Advanced Cefathiamidine Manufacturing: Cost-Effective Chloroacetyl Chloride Route for Global Supply Chains
The pharmaceutical landscape for first-generation cephalosporins is constantly evolving, driven by the need for more cost-effective and stable manufacturing processes. Patent CN101486718B introduces a transformative methodology for the preparation of cefathiamidine, a broad-spectrum antibiotic known for its potent activity against gram-positive organisms and enterococci. This innovation specifically addresses the long-standing economic and quality bottlenecks associated with traditional synthesis routes by substituting the expensive bromoacetyl bromide with the significantly more economical chloroacetyl chloride. For R&D directors and procurement strategists, this shift represents a critical opportunity to optimize the cost of goods sold (COGS) without compromising the stringent purity specifications required for injectable formulations. The technical breakthrough lies not merely in the raw material swap, but in the sophisticated optimization of reaction conditions and catalytic systems that make the less reactive chlorine species viable for large-scale production.
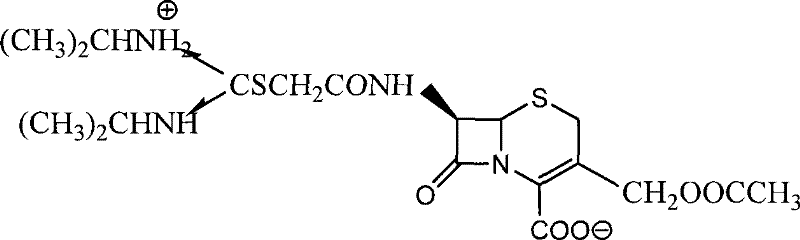
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial standard for synthesizing cefathiamidine has relied heavily on bromoacetyl bromide as the acylating agent for 7-amino-cephalosporanic acid (7-ACA). While the high reactivity of the bromine atom facilitates a straightforward condensation reaction with thiourea derivatives, this conventional pathway suffers from severe economic and quality drawbacks. The primary constraint is the exorbitant cost of bromoacetyl bromide, which acts as a major driver of raw material expenses in the final API pricing structure. Furthermore, products synthesized via the brominated route are prone to stability issues, specifically regarding coloration; over time, typically within three months of storage at ambient temperatures, the product color deepens significantly, indicating degradation or impurity formation that complicates inventory management and shelf-life validation. These factors collectively burden the supply chain with higher carrying costs and stricter quality control requirements.
The Novel Approach
The novel approach detailed in the patent data fundamentally re-engineers the synthesis by leveraging chloroacetyl chloride, a congener that is chemically similar but economically superior. By successfully adapting the reaction conditions to accommodate the lower nucleophilic leaving group ability of chlorine, this method achieves a drastic reduction in raw material costs, as chloroacetyl chloride is priced at merely one-sixth of its brominated counterpart. Beyond the immediate financial benefits, the elimination of alkali addition in the second reaction step—where the intermediate reacts with N,N-di-isopropyl thiourea—results in a product with markedly lighter color and enhanced stability. This improvement in the impurity profile ensures that the cefathiamidine produced is more conducive to long-term storage and transportation, thereby reducing waste and enhancing the reliability of the supply chain for global distribution networks.
Mechanistic Insights into Catalytic Halogen Activation
The core technical challenge in utilizing chloroacetyl chloride lies in the inherently lower reactivity of the carbon-chlorine bond compared to the carbon-bromine bond, which traditionally makes the subsequent substitution with thiourea kinetically sluggish and difficult to drive to completion. To overcome this barrier, the patented process employs a strategic catalytic system involving sodium iodide dissolved in a mixed solvent of tetrahydrofuran (THF) and N,N-dimethylacetamide (DMA). Mechanistically, the iodide ion acts as a potent nucleophilic catalyst, likely facilitating an in-situ Finkelstein-type halogen exchange where the chloro-intermediate is transiently converted into a more reactive iodo-species. This activated intermediate then undergoes rapid nucleophilic attack by the sulfur atom of the N,N-di-isopropyl thiourea, effectively bypassing the high activation energy barrier associated with direct chlorine displacement. This catalytic cycle allows the reaction to proceed efficiently at moderate temperatures (30-35°C) within a practical timeframe of two hours, ensuring high conversion rates without the need for harsh thermal conditions that could degrade the sensitive beta-lactam ring.
Controlling the impurity profile in beta-lactam antibiotics is paramount, and this mechanism offers distinct advantages in that regard. By avoiding the addition of alkali in the second step, the process minimizes the risk of base-catalyzed hydrolysis of the beta-lactam ring, a common degradation pathway that leads to open-ring impurities and reduced potency. The use of specific crystallization solvents, such as acetone or chloroform, further refines the product by selectively precipitating the desired cefathiamidine while keeping soluble impurities in the mother liquor. The result is a crude product that, upon simple recrystallization from aqueous ethanol, yields a final API with a content exceeding 97% and a color profile that remains stable over extended periods. This robust control over the reaction environment ensures that the final product meets the rigorous standards of the Chinese Pharmacopoeia and international equivalents, providing R&D teams with a reliable and reproducible synthetic route.
How to Synthesize Cefathiamidine Efficiently
The synthesis of cefathiamidine via this optimized chloroacetyl chloride route is designed for scalability and operational simplicity, making it highly attractive for commercial manufacturing. The process begins with the controlled acylation of 7-ACA in a biphasic or mixed solvent system, followed by a catalytic substitution step that leverages inexpensive commodity chemicals. Detailed below is the high-level workflow derived from the patent embodiments, which serves as a foundational guide for process engineers looking to implement this technology. For the complete standardized operating procedures, including precise stirring rates and drying parameters, please refer to the technical documentation provided in the subsequent section.
- Dissolve 7-ACA in a solvent mixture (e.g., acetone/water) with alkali, then add chloroacetyl chloride at controlled temperatures to form chloracetyl 7-ACA.
- Isolate the chloracetyl 7-ACA intermediate by acidification and filtration.
- React the intermediate with N,N-di-isopropyl thiourea in THF/DMA solvent using a sodium iodide catalyst to yield crude cefathiamidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chloroacetyl chloride-based synthesis offers a compelling value proposition centered on cost reduction and supply security. The most immediate impact is the dramatic decrease in raw material expenditure, driven by the substitution of a high-cost specialty reagent with a widely available commodity chemical. This shift not only lowers the direct cost of manufacturing but also mitigates the risk of supply disruptions often associated with specialized brominated reagents. Furthermore, the improved stability of the final product reduces the likelihood of batch rejections due to color specification failures during quality control testing, thereby enhancing overall yield efficiency and reducing waste disposal costs associated with off-spec material.
- Cost Reduction in Manufacturing: The economic argument for this process is anchored in the substantial price differential between the key acylating agents; with chloroacetyl chloride costing approximately one-sixth of bromoacetyl bromide, the potential for margin expansion is significant. Additionally, the process eliminates the need for alkali in the second reaction step, which simplifies the workup procedure and reduces the consumption of auxiliary chemicals and water for washing. This streamlined workflow translates to lower utility costs and reduced labor hours per batch, contributing to a leaner and more cost-efficient production model that can withstand market price fluctuations.
- Enhanced Supply Chain Reliability: From a sourcing perspective, chloroacetyl chloride is a mature, high-volume commodity chemical with a robust global supply network, unlike bromoacetyl bromide which may be subject to tighter supply constraints or regulatory scrutiny. By aligning the production process with readily available feedstocks, manufacturers can secure longer-term supply agreements and reduce the lead time for raw material procurement. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical customers who require just-in-time inventory management.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are mild and operate at near-ambient temperatures, which significantly reduces the energy load required for heating or cryogenic cooling compared to more aggressive synthetic routes. The use of standard organic solvents like THF, DMA, and acetone allows for established recovery and recycling protocols, minimizing the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only supports corporate sustainability goals but also ensures compliance with increasingly stringent environmental regulations, safeguarding the facility against potential operational shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cefathiamidine synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the process capabilities and limitations. Stakeholders are encouraged to review these points to assess the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: Why is chloroacetyl chloride preferred over bromoacetyl bromide for cefathiamidine production?
A: Chloroacetyl chloride offers a significant cost advantage, priced at approximately one-sixth of bromoacetyl bromide. Additionally, the resulting product exhibits superior color stability and quality, avoiding the deepening of color often seen with brominated routes during storage.
Q: How is the lower reactivity of chlorine managed in this synthesis?
A: The process utilizes a catalytic amount of sodium iodide in a polar aprotic solvent system (THF/DMA). This facilitates an in-situ halogen exchange, effectively activating the chloro-intermediate for nucleophilic attack by the thiourea, overcoming the kinetic barriers associated with chlorine.
Q: What purity levels can be achieved with this method?
A: Following recrystallization from ethanol, the process consistently yields cefathiamidine with a content greater than 97% as measured by HPLC, meeting stringent pharmacopoeia standards for color and impurity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefathiamidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes that balance cost efficiency with uncompromising quality standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and risk-mitigated. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cefathiamidine delivered meets the exacting requirements of global regulatory bodies. Our commitment to technical excellence allows us to offer a supply chain partnership that is both resilient and responsive to the dynamic needs of the pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this innovative chloroacetyl chloride route can be tailored to your specific production volumes and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the potential economic benefits specific to your operation. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive value and efficiency in your antibiotic manufacturing supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
