Advanced Synthesis of Fenoxaprop-p-ethyl: Technical Breakthroughs for Commercial Scale-up
Advanced Synthesis of Fenoxaprop-p-ethyl: Technical Breakthroughs for Commercial Scale-up
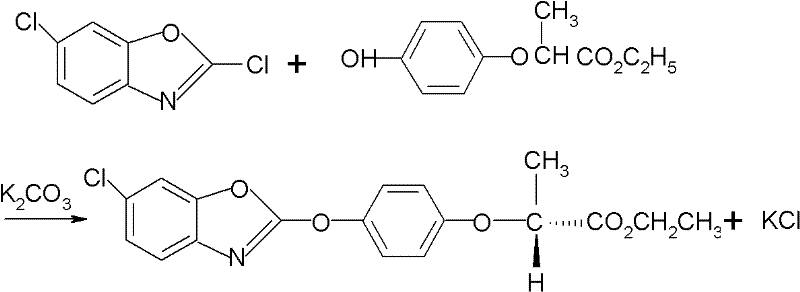
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key agrochemical intermediates. Patent CN102558087A introduces a transformative method for combining high-quality fenoxaprop-p-ethyl, addressing critical bottlenecks in yield and optical purity that have long plagued the industry. This technical disclosure outlines a streamlined process that utilizes R-phenoxy ethyl propionate and 2,6-dichlorobenzoxazole under optimized phase transfer catalytic conditions. By shifting away from complex multi-step derivatizations, this approach achieves a reaction yield exceeding 98% and an optical effective body content greater than 98% without the need for intricate post-processing purification. For R&D directors and procurement specialists, this represents a significant opportunity to enhance the reliability of the supply chain for grass-control herbicides. The method not only ensures stringent quality specifications but also aligns with modern green chemistry principles by reducing waste generation. As a reliable agrochemical intermediate supplier, understanding these mechanistic advancements is crucial for evaluating long-term partnership viability and cost structures in herbicide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
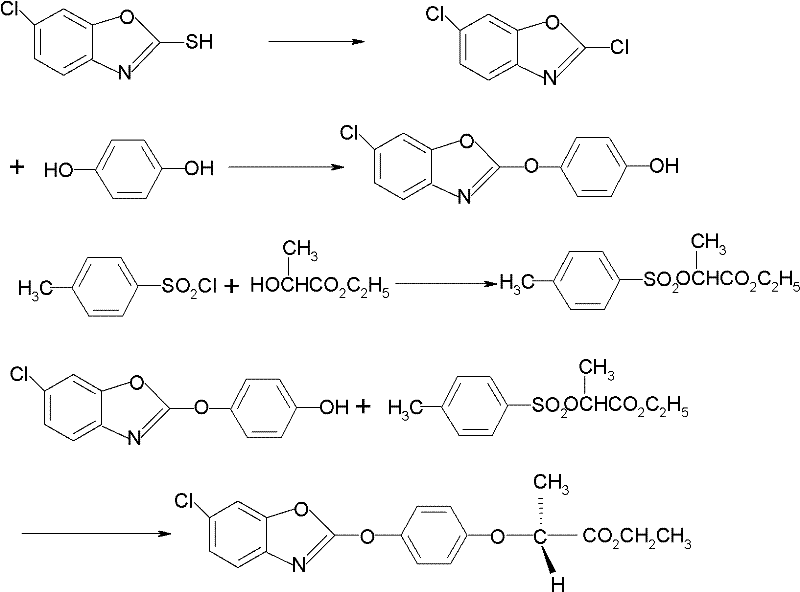
Historically, the production of fenoxaprop-p-ethyl has been hindered by inefficient synthetic routes that compromise both economic viability and environmental compliance. Traditional Route 1, for instance, relies on the condensation of 6-chloro-2-mercaptobenzoxazole with resorcinol, followed by a complex esterification involving L-ethyl lactate and p-toluenesulfonyl chloride. This pathway is fraught with technical deficiencies, including low reaction conversion efficiency and the generation of substantial byproduct waste, specifically p-toluenesulfonic acid, which is difficult to reclaim and poses serious environmental pollution risks. Furthermore, the optical purity in these conventional processes is often suboptimal, typically hovering around 95%, necessitating expensive chiral resolution steps to meet market standards. The reliance on harsh reagents and the inability to effectively recover solvents further exacerbates the production cost, making cost reduction in agrochemical manufacturing a challenging objective for producers adhering to these legacy methods.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data simplifies the molecular construction to a direct coupling reaction that preserves stereochemistry and maximizes atom economy. By reacting R-phenoxy ethyl propionate directly with 2,6-dichlorobenzoxazole in the presence of potassium carbonate and a specific phase transfer catalyst, the process bypasses the need for unstable sulfonate intermediates. This method operates effectively in aromatic hydrocarbon solvents like toluene or xylene, which are far easier to recover and recycle compared to the polar aprotic solvents used in older routes. The result is a direct obtainment of high-quality fenoxaprop-p-ethyl with content and optical purity both surpassing 98%. This drastic simplification of the synthetic pathway not only enhances the commercial scale-up of complex herbicide intermediates but also significantly reduces the operational complexity associated with waste treatment and solvent management, offering a robust solution for industrial application.
Mechanistic Insights into Phase Transfer Catalyzed Nucleophilic Substitution
The core of this technological breakthrough lies in the precise manipulation of nucleophilic substitution dynamics facilitated by phase transfer catalysis (PTC). In this system, the potassium carbonate acts as a base to deprotonate the phenolic hydroxyl group of the R-phenoxy ethyl propionate, generating a reactive phenoxide anion. However, since the reaction occurs in an organic solvent phase where the inorganic base is insoluble, the addition of a quaternary ammonium salt catalyst, such as tetrabutylammonium bromide, is critical. The catalyst transports the phenoxide anion into the organic phase, where it can efficiently attack the electrophilic carbon of the 2,6-dichlorobenzoxazole. This mechanism ensures that the reaction proceeds rapidly at moderate temperatures between 70°C and 110°C, minimizing thermal degradation of the chiral center. The careful control of the molar ratio between the dichloro compound and the phenoxy ester, ideally between 1:0.9 and 1:1.2, further drives the equilibrium towards product formation, ensuring that the optical integrity of the R-enantiomer is maintained throughout the transformation.
Impurity control is another critical aspect where this mechanism outperforms traditional methods. In conventional syntheses, racemization often occurs during the activation of the chiral center or during high-temperature condensation steps. The novel method mitigates this by avoiding the formation of reactive sulfonate esters that are prone to SN1-type racemization pathways. Instead, the direct SN2-type displacement on the benzoxazole ring proceeds with inversion or retention characteristics that are highly predictable and controllable under the specified conditions. Furthermore, the use of water washing for work-up effectively removes inorganic salts and catalyst residues without requiring complex chromatographic separation. This inherent selectivity means that the crude product already meets high-purity fenoxaprop-p-ethyl specifications, reducing the burden on downstream purification units and ensuring a consistent impurity profile that is vital for regulatory approval in key agricultural markets.
How to Synthesize Fenoxaprop-p-ethyl Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters defined in the patent to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of the reaction mixture, where R-phenoxy ethyl propionate, potassium carbonate, and the phase transfer catalyst are suspended in a dry aromatic solvent. Temperature control is paramount during the addition of the 2,6-dichlorobenzoxazole, which should be added slowly to manage the exotherm and maintain the reaction temperature within the optimal 65-90°C range initially. Following the addition, the mixture is heated to 70-110°C and maintained for a duration of 0.5 to 5 hours to ensure complete conversion. The detailed standardized synthesis steps, including specific stirring rates, addition velocities, and quenching protocols, are outlined in the technical guide below to assist process engineers in validating this route for pilot and production scales.
- Mix R-phenoxy ethyl propionate, potassium carbonate, and a phase transfer catalyst in a solvent such as toluene or xylene.
- Heat the mixture to 65-90°C and slowly add 2,6-dichlorobenzoxazole dissolved in the solvent.
- Maintain the reaction temperature between 70-110°C for 0.5 to 5 hours, then separate the oil layer via water washing to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates into tangible strategic advantages that extend beyond simple unit price negotiations. The elimination of complex intermediate isolation steps and the removal of difficult-to-treat byproducts like sulfonic acids drastically simplify the manufacturing workflow. This simplification directly correlates to a reduction in operational expenditures, as fewer unit operations are required to bring the product to market specification. Moreover, the high yield and purity achieved without extensive post-processing mean that raw material utilization is maximized, leading to substantial cost savings in the overall production budget. These efficiencies make the supply chain more resilient to fluctuations in raw material pricing, as the process is less sensitive to yield losses that typically erode margins in less optimized synthetic routes.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive chiral resolving agents and complex purification columns that were previously necessary to achieve acceptable optical purity. By achieving over 98% optical purity directly from the reaction, the manufacturing cost is significantly reduced through the removal of these value-adding but cost-intensive steps. Additionally, the ability to use common, recoverable solvents like toluene instead of specialized polar solvents reduces solvent procurement costs and waste disposal fees. The qualitative improvement in process efficiency means that the same production capacity can output a higher volume of saleable product, effectively lowering the fixed cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes that rely on niche reagents or generate hazardous waste that limits production capacity. This method utilizes readily available starting materials and generates minimal hazardous waste, reducing the risk of regulatory shutdowns or supply interruptions. The robustness of the reaction conditions allows for flexible scheduling and faster turnaround times between batches. Consequently, reducing lead time for high-purity agrochemical intermediates becomes achievable, ensuring that downstream formulators receive their materials on schedule. This reliability is crucial for maintaining just-in-time inventory levels and meeting the seasonal demands of the global agricultural market without the need for excessive safety stock.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this reaction operates under mild thermal conditions that are easily managed in large-scale reactors. The absence of highly toxic reagents and the generation of benign inorganic salts as byproducts simplify the environmental compliance landscape. Waste treatment facilities can handle the effluent more easily, reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the brand value of the final product in markets that prioritize sustainable sourcing. The process is inherently designed for commercial scale-up of complex herbicide intermediates, ensuring that quality remains consistent from the gram scale to the multi-ton scale.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of adopting this route for industrial production. Understanding these details is essential for technical teams evaluating the integration of this process into existing manufacturing lines. The data provided reflects the specific conditions and outcomes reported in the intellectual property, offering a transparent view of the technology's capabilities and limitations in a commercial context.
Q: How does the novel synthesis method improve optical purity compared to conventional routes?
A: The novel method utilizes R-phenoxy ethyl propionate directly with 2,6-dichlorobenzoxazole under specific phase transfer catalytic conditions. This avoids the racemization issues common in older routes that required complex transposition steps, ensuring optical effective body content exceeds 98% without extensive post-processing.
Q: What are the primary cost drivers eliminated in this new manufacturing process?
A: Traditional methods often generate difficult-to-recover byproducts like p-toluenesulfonic acid or require expensive solvents like DMSO that are hard to reclaim. The new process uses recoverable aromatic hydrocarbon solvents and eliminates the formation of heavy sulfonic acid waste, drastically simplifying waste treatment and raw material costs.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the reaction conditions operate at moderate temperatures (70-110°C) using common solvents like toluene. The high yield (>98%) and simplified work-up (direct liquid separation) make it highly scalable for commercial manufacturing, reducing lead time for high-purity agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fenoxaprop-p-ethyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent data to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of fenoxaprop-p-ethyl meets the highest international standards for agrochemical intermediates. Our commitment to quality assurance means that we can consistently deliver the high optical purity and chemical content required by modern herbicide formulations, providing a stable foundation for your product development and market expansion strategies.
We invite you to collaborate with us to optimize your supply chain and leverage these technical advancements for your business growth. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. By partnering with us, you gain access to a reliable agrochemical intermediate supplier dedicated to driving innovation and efficiency in the global agricultural chemical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
