex solid-liquid handling equipment and the reduction in solvent inventory requirements directly lower capital expenditure and working capital needs. By integrating solvent recovery directly into the reaction phase, the process minimizes the volume of waste streams requiring external treatment, thereby reducing environmental compliance costs and enhancing the sustainability profile of the manufacturing site. These efficiencies contribute to a more resilient supply chain capable of withstanding market fluctuations in raw material availability.
- Cost Reduction in Manufacturing: The innovative solvent management system allows for the continuous recovery of acetone, which can be purified and reused, significantly lowering the consumption of fresh organic solvents. Furthermore, the use of inexpensive inorganic bases like sodium carbonate instead of costly organic bases or strong alkalis reduces raw material expenses. The simplified workup procedure, which avoids extensive chromatography or complex crystallization steps, decreases labor and utility costs, resulting in substantial overall cost savings for the production of high-volume herbicide intermediates.
- Enhanced Supply Chain Reliability: The robustness of the liquid-phase reaction ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. The ability to operate at atmospheric pressure and moderate temperatures enhances process safety, minimizing the likelihood of unplanned shutdowns due to equipment failure or safety incidents. This reliability is crucial for maintaining steady inventory levels and meeting the just-in-time delivery expectations of downstream formulators and agrochemical companies seeking a reliable agrochemical intermediate supplier.
- Scalability and Environmental Compliance: The process is inherently scalable, as the heat transfer and mixing dynamics in a liquid-liquid system are easier to manage in large reactors compared to heterogeneous slurries. The significant reduction in nitrogen-containing wastewater and organic solvent waste aligns with increasingly strict global environmental regulations, facilitating smoother permitting and operation in regulated jurisdictions. This environmental advantage future-proofs the manufacturing asset against tightening emission standards, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of fenoxaprop-p-ethyl using this advanced methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and product quality attributes. Understanding these details is vital for partners evaluating the feasibility of integrating this intermediate into their broader agricultural chemical portfolios.
Q: What are the key advantages of the liquid-phase synthesis method for fenoxaprop-p-ethyl?
A: The liquid-phase method described in patent CN111732553B eliminates the difficulties associated with traditional solid-liquid reactions, such as excessive solvent usage and nitrogen-containing wastewater generation. It allows for in-situ solvent recovery and simplifies post-treatment through phase separation.
Q: How is high optical purity maintained during the synthesis process?
A: The process utilizes R-(+)-2-(4-hydroxyphenoxy) ethyl propionate as a starting material which does not undergo stereoisomerism under the reaction conditions. Combined with mild inorganic bases like sodium carbonate, the method ensures the effective optical rotation content remains above 99.9%.
Q: Can the acetone solvent be recovered efficiently in this process?
A: Yes, acetone is evaporated at normal pressure during the reaction at 70-75°C. The resulting acetone-water azeotrope can be separated and recovered at high purity using membrane separation technology, significantly reducing raw material costs and environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fenoxaprop-p-ethyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern agrochemical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111732553B are fully realized in practical manufacturing settings. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that verify every batch for optical rotation content and chemical purity. Our infrastructure is designed to handle complex chemistries safely and efficiently, making us an ideal partner for long-term supply agreements.
We invite global agrochemical manufacturers to collaborate with us to leverage these technological advancements for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our fenoxaprop-p-ethyl solutions align perfectly with your quality and efficiency goals.
