Scalable Manufacturing of Avapritinib Intermediates Using Novel Rhodium-Catalyzed Asymmetric Addition
Scalable Manufacturing of Avapritinib Intermediates Using Novel Rhodium-Catalyzed Asymmetric Addition
The pharmaceutical landscape for oncology treatments has been significantly advanced by the development of Avapritinib, a precision therapy approved for treating gastrointestinal stromal tumors (GIST) with specific PDGFRA exon 18 mutations. As demand for this critical medication grows globally, the efficiency and safety of its supply chain become paramount. A pivotal advancement in this domain is detailed in Chinese patent CN111454217A, which discloses a robust preparation method for the key chiral intermediate, (S)-1-(4-fluorophenyl)-1-[2-(piperazine-1-yl)pyrimidine-5-yl]ethylamine. This patent represents a strategic shift from hazardous traditional methodologies to a greener, organometallic-catalyzed approach. By leveraging a novel rhodium-catalyzed asymmetric addition reaction, this technology addresses long-standing challenges in stereochemical control and process safety. For procurement leaders and R&D directors seeking a reliable pharmaceutical intermediates supplier, understanding this technological leap is essential for securing a stable, high-quality supply of this complex API precursor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amines like the Avapritinib intermediate has relied heavily on the addition of Grignard reagents to chiral sulfinyl imines. While effective on a small laboratory scale, this conventional pathway presents severe drawbacks for industrial manufacturing. The use of Grignard reagents necessitates strictly anhydrous conditions and often requires cryogenic temperatures to maintain selectivity and prevent side reactions, leading to substantial energy consumption and operational complexity. Furthermore, the preparation of chiral sulfinyl imines often involves multiple steps with expensive chiral auxiliaries that must be recovered and recycled, adding to the overall process mass intensity. The inherent instability of these reagents poses significant safety risks during scale-up, including potential thermal runaways. These factors collectively result in a fragile supply chain with higher production costs and extended lead times, making the conventional route less attractive for cost reduction in pharmaceutical intermediates manufacturing.
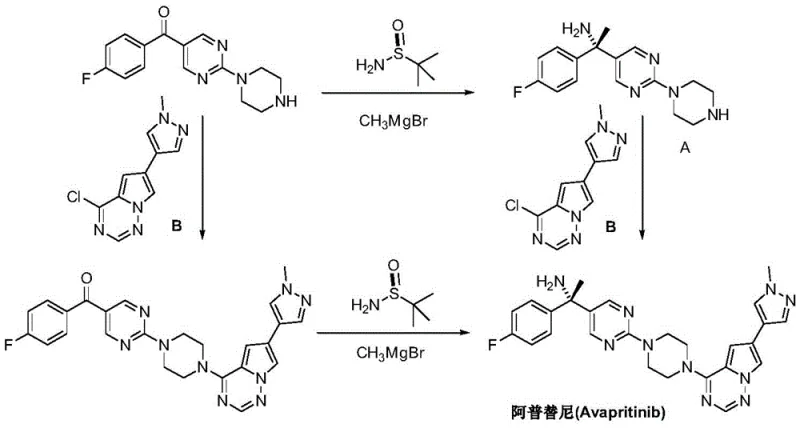
The Novel Approach
In stark contrast, the methodology outlined in patent CN111454217A introduces a streamlined three-step sequence that bypasses the hazards of Grignard chemistry. The process initiates with a straightforward imidization of 4-fluoroacetophenone, followed by a highly stereoselective addition reaction utilizing a stable organoboron species—specifically 2-(1-piperazinyl)pyrimidine-5-potassium trifluoroborate. This substitution of reactive organomagnesium compounds with robust organoboron salts is a game-changer for process safety. The reaction is mediated by a sophisticated chiral rhodium catalyst, which ensures the precise formation of the S-configuration without the need for extreme temperatures. Finally, a mild hydrolytic deprotection step yields the target amine. This route not only simplifies the operational workflow but also significantly enhances the environmental profile of the synthesis by reducing hazardous waste generation.
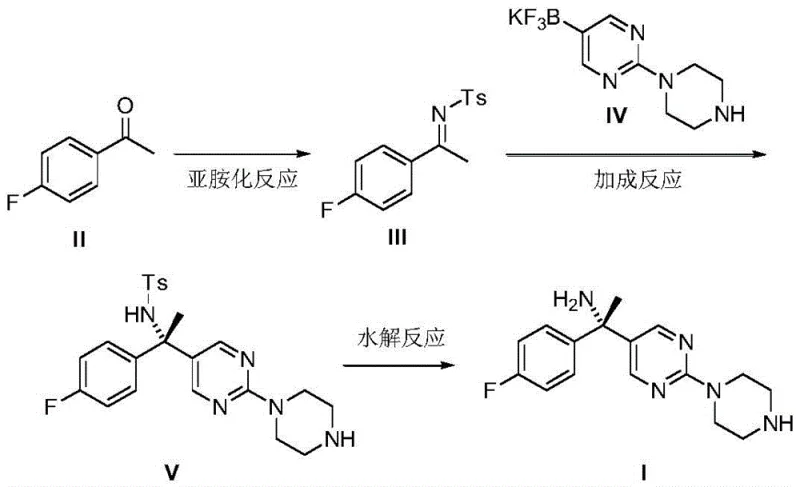
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Addition
The core innovation of this patent lies in the mechanistic elegance of the rhodium-catalyzed 1,2-addition. The catalyst employed, bis[(2,3,5,6-η)-(1R,4R)-2,5-2, 5-bis(4-trifluoromethylphenyl)bicyclo[2.2.2]octane-2-diene]bis-mu-rhodium chloride complex, features a bulky, chiral ligand system. During the reaction, this catalyst coordinates with the nitrogen atom of the imine substrate and the carbon of the organoboron reagent. The chiral environment created by the ligand forces the nucleophilic attack to occur from a specific face of the imine double bond, thereby exerting powerful stereocontrol. This mechanism effectively locks the stereochemistry into the desired S-configuration with high fidelity. Unlike non-catalytic methods that might produce racemic mixtures requiring costly chiral chromatography for separation, this catalytic approach builds the chirality directly into the molecular skeleton. This "chiral pool" strategy minimizes material loss and maximizes the optical purity of the final product, which is a critical quality attribute for any high-purity pharmaceutical intermediate.
Furthermore, the subsequent hydrolysis step is engineered for efficiency. The use of thiophenol as a co-catalyst alongside a mild base like potassium carbonate facilitates the cleavage of the sulfonyl group under relatively gentle conditions (30°C). This prevents the racemization of the sensitive chiral center, a common pitfall in harsh acidic or basic hydrolysis conditions. The combination of a highly selective catalytic addition and a gentle deprotection strategy ensures that the impurity profile of the final intermediate remains exceptionally clean. For R&D teams, this implies a more robust process with fewer purification bottlenecks, directly translating to higher throughput and consistent batch-to-batch quality in the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize (S)-1-(4-fluorophenyl)-1-[2-(piperazine-1-yl)pyrimidine-5-yl]ethylamine Efficiently
The synthesis protocol described in the patent offers a clear roadmap for laboratories and pilot plants aiming to adopt this superior methodology. The process begins with the condensation of readily available 4-fluoroacetophenone and p-toluenesulfonamide in toluene, driven by p-toluenesulfonic acid at reflux temperatures to form the imine intermediate. This is followed by the critical asymmetric addition step in dioxane, where the organoboron reagent and the chiral rhodium catalyst are combined at 80°C. The final step involves treating the protected amine with potassium carbonate and thiophenol in DMF to reveal the free amine. Each step utilizes common industrial solvents and reagents, avoiding the need for specialized equipment required for cryogenic or ultra-high vacuum processes. The detailed standardized synthesis steps for implementing this route are provided below.
- Perform imidization of 4-fluoroacetophenone with p-toluenesulfonamide using p-toluenesulfonic acid catalyst in toluene at 110°C.
- Execute stereoselective addition using a chiral rhodium complex catalyst and 2-(1-piperazinyl)pyrimidine-5-potassium trifluoroborate in dioxane at 80°C.
- Conduct alkaline hydrolysis using potassium carbonate and thiophenol catalyst in DMF at 30°C to remove the sulfonyl protecting group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of raw material handling. By eliminating the need for pyrophoric Grignard reagents, facilities can reduce their safety infrastructure costs and insurance premiums associated with hazardous chemical storage. The stability of the potassium trifluoroborate salt allows for bulk purchasing and longer shelf-life inventory management, mitigating the risk of supply disruptions caused by reagent degradation. This stability is a key factor in reducing lead time for high-purity pharmaceutical intermediates, as it decouples production schedules from the immediate availability of freshly prepared reactive species.
- Cost Reduction in Manufacturing: The economic implications of this process are profound. The elimination of cryogenic cooling systems for the addition step results in significant energy savings. Additionally, the high stereoselectivity of the rhodium catalyst reduces the burden on downstream purification processes such as chiral resolution or recrystallization, which are often the most expensive unit operations in API manufacturing. The use of a catalytic amount of rhodium, despite the metal's cost, is offset by the high turnover and the avoidance of stoichiometric chiral auxiliaries. Consequently, the overall cost of goods sold (COGS) for the intermediate is optimized through process intensification and waste minimization.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commodity chemicals. Starting materials like 4-fluoroacetophenone and p-toluenesulfonamide are widely produced and available from multiple global vendors, reducing dependency on single-source suppliers. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or humidity, leading to higher first-pass yields and more predictable production timelines. This reliability is crucial for maintaining the inventory levels required to support the continuous manufacturing of the final Avapritinib drug product.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route aligns perfectly with modern green chemistry principles. The replacement of volatile ether solvents often used in Grignard reactions with higher boiling point solvents like dioxane and DMF reduces VOC emissions. Furthermore, the aqueous workup procedures described are straightforward, facilitating easier wastewater treatment. The process is inherently safer to scale from kilogram to multi-ton quantities, as the exothermic risks associated with organometallic additions are significantly mitigated. This makes the technology ideal for commercial scale-up in regulated environments where environmental compliance is strictly enforced.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within patent CN111454217A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this intermediate into their existing supply networks.
Q: Why is the rhodium-catalyzed organoboron addition superior to traditional Grignard methods?
A: Traditional methods rely on hazardous Grignard reagents which require strict anhydrous conditions and cryogenic temperatures. The patented rhodium-catalyzed route utilizes stable potassium trifluoroborate salts, allowing for milder reaction conditions (80°C) and significantly improved operational safety and handling.
Q: How is the S-configuration chirality controlled in this synthesis?
A: Chirality is introduced during the addition step using a specialized chiral rhodium catalyst, specifically bis[(2,3,5,6-η)-(1R,4R)-2,5-2, 5-bis(4-trifluoromethylphenyl)bicyclo[2.2.2]octane-2-diene]bis-mu-rhodium chloride complex. This catalyst induces high stereoselectivity, ensuring the formation of the required S-enantiomer without the need for difficult resolution steps.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It utilizes common solvents like toluene, dioxane, and DMF, and operates at moderate temperatures ranging from 30°C to 110°C. The use of stable organoboron reagents and efficient hydrolytic deprotection simplifies downstream processing and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avapritinib Intermediate Supplier
The technological advancements presented in patent CN111454217A underscore the evolving complexity of oncology drug manufacturing. At NINGBO INNO PHARMCHEM, we recognize that accessing such cutting-edge synthetic routes requires a partner with deep technical expertise and robust manufacturing capabilities. As a dedicated CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of organometallic catalysis, including the safe handling of rhodium complexes and the rigorous purification needed to meet stringent purity specifications. Our rigorous QC labs ensure that every batch of Avapritinib intermediate delivered meets the highest international standards for chiral purity and chemical identity.
We invite global pharmaceutical partners to collaborate with us to leverage this efficient synthesis technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for this critical cancer therapy intermediate, ensuring that life-saving medications reach patients without delay.
