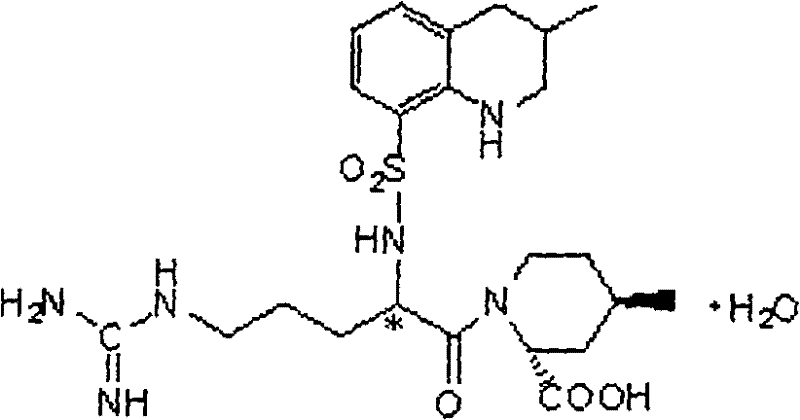
Scalable Synthesis of Argatroban Intermediates: Overcoming Moisture Control Barriers in API Manufacturing
The global pharmaceutical landscape continues to demand more efficient and robust synthetic routes for critical anticoagulant therapies, particularly for Argatroban, a direct thrombin inhibitor essential for treating acute ischemic cerebral apoplexy. Patent CN101348463B introduces a groundbreaking refinement in the manufacturing of the key intermediate, N-(3-methyl-8-quinoline sulfonyl)-N-nitryl-L-arginine, addressing the longstanding bottleneck of moisture sensitivity in amidation reactions. This technical breakthrough is pivotal for any reliable pharmaceutical intermediates supplier aiming to secure the supply chain for this life-saving medication. The invention specifically targets the post-treatment of the condensation between N-nitryl-L-arginine and 3-methyl-8-quinoline sulfonyl chloride, solving the thorny issue of water removal that has historically plagued industrial scale-up efforts. By implementing a novel two-stage drying protocol, the process ensures water content is rigorously controlled below 0.2 percent, a threshold previously difficult to achieve without risking thermal decomposition of the product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Argatroban intermediates has been fraught with operational complexities that hinder cost reduction in API manufacturing. Prior art methods, such as those described in earlier patents, often relied on isolating the intermediate N-(3-methyl-8-quinoline sulfonyl)-N-nitryl-L-arginine as a viscous syrup. This physical state presented a nightmare for process engineers, as removing trace water from a hygroscopic syrup typically required prolonged vacuum distillation at elevated temperatures. Such harsh conditions not only consumed excessive energy but also significantly increased the risk of thermal decomposition, leading to lower overall yields and a darker, more difficult-to-purify crude product. Furthermore, the requirement for absolute anhydrous conditions meant that even minor deviations in vacuum tightness or heating duration could result in batch failures, creating unacceptable variability for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
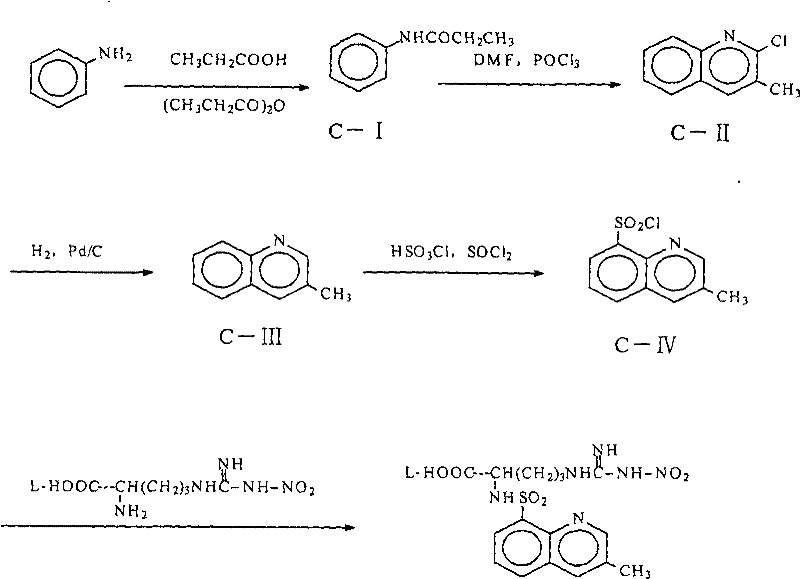
The methodology disclosed in CN101348463B represents a paradigm shift by abandoning the isolation of the syrupy intermediate in favor of a streamlined solution-phase processing technique. Instead of relying solely on thermal vacuum stripping, the inventors introduced a sophisticated extraction and drying sequence involving tetrahydrofuran (THF) and inorganic salts. The process cleverly utilizes pH adjustment and salt saturation to drive the product into the organic phase while leaving inorganic impurities behind. Crucially, the innovation lies in the sequential drying strategy: an initial drying with standard desiccants like anhydrous magnesium sulfate reduces bulk water, followed by a polishing step using activated molecular sieves. This approach effectively drives the residual water content down to the critical sub-0.2 percent level required for the subsequent acylation step, all while maintaining the integrity of the thermally sensitive arginine derivative.
Mechanistic Insights into Moisture-Controlled Amidation
To fully appreciate the technical merit of this invention, one must understand the underlying chemical kinetics governing the amidation reaction. The core transformation involves the nucleophilic attack of the alpha-amino group of the nitro-arginine derivative onto the sulfur atom of the 3-methyl-8-quinoline sulfonyl chloride. This reaction is inherently competitive; water molecules act as potent nucleophiles that can hydrolyze the sulfonyl chloride starting material into the corresponding sulfonic acid, a dead-end byproduct that consumes reagents and complicates purification. Therefore, the high-purity pharmaceutical intermediates required for GMP production depend entirely on suppressing this hydrolysis pathway. The patent's specific instruction to measure THF water content and ensure it remains below 1 percent before adding molecular sieves is not arbitrary; it prevents the premature saturation of the molecular sieves, allowing them to function efficiently in the final polishing stage to reach the stringent < 0.2 percent target.
Furthermore, the choice of reaction media and pH control plays a vital role in the selectivity of the process. The initial reaction in aqueous sodium carbonate ensures that the amino group is sufficiently deprotonated to act as a nucleophile, yet the pH is carefully managed to prevent the degradation of the guanidine moiety present in the arginine side chain. The subsequent extraction into THF serves a dual purpose: it separates the organic-soluble product from the aqueous salt waste, and it provides a solvent environment compatible with the downstream coupling with (2R, 4R)-4-methyl-pipecolic acid ethyl ester. This seamless transition between aqueous workup and anhydrous coupling is the hallmark of a well-designed industrial process, minimizing unit operations and maximizing throughput.
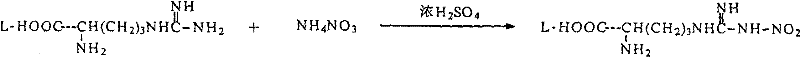
How to Synthesize N-(3-methyl-8-quinoline sulfonyl)-N-nitryl-L-arginine Efficiently
The execution of this synthesis requires precise adherence to the drying protocols to guarantee success. The process begins with the preparation of the nitro-arginine precursor, followed by its coupling with the quinoline sulfonyl chloride under biphasic conditions. The critical path involves the rigorous removal of water from the THF extract before proceeding to the final amide bond formation. Operators must monitor water content analytically at each stage to ensure the molecular sieves are not overloaded. For a detailed breakdown of the specific reagent quantities, temperature profiles, and stirring times required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Prepare N-nitro-L-arginine by reacting L-arginine with ammonium nitrate in concentrated sulfuric acid, followed by neutralization and isolation.
- React N-nitro-L-arginine with 3-methyl-8-quinoline sulfonyl chloride in an aqueous sodium carbonate solution, followed by extraction into THF.
- Execute a critical two-stage drying process: first with anhydrous salts (e.g., MgSO4) to reduce water below 1%, then with 4A molecular sieves to achieve <0.2% moisture before final coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical elegance. By eliminating the need for high-vacuum distillation of heat-sensitive syrups, the process drastically simplifies the equipment requirements, allowing for the use of standard glass-lined reactors rather than specialized thin-film evaporators. This reduction in capital expenditure and maintenance complexity translates directly into a more resilient supply chain, as the process is less susceptible to equipment failure or bottlenecks. Moreover, the ability to run the reaction in larger batches, as evidenced by the successful 300L pilot trials mentioned in the patent data, suggests that reducing lead time for high-purity pharmaceutical intermediates is achievable through straightforward linear scale-up rather than complex campaign adjustments.
- Cost Reduction in Manufacturing: The elimination of energy-intensive vacuum distillation steps results in significant utility savings per kilogram of product. Additionally, by preventing the thermal decomposition associated with prolonged heating, the process improves the effective yield of the valuable quinoline starting material. This enhancement in atom economy means that less raw material is wasted as byproducts, leading to substantial cost savings in the bill of materials without compromising the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of the two-stage drying protocol ensures consistent batch-to-batch quality, a critical factor for regulatory compliance. Because the process does not rely on pushing equipment to its thermal limits, the risk of unplanned downtime due to reactor fouling or decomposition events is minimized. This reliability allows suppliers to offer more stable delivery schedules, mitigating the risk of stockouts for downstream drug manufacturers who depend on a steady flow of this critical anticoagulant intermediate.
- Scalability and Environmental Compliance: The use of THF and standard inorganic salts aligns well with existing solvent recovery infrastructure found in most modern chemical plants. The process avoids the generation of hazardous phosgene gas, which was a concern in older synthetic routes, thereby improving the overall safety profile and reducing the burden on废气 treatment systems. This greener profile facilitates easier permitting for capacity expansion and aligns with the increasing environmental, social, and governance (ESG) mandates of major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Argatroban intermediates using this advanced moisture-control technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view into the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their broader API synthesis strategies.
Q: Why is strict moisture control critical in this synthesis?
A: The reaction involves a highly reactive sulfonyl chloride intermediate. Trace moisture causes hydrolysis of the sulfonyl chloride, leading to significant yield loss and the formation of sulfonic acid impurities that are difficult to separate.
Q: What yields can be expected from this improved process?
A: According to the patent data, the optimized process achieves isolated yields ranging from 46% to 55% for the key intermediate, with high purity (>83%) and extremely low water content (<0.11%), ensuring successful downstream amidation.
Q: Is this method suitable for large-scale production?
A: Yes, the patent explicitly demonstrates scalability with successful trials conducted in 300L reactors. The elimination of complex vacuum distillation steps for syrupy intermediates makes it highly viable for industrial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(3-methyl-8-quinoline sulfonyl)-N-nitryl-L-arginine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate moisture-control protocols described in CN101348463B are executed with precision at every scale. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities and moisture levels, guaranteeing that every batch of intermediate meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to optimize your supply chain for Argatroban production. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our refined process can lower your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to quality and efficiency can support your long-term commercial goals.
