Advanced Purification Technology for Commercial Scale Argatroban Production
The pharmaceutical landscape for thrombin inhibitors demands exceptionally high purity standards to ensure patient safety and therapeutic efficacy. Patent CN102408468A introduces a groundbreaking purification methodology for Argatroban, a potent synthetic antithrombotic agent, addressing critical limitations found in earlier manufacturing protocols. This technical disclosure outlines a sophisticated three-step process involving strong-base ion exchange resin adsorption, catalytic hydrogenation, and controlled recrystallization. By shifting away from hazardous traditional reagents, this innovation not only enhances the chemical profile of the final active pharmaceutical ingredient but also aligns with modern green chemistry principles. For global procurement and R&D teams, understanding this patent is essential for securing a reliable Argatroban supplier capable of delivering material with purity not lower than 99.5% and minimal heavy metal contamination.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Argatroban has relied on routes that present significant safety and operational challenges for large-scale manufacturing. Prior art, including patents such as EP8746 and US4258192, often necessitates the use of highly toxic phosgene gas and requires strict nitrogen protection throughout the reaction sequence. These conditions impose severe engineering constraints, increasing the complexity of the reactor setup and the risk profile for plant operators. Furthermore, alternative pathways utilizing phosphorus oxychloride (POCl3) or phosphorus trichloride introduce corrosive hazards and pungent odors that complicate waste management and environmental compliance. The moisture sensitivity of these reagents often leads to side reactions, resulting in lower overall yields and the formation of difficult-to-remove diastereomeric impurities. Consequently, the final product from these conventional methods frequently suffers from inconsistent purity and color issues, necessitating costly and inefficient downstream purification steps like repeated column chromatography which are not viable for ton-scale production.
The Novel Approach
In stark contrast, the methodology disclosed in CN102408468A offers a streamlined and safer alternative that fundamentally reengineers the purification workflow. By leveraging the selective adsorption properties of strong-base ion exchange resins, the process effectively captures the target molecule while excluding a wide range of organic and inorganic impurities at the initial stage. This is followed by a catalytic hydrogenation step using palladium carbon, which serves a dual purpose of reducing specific impurities and scavenging heavy metals that are critical quality attributes for injectable formulations. The final recrystallization from purified water eliminates the need for large volumes of organic solvents, reducing both cost and environmental impact. This approach transforms the production of this complex pharmaceutical intermediate from a hazardous, low-yield operation into a robust, high-efficiency process suitable for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ion Exchange and Catalytic Purification
The core of this technological advancement lies in the synergistic application of ion exchange chromatography and heterogeneous catalysis. The strong-base anion exchange resin, such as the A850 or A500P types mentioned, functions through a quaternary amine base structure that dissociates hydroxide ions to create a highly alkaline environment locally. When the crude Argatroban solution passes through the resin bed, the carboxylic acid moiety of the Argatroban molecule interacts with the positively charged groups on the resin matrix. This interaction allows for the selective retention of the product while allowing neutral organic impurities and certain colored byproducts to pass through or be washed away. The subsequent elution with a controlled concentration of alkaline solution, such as 4% to 10% sodium hydroxide, releases the Argatroban in a significantly purified salt form. This step is crucial for removing bacterial endotoxins and inorganic salts that often co-precipitate in traditional isolation methods, thereby laying the foundation for the high purity specifications required by regulatory bodies.
Following the ion exchange step, the solution undergoes a critical neutralization and hydrogenation phase. Adjusting the pH to a range of 3 to 6, preferably between 4.5 and 5.5 using acetic acid, prepares the molecule for the subsequent catalytic treatment. The addition of palladium carbon under hydrogen pressure (3.92 to 5 Mpa) at elevated temperatures (50 to 80°C) facilitates the reduction of trace unsaturated impurities and, more importantly, acts as a scavenger for heavy metal ions. The hot filtration step immediately following hydrogenation ensures that the palladium catalyst and any insoluble particulate matter are removed before crystallization begins. This mechanism is vital for achieving the extremely low heavy metal content (e.g., 4-5 ppm) reported in the examples. Finally, the controlled addition of purified water and slow cooling promotes the formation of a stable crystal lattice, excluding remaining soluble impurities and yielding the monohydrate form which is then dried to obtain the pure anhydrous or stable hydrate compound.
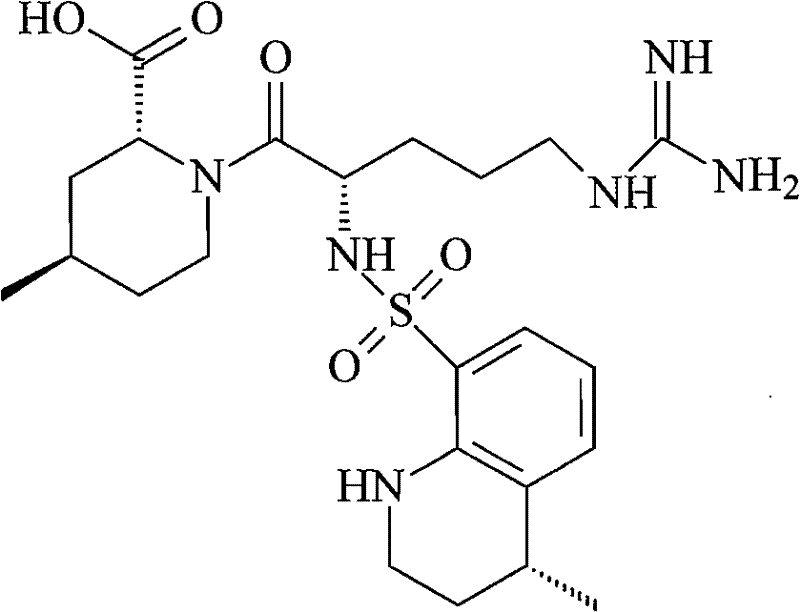
How to Synthesize Argatroban Efficiently
Implementing this purification strategy requires precise control over reaction parameters to maximize yield and quality. The process begins with the preparation of the ion exchange column, followed by the careful loading of the crude material. Operators must monitor the pH and temperature during the neutralization and hydrogenation steps closely, as deviations can impact the efficiency of impurity removal. The recrystallization phase demands a slow cooling profile to ensure the growth of large, pure crystals rather than amorphous precipitates. For detailed operational parameters and specific equipment configurations, please refer to the standardized synthesis steps provided in the technical guide below.
- Adsorb crude Argatroban using strong-base ion exchange resin, elute with alkaline solution, and concentrate under reduced pressure.
- Neutralize the eluent with acid to pH 3-6, add palladium carbon for catalytic hydrogenation, and filter while hot to remove insolubles.
- Add purified water to the solution, cool slowly to room temperature for recrystallization, and dry at 60-80°C to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this purification technology offers substantial benefits for supply chain stability and cost management. By eliminating the need for hazardous reagents like phosgene and phosphorus oxychloride, manufacturers can significantly reduce the costs associated with specialized safety equipment, hazardous waste disposal, and regulatory compliance. The simplification of the process flow also translates to shorter production cycles and higher throughput, enhancing the overall reliability of the supply chain. Furthermore, the use of water as a primary recrystallization solvent drastically cuts down on the consumption of expensive organic solvents, contributing to significant cost savings in raw material procurement. These factors combined make the production of high-purity Argatroban more economically viable and less susceptible to disruptions caused by environmental regulations or raw material shortages.
- Cost Reduction in Manufacturing: The elimination of toxic and corrosive reagents removes the need for expensive corrosion-resistant reactors and complex gas scrubbing systems. Additionally, the high yield reported in the patent examples, exceeding 93%, means less raw material is wasted per unit of final product. This efficiency directly lowers the cost of goods sold (COGS) and allows for more competitive pricing structures in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available ion exchange resins and standard hydrogenation equipment reduces dependency on specialized or scarce reagents. This accessibility ensures that production can be maintained consistently even during periods of raw material volatility. The robustness of the purification steps also minimizes the risk of batch failures due to impurity profiles, ensuring a steady flow of material to downstream formulation partners and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing unit operations that are common in fine chemical manufacturing. The reduction in organic solvent usage and the avoidance of hazardous gas generation align with increasingly strict environmental regulations worldwide. This compliance reduces the risk of production shutdowns due to environmental violations and positions the supply chain as a sustainable partner for green-conscious pharmaceutical companies seeking cost reduction in pharmaceutical intermediates manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this purification technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential partners.
Q: How does this purification method improve upon conventional synthesis routes?
A: Conventional routes often utilize toxic phosgene or corrosive phosphorus oxychloride, leading to complex operations and lower yields. This patented method replaces hazardous reagents with ion exchange and catalytic hydrogenation, significantly simplifying the process while achieving purity levels exceeding 99.5%.
Q: What is the effectiveness of heavy metal removal in this process?
A: The process incorporates a specific catalytic hydrogenation step using palladium carbon followed by hot filtration. This mechanism effectively reduces heavy metal content to extremely low levels, typically around 4-5 ppm, meeting stringent pharmaceutical safety standards.
Q: Is this purification method suitable for industrial scale-up?
A: Yes, the method is designed for industrial applicability. It utilizes commercially available strong-base ion exchange resins and standard hydrogenation equipment, demonstrating high yields of over 93% and consistent quality suitable for mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Argatroban Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of thrombin inhibitors like Argatroban. Our technical team has extensively analyzed the pathways described in CN102408468A and possesses the expertise to implement these advanced purification protocols at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying heavy metal content and diastereomeric ratios to meet the most demanding global pharmacopoeia standards.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for this vital anticoagulant. By leveraging our technical capabilities, you can secure a source of material that balances high quality with commercial viability. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing processes can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
