Advanced Manufacturing of Avapritinib via Selective Boc-Protection Strategy
Advanced Manufacturing of Avapritinib via Selective Boc-Protection Strategy
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for potent kinase inhibitors, particularly for oncology applications where purity and supply continuity are paramount. Patent CN110950872A introduces a refined methodology for the preparation of Avapritinib, a selective inhibitor of PDGFRα and KIT mutations, which addresses critical limitations in prior art synthesis. This technical disclosure outlines a strategic approach that prioritizes the protection of reactive functional groups to enhance reaction selectivity and total yield. By implementing a Boc-protection strategy on the amino group of the initial intermediate prior to the halogenation and coupling steps, the process effectively mitigates the formation of complex byproduct profiles. This innovation is particularly significant for commercial manufacturers aiming to streamline purification workflows while maintaining stringent quality standards required for clinical and commercial grade Active Pharmaceutical Ingredients (APIs).
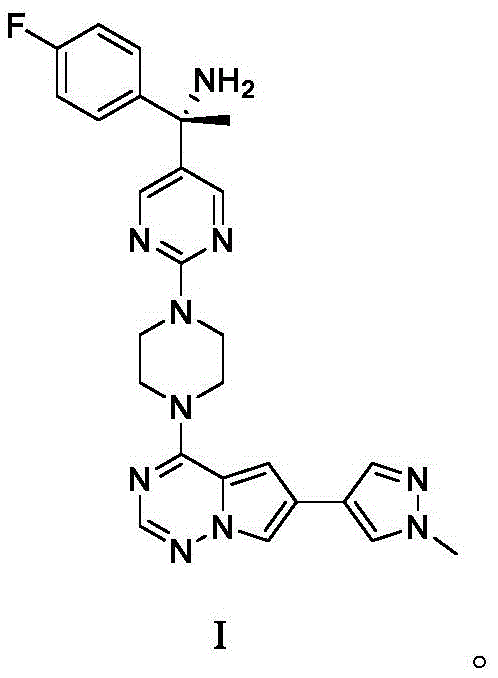
The structural complexity of Avapritinib necessitates a synthesis route that can reliably construct the central heterocyclic core while managing the reactivity of the chiral amine side chain. The patent highlights that traditional methods often suffer from lower selectivity during the coupling of the piperazine-containing fragment with the halogenated heterocycle. The disclosed method overcomes these challenges by temporarily masking the primary amine, thereby altering the electronic environment of the molecule to favor the desired nucleophilic attack. This level of control is essential for R&D directors focused on impurity profiling, as it reduces the burden on downstream purification processes such as chromatography or recrystallization. Furthermore, the use of readily available starting materials ensures that the supply chain remains resilient against raw material shortages, a key consideration for procurement managers evaluating long-term vendor viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic routes for Avapritinib and related kinase inhibitors often encounter significant hurdles regarding chemoselectivity, particularly when dealing with multifunctional intermediates containing both nucleophilic amines and electrophilic halides. In many prior art processes, the direct coupling of an unprotected amine-containing piperazine with a chloro-substituted pyrrolopyrimidine can lead to competitive side reactions. These side reactions may include N-halogenation or the formation of oligomeric byproducts, which complicate the isolation of the target molecule. Such impurities not only reduce the overall yield but also pose severe risks to patient safety if not completely removed, requiring extensive and costly purification steps. Additionally, harsh reaction conditions sometimes employed to drive these sluggish couplings can lead to racemization of chiral centers, further compromising the quality of the final API. For supply chain heads, these inefficiencies translate into longer production cycles and higher waste generation, impacting both cost and environmental compliance metrics.
The Novel Approach
The novel approach detailed in patent CN110950872A fundamentally shifts the synthetic sequence by introducing a protective group strategy at the earliest feasible stage. By reacting the starting amine intermediate with di-tert-butyl dicarbonate ((Boc)2O), the process generates a stable carbamate derivative that is inert to the subsequent coupling conditions. This modification effectively 'switches off' the reactivity of the amine nitrogen, ensuring that the nucleophilic attack occurs exclusively at the intended piperazine nitrogen or prevents unwanted interactions at the amine site during the coupling with the chloro-heterocycle. The result is a dramatic improvement in reaction selectivity, allowing the coupling to proceed smoothly under mild conditions, typically at room temperature in THF. This methodological shift not only simplifies the reaction profile but also enhances the robustness of the process, making it highly suitable for scale-up operations where consistency is key. The ability to avoid halogenated byproducts on the imine moiety specifically addresses a known pain point in the synthesis of this chemical class.
Mechanistic Insights into Boc-Protection and Nucleophilic Substitution
The mechanistic advantage of this route lies in the temporal control of functional group reactivity. In the first step, the primary amine of Compound 1 reacts with (Boc)2O in the presence of a mild base like sodium bicarbonate. This reaction proceeds through a nucleophilic attack of the amine on the carbonyl carbon of the anhydride, releasing tert-butanol and forming the tert-butyl carbamate (Boc-amine). This protecting group is bulky and electron-withdrawing, which significantly reduces the nucleophilicity of the nitrogen atom. Consequently, when this protected intermediate (Compound 2) is subjected to the coupling reaction with Compound 3 (the chloro-pyrrolopyrimidine derivative), the risk of the amine participating in side reactions is nullified. The coupling itself is a classic nucleophilic aromatic substitution (SnAr) or metal-catalyzed cross-coupling (depending on the specific activation of Compound 3, though the patent implies direct displacement or catalyzed coupling facilitated by DIPEA). The base DIPEA serves to scavenge the hydrochloric acid generated during the displacement of the chloride leaving group, driving the equilibrium towards the product Compound 4.
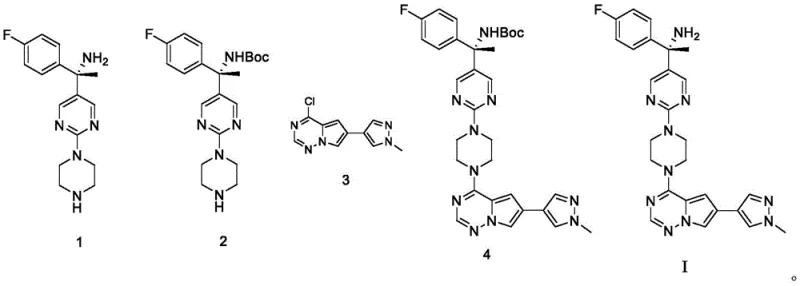
Impurity control is intrinsically built into this mechanism. Without the Boc group, the free amine could potentially act as a competing nucleophile or react with the electrophilic chlorine on the heterocycle to form undesired regioisomers. Moreover, the patent notes that this sequence avoids generating halogenated byproducts on the imine, suggesting that the protection prevents oxidative or electrophilic degradation of the sensitive imine-like structures within the scaffold. The final deprotection step utilizes hydrochloric acid in dioxane, a standard and efficient method for cleaving Boc groups. This acidolysis proceeds via the protonation of the carbamate oxygen, followed by the elimination of isobutylene and carbon dioxide, regenerating the free amine as its hydrochloride salt, which is then neutralized to yield the free base Avapritinib. This clean cleavage mechanism ensures that no residual protecting group fragments remain in the final product, contributing to the high purity levels observed in the examples.
How to Synthesize Avapritinib Efficiently
The synthesis of Avapritinib according to this patent involves a streamlined three-step sequence that balances operational simplicity with high chemical efficiency. The process begins with the protection of the chiral amine, followed by the construction of the core linkage, and concludes with the removal of the protecting group. Each step utilizes common organic solvents and reagents that are readily available on the global chemical market, reducing procurement risks. The reaction conditions are mild, generally operating between 0°C and room temperature, which minimizes energy consumption and thermal hazards. For process chemists, the workup procedures described, such as aqueous washes and standard column chromatography, are well-established unit operations that can be easily adapted for larger scale manufacturing. The following guide summarizes the critical operational parameters derived from the patent examples to ensure successful replication of this high-yielding route.
- React the starting amine intermediate (Compound 1) with di-tert-butyl dicarbonate ((Boc)2O) in DCM and aqueous NaHCO3 to form the protected Compound 2.
- Couple Compound 2 with the chloro-pyrrolopyrimidine derivative (Compound 3) using DIPEA in THF at room temperature to generate Compound 4.
- Remove the Boc protecting group from Compound 4 using hydrochloric acid in dioxane to yield the final Avapritinib product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial advantages for procurement managers and supply chain leaders tasked with securing reliable sources of high-value oncology APIs. The primary benefit stems from the significant simplification of the purification process. By effectively suppressing the formation of halogenated and imine-based byproducts, the method reduces the complexity of the crude reaction mixture. This directly translates to reduced solvent consumption and shorter processing times during isolation and purification, which are major cost drivers in API manufacturing. Furthermore, the avoidance of transition metal catalysts in the described steps (relying instead on organic bases and standard reagents) eliminates the need for expensive and time-consuming heavy metal scavenging processes. This not only lowers the cost of goods sold (COGS) but also simplifies regulatory filings related to elemental impurities, accelerating time-to-market for generic or new formulations.
- Cost Reduction in Manufacturing: The strategic use of Boc protection leads to a drastic improvement in reaction selectivity, which inherently boosts the yield of the desired intermediate and final product. Higher yields mean less raw material is wasted per kilogram of output, providing a direct material cost saving. Additionally, the use of mild reaction conditions and common solvents like DCM and THF allows for the utilization of standard reactor infrastructure without the need for specialized high-pressure or cryogenic equipment. The elimination of complex byproduct removal steps further reduces the operational expenditure associated with waste disposal and solvent recovery, creating a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: The starting materials identified in this route, such as the amine intermediate and the chloro-heterocycle, are structurally simple and can be sourced from multiple established chemical suppliers. This diversification of the supply base mitigates the risk of single-source dependency, a critical factor for maintaining continuous production schedules. The robustness of the chemistry, characterized by its tolerance to mild variations in temperature and stoichiometry, ensures consistent batch-to-batch quality. For supply chain heads, this reliability means fewer production delays due to failed batches or out-of-specification results, ensuring a steady flow of material to meet downstream formulation demands.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction vessels and separation techniques that are standard in the fine chemical industry. The absence of hazardous reagents or extreme conditions facilitates a smoother technology transfer from laboratory to pilot and commercial plant scales. From an environmental standpoint, the improved atom economy and reduced solvent usage contribute to a lower E-factor (mass of waste per mass of product). This aligns with increasingly stringent global environmental regulations and corporate sustainability goals, making the manufacturer a more attractive partner for eco-conscious pharmaceutical companies seeking to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Avapritinib. These answers are derived directly from the experimental data and technical disclosures within patent CN110950872A, providing clarity on the process capabilities and quality attributes. Understanding these details is crucial for stakeholders evaluating the feasibility of this route for commercial production. The responses cover aspects of reaction mechanism, impurity control, and operational parameters to support informed decision-making.
Q: Why is Boc protection used before the coupling reaction in this Avapritinib synthesis?
A: The Boc protection step is critical to prevent unwanted halogenation byproducts on the imine nitrogen and to improve the selectivity of the subsequent nucleophilic substitution reaction, thereby increasing overall yield.
Q: What are the key solvents used in the patented process for Avapritinib?
A: The process utilizes common industrial solvents including Dichloromethane (DCM) for the protection step, Tetrahydrofuran (THF) for the coupling reaction, and Dioxane for the final deprotection, ensuring scalability and ease of recovery.
Q: How does this method improve impurity control compared to conventional routes?
A: By masking the reactive amine group early in the sequence, the method effectively avoids the formation of difficult-to-remove halogenated impurities, resulting in a final product with HPLC purity exceeding 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avapritinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of oncology drug development. Our team of expert process chemists has extensively analyzed the methodology presented in CN110950872A and possesses the technical capability to implement this Boc-protection strategy with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch of Avapritinib meets stringent purity specifications, including tight control over genotoxic impurities and residual solvents.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis for your supply chain needs. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal for the commercial supply of high-purity Avapritinib. Let us help you secure a reliable, cost-effective, and compliant source for this vital anticancer agent.
