Scalable Production of Chiral Avapritinib Intermediates Using Novel Rhodium Catalysis
Scalable Production of Chiral Avapritinib Intermediates Using Novel Rhodium Catalysis
The pharmaceutical landscape for oncology treatments continues to evolve with the demand for high-purity kinase inhibitors like Avapritinib (Ayvakit), a breakthrough therapy for gastrointestinal stromal tumors (GIST). A critical challenge in the supply chain for this potent drug lies in the efficient and stereoselective synthesis of its key chiral building blocks. Patent CN111471020B, published in March 2022, introduces a transformative preparation method for the Avapritinib intermediate (S)-1-(4-fluorophenyl)-1-[2-(piperazine-1-yl)pyrimidin-5-yl]ethylamine. This technology represents a significant departure from conventional synthetic strategies by leveraging advanced organometallic catalysis to establish the crucial chiral center. For global procurement teams and R&D directors, this patent offers a pathway to a more robust, safe, and economically viable supply of high-purity pharmaceutical intermediates. By replacing hazardous reagents with stable organoboron species and utilizing a sophisticated rhodium catalyst system, this method addresses the twin pressures of regulatory compliance and cost efficiency in pharmaceutical intermediates manufacturing.
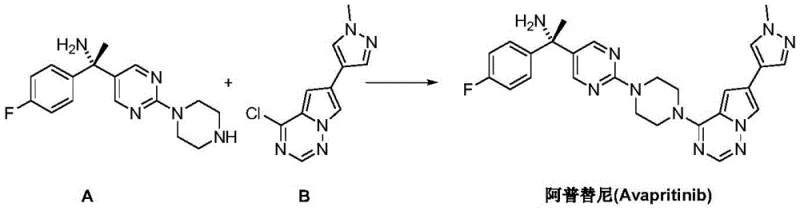
The structural complexity of Avapritinib necessitates precise control over stereochemistry, as the biological activity is strictly dependent on the (S)-configuration. The traditional approach to synthesizing such chiral amines often relies on the formation of chiral sulfinyl imines followed by nucleophilic addition. However, these legacy methods frequently involve harsh conditions, expensive chiral auxiliaries that must be recovered, and reagents that pose significant safety risks on a large scale. The innovation disclosed in CN111471020B circumvents these bottlenecks by introducing a streamlined three-step sequence: imidization, rhodium-catalyzed asymmetric addition, and hydrolysis. This strategic shift not only simplifies the process flow but also enhances the overall atom economy and safety profile, positioning it as a superior choice for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral benzylic amines required for kinase inhibitors has relied heavily on the addition of organometallic reagents to imines derived from chiral sulfinamides. As illustrated in prior art, this typically involves the reaction of a ketone precursor with a chiral sulfinamide to form a sulfinyl imine, followed by the addition of a methyl Grignard reagent. This conventional pathway presents multiple critical vulnerabilities for industrial supply chains. Firstly, Grignard reagents are pyrophoric and moisture-sensitive, requiring stringent anhydrous conditions and specialized handling equipment, which drastically increases capital expenditure and operational risk. Secondly, the reaction often demands cryogenic temperatures (e.g., -78°C) to maintain stereoselectivity, leading to excessive energy consumption and limiting reactor throughput. Furthermore, the chiral sulfinamide auxiliary adds significant material cost and requires an additional cleavage and recovery step, complicating the purification process and generating substantial chemical waste.
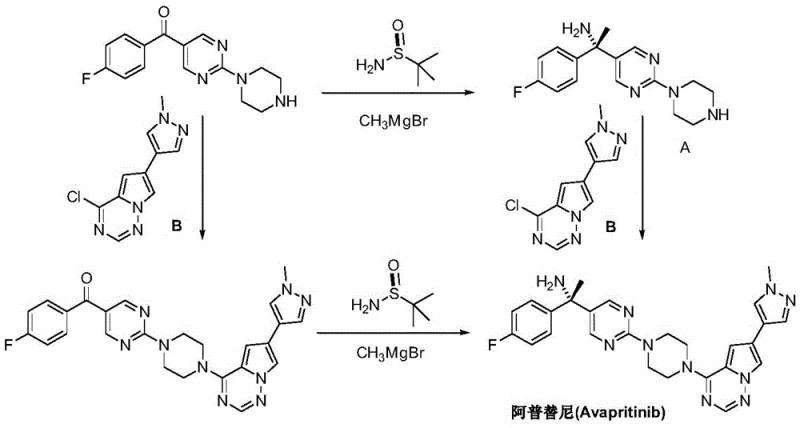
The Novel Approach
In stark contrast, the methodology outlined in patent CN111471020B employs a transition metal-catalyzed asymmetric addition of organoboron reagents to N-sulfonyl imines. This novel route utilizes 4-fluorophenyl potassium trifluoroborate as a stable, non-pyrophoric nucleophile, eliminating the safety hazards associated with Grignard chemistry. The reaction proceeds under mild thermal conditions (50-100°C), removing the need for energy-intensive cryogenic cooling. The core of this innovation is the use of a chiral rhodium complex which directs the stereochemical outcome with high fidelity, directly installing the required (S)-configuration without the need for stoichiometric chiral auxiliaries that must be discarded or recycled. This approach streamlines the synthesis into a more direct and manageable process, significantly reducing lead time for high-purity pharmaceutical intermediates while improving the environmental footprint of the manufacturing operation.
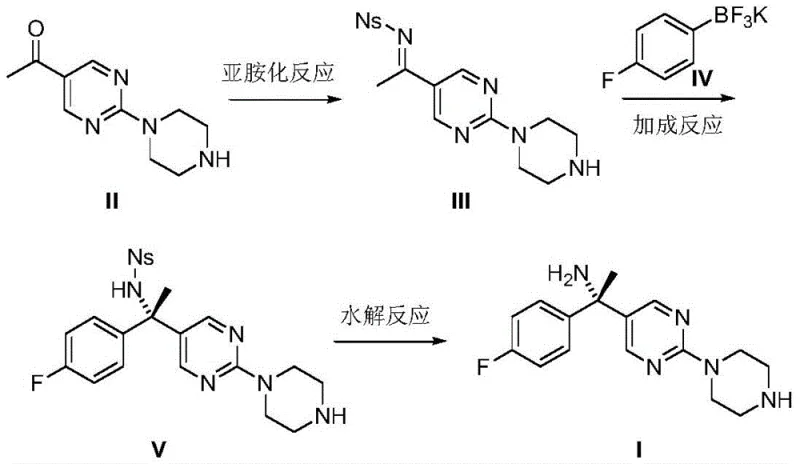
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Addition
The heart of this technological advancement lies in the sophisticated catalytic cycle driven by the chiral rhodium complex. The reaction initiates with the formation of an N-p-toluenesulfonyl imine intermediate via the condensation of the ketone precursor with p-toluenesulfonamide. This imine serves as the electrophile in the subsequent stereodetermining step. The chiral rhodium catalyst, specifically bis[(2,3,5,6-eta)-(1R,4R)-2,5-2,5-bis(4-trifluoromethylphenyl)bicyclo[2.2.2]octane-2-diene]bis-mu-rhodium chloride complex, facilitates the transmetallation of the aryl group from the potassium trifluoroborate to the metal center. This generates a reactive aryl-rhodium species that undergoes a highly organized 1,2-addition to the C=N bond of the imine. The bulky chiral ligands on the rhodium center create a specific steric environment that favors the attack from one face of the imine, thereby ensuring the exclusive formation of the (S)-enantiomer. This mechanism bypasses the need for low-temperature kinetic control, relying instead on the thermodynamic stability and precise geometry of the catalyst-substrate complex.
Following the asymmetric addition, the resulting sulfonamide-protected amine undergoes a mild hydrolysis to reveal the free amine functionality. The patent specifies the use of potassium carbonate and thiophenol in DMF at a remarkably low temperature of 30°C. This gentle deprotection strategy is crucial for maintaining the integrity of the sensitive heterocyclic core and preventing racemization of the newly formed chiral center. By avoiding strong acids or harsh basic conditions typically used in sulfinamide cleavage, this step minimizes the formation of degradation byproducts and simplifies downstream purification. The result is a crude product with high optical purity, reducing the burden on crystallization or chromatography steps and ensuring a consistent quality profile essential for regulatory approval of the final API.
How to Synthesize (S)-1-(4-fluorophenyl)-1-[2-(piperazine-1-yl)pyrimidin-5-yl]ethylamine Efficiently
The implementation of this synthesis route requires careful attention to catalyst loading and solvent selection to maximize yield and enantiomeric excess. The process begins with the imidization of the ketone in toluene, followed by the critical rhodium-catalyzed coupling in dioxane, and concludes with hydrolysis in DMF. Each step has been optimized to balance reaction rate with selectivity, ensuring that the process is robust enough for multi-kilogram production. The detailed standardized operating procedures, including specific molar ratios and workup protocols, are essential for replicating the high performance reported in the patent data.
- Perform imidization of 1-[2-(piperazine-1-yl)pyrimidin-5-yl]ethanone with p-toluenesulfonamide using p-toluenesulfonic acid catalyst in toluene at 110°C.
- Execute stereoselective addition using 4-fluorophenyl potassium trifluoroborate and a chiral rhodium complex catalyst in dioxane at 80°C.
- Conduct alkaline hydrolysis of the sulfonamide protecting group using potassium carbonate and thiophenol catalyst in DMF at 30°C to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed route offers tangible strategic benefits beyond mere technical elegance. The shift away from hazardous reagents and extreme conditions translates directly into improved operational continuity and reduced risk exposure. By stabilizing the supply chain against the volatility associated with dangerous chemical handling, manufacturers can ensure more reliable delivery schedules for their downstream API partners. Furthermore, the simplified workflow reduces the number of unit operations and potential failure points, enhancing the overall resilience of the production line.
- Cost Reduction in Manufacturing: The elimination of cryogenic infrastructure and pyrophoric reagents leads to a substantial decrease in both capital and operational expenditures. The use of stable organoboron reagents reduces waste disposal costs associated with quenching reactive metals, while the high selectivity of the rhodium catalyst minimizes the loss of valuable starting materials to unwanted isomers. Additionally, the avoidance of stoichiometric chiral auxiliaries removes a significant cost driver, resulting in a more economically efficient process overall.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel pathway, such as potassium trifluoroborates and standard organic solvents, are commercially available and stable upon storage, unlike moisture-sensitive Grignard reagents that require fresh preparation or specialized logistics. This stability ensures that raw material inventory can be maintained safely, preventing production delays caused by reagent degradation. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for specialized low-temperature jackets, increasing equipment availability.
- Scalability and Environmental Compliance: The process operates within a moderate temperature range (30-110°C), which is easily manageable in large-scale reactors, facilitating seamless technology transfer from pilot plant to commercial production. The reduced generation of hazardous waste and the use of less toxic reagents align with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. This compliance reduces the administrative burden and potential fines associated with waste management, further supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is vital for R&D teams evaluating the feasibility of adopting this technology for their own production lines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation.
Q: What are the safety advantages of this Rh-catalyzed route over traditional Grignard methods?
A: Unlike traditional methods requiring pyrophoric methyl Grignard reagents and cryogenic temperatures, this patent utilizes stable organoboron reagents and operates at mild temperatures (50-100°C), significantly reducing operational hazards and energy consumption.
Q: How is high chiral purity achieved in this synthesis?
A: High enantioselectivity is achieved through the use of a specialized chiral rhodium complex catalyst, specifically bis[(2,3,5,6-eta)-(1R,4R)-2,5-2,5-bis(4-trifluoromethylphenyl)bicyclo[2.2.2]octane-2-diene]bis-mu-rhodium chloride, which induces the S-configuration during the carbon-nitrogen double bond addition.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process avoids extreme conditions and uses common solvents like toluene and dioxane. The mild hydrolysis step at 30°C further enhances scalability, making it highly suitable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avapritinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving oncology therapies. Our technical team has extensively analyzed the advancements presented in patent CN111471020B and possesses the expertise to implement this rhodium-catalyzed methodology effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of monitoring chiral purity and impurity profiles to meet stringent purity specifications required by global regulatory bodies.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their Avapritinib supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer, more efficient route. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments tailored to your project timelines and volume requirements.
