Scalable Manufacturing of High-Purity BTK Inhibitors Using Novel Slurry Purification Technology
The pharmaceutical industry is constantly seeking robust synthetic routes that balance high purity with industrial feasibility, particularly for complex kinase inhibitors. Patent CN115385920A introduces a groundbreaking refinement method for synthesizing BTK inhibitor compounds of Formula III, addressing critical bottlenecks in traditional manufacturing. This technology leverages a quaternary ammonium salt catalytic system combined with a unique multi-stage slurry purification process, effectively bypassing the need for expensive microwave reactors and labor-intensive column chromatography. By shifting from specialized microwave conditions to conventional thermal heating at 80-85°C, the process significantly lowers the barrier for commercial scale-up. The result is a streamlined workflow that delivers yields up to 92% and exceptional purity levels reaching 99.4%, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their oncology drug pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrolopyrimidine-based BTK inhibitors has relied heavily on technologies that are notoriously difficult to scale, such as microwave-assisted organic synthesis and silica gel column chromatography. While effective on a laboratory benchtop, microwave reactors require specialized, high-cost equipment that poses significant safety and engineering challenges when transitioning to multi-kilogram or ton-scale production. Furthermore, the reliance on column chromatography for purification creates a massive bottleneck; it consumes vast quantities of silica gel and organic solvents, generates substantial hazardous waste, and requires extensive manual labor for fraction collection and analysis. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, rendering many promising candidates economically unviable for mass market distribution in the competitive landscape of cost reduction in API manufacturing.
The Novel Approach
The innovative strategy outlined in the patent data replaces these cumbersome steps with a thermally driven reaction mediated by quaternary ammonium salts, such as tetrabutylammonium iodide, in the presence of an amine base like DIPEA. Instead of chromatography, the purification relies on a sophisticated "beating" or slurry method involving sequential washes with water and benign solvents like ethyl acetate. This approach not only simplifies the operational workflow but also dramatically reduces solvent consumption and waste generation. The reaction proceeds cleanly in isopropanol, a green solvent choice, and the subsequent purification steps effectively strip away unreacted starting materials and side products. This transition from complex separation techniques to crystallization-based purification represents a paradigm shift towards greener, more economical chemical production suitable for high-volume commercial demands.
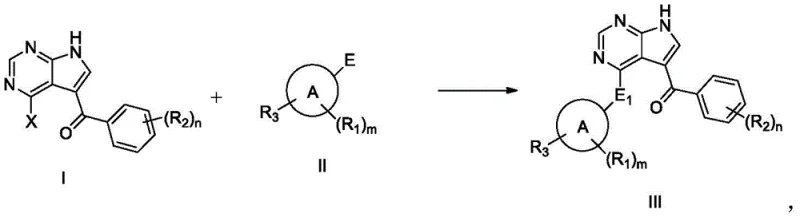
Mechanistic Insights into Quaternary Ammonium Salt-Catalyzed Substitution
The core of this synthetic advancement lies in the efficient nucleophilic substitution facilitated by the phase-transfer capabilities of the quaternary ammonium catalyst. In the reaction between the halogenated pyrrolopyrimidine precursor (Formula I) and the amine-containing tetrahydropyran derivative (Formula II), the catalyst enhances the nucleophilicity of the amine species within the organic phase. The bulky organic cation of the salt helps to solubilize the reactive anionic species or stabilize the transition state, allowing the reaction to proceed smoothly at moderate temperatures of 80-85°C without the need for extreme thermal energy or microwave irradiation. This mechanistic efficiency ensures that the conversion is thorough, minimizing the presence of residual starting materials which are often the most difficult impurities to remove during downstream processing.
Furthermore, the purification mechanism is designed to exploit the differential solubility profiles of the product versus its impurities. The multi-step slurry process begins with a water wash to remove polar inorganic salts and highly water-soluble by-products. Subsequent beating with benign organic solvents at elevated temperatures followed by cooling allows for the selective crystallization of the target BTK inhibitor while keeping lipophilic impurities in solution. The final precipitation step using a strong polar organic solvent like DMF followed by water addition ensures that any remaining trace impurities are excluded from the crystal lattice. This rigorous physical separation strategy guarantees the high purity of 99.4% observed in the experimental data, providing R&D teams with a material that requires minimal further processing before formulation.
How to Synthesize BTK Inhibitor Formula III Efficiently
Implementing this refined synthesis route requires precise control over reaction stoichiometry and purification parameters to maximize yield and quality. The process begins by charging the reactor with the halogenated precursor and the amine coupling partner in isopropanol, followed by the addition of the base and the quaternary ammonium catalyst. Once the reaction reaches completion, typically monitored by HPLC, the workup involves a series of concentration and slurry steps that are critical for impurity rejection. Operators must carefully manage the temperature gradients during the beating phases, specifically heating to 60-80°C to dissolve surface impurities and then cooling to 20-30°C to induce crystallization of the pure product. For a detailed breakdown of the specific operational parameters and solvent ratios, please refer to the standardized guide below.
- React Formula I and Formula II compounds with DIPEA and a quaternary ammonium salt catalyst in isopropanol at 80-85°C.
- Concentrate the reaction mixture and perform an initial water slurry purification at room temperature to remove bulk impurities.
- Execute a multi-step beating process using benign solvents like ethyl acetate and polar solvent precipitation to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patent technology offers transformative benefits that directly impact the bottom line and operational resilience. By eliminating the dependency on column chromatography, manufacturers can drastically reduce the consumption of silica gel and high-grade chromatographic solvents, which are significant cost drivers in fine chemical production. This reduction in material usage translates directly into substantial cost savings and a smaller environmental footprint, aligning with modern sustainability goals. Additionally, the removal of microwave synthesis requirements means that production can be executed in standard stainless steel reactors available in most CDMO facilities, removing the need for capital-intensive equipment upgrades and facilitating faster technology transfer between sites.
- Cost Reduction in Manufacturing: The elimination of column chromatography is the single most impactful cost-saving measure in this process. Chromatography is inherently inefficient on a large scale, requiring large columns, massive solvent volumes, and significant labor for operation and solvent recovery. By replacing this with a slurry-based purification, the process reduces solvent usage by a significant margin and eliminates the cost of silica gel entirely. Furthermore, the use of common solvents like isopropanol and ethyl acetate, rather than exotic or highly regulated solvents, ensures that raw material costs remain low and stable. This economic efficiency allows for more competitive pricing structures for the final BTK inhibitor intermediate, enhancing the overall value proposition for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Relying on standard thermal reaction conditions rather than specialized microwave technology significantly de-risks the supply chain. Microwave reactors are often single-source or limited-supply items with long lead times for maintenance and parts. In contrast, the heating requirements for this process (80-85°C) can be met by ubiquitous jacketed reactors found in virtually every chemical plant globally. This universality ensures that production is not bottlenecked by equipment availability. Moreover, the robustness of the slurry purification method means that minor variations in reaction conditions do not necessitate complex re-purification steps, leading to more consistent batch-to-batch quality and reliable delivery schedules for customers awaiting critical oncology intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale, moving seamlessly from gram-scale optimization to multi-ton commercial production without fundamental changes to the unit operations. The reduction in solvent waste and the avoidance of silica gel disposal simplify waste management protocols and reduce the burden on environmental compliance teams. The use of "benign solvents" as defined in the patent further supports green chemistry initiatives, making the manufacturing process more attractive to partners with strict ESG (Environmental, Social, and Governance) mandates. This scalability ensures that as demand for BTK inhibitors grows in the global market, the supply can be ramped up quickly without encountering the technical ceilings often associated with chromatographic purifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this BTK inhibitor refining method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or for assessing the quality of intermediates sourced from external suppliers utilizing this methodology.
Q: How does this method improve scalability compared to prior art?
A: By eliminating microwave synthesis and column chromatography, the process utilizes standard thermal reactors and simple filtration equipment, drastically reducing equipment complexity and operational costs for large-scale production.
Q: What purity levels can be achieved with this refining method?
A: The patented slurry purification technique consistently achieves product purity levels up to 99.4%, meeting stringent specifications required for pharmaceutical intermediate supply without the need for extensive chromatographic separation.
Q: Is the quaternary ammonium catalyst difficult to remove?
A: The specific beating and washing protocols described in the patent effectively remove catalyst residues and by-products, ensuring the final filter cake meets rigorous quality control standards for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BTK Inhibitor Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-quality synthesis routes for next-generation kinase inhibitors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are fully equipped to implement the quaternary ammonium catalyzed synthesis described in CN115385920A, leveraging our rigorous QC labs to maintain stringent purity specifications that exceed industry standards. Our commitment to process excellence means we can deliver high-purity BTK inhibitor intermediates that empower your drug development programs without the delays associated with traditional purification bottlenecks.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing capability can benefit your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new analogues, we are ready to provide the support you need. Contact us today to secure a reliable supply of high-quality pharmaceutical intermediates and accelerate your path to clinical and commercial success.
