Advanced Purification Technology for High-Purity Cefathiamidine Manufacturing
The pharmaceutical landscape for first-generation cephalosporins continues to evolve, driven by the relentless demand for higher purity profiles and reduced toxicological risks in active pharmaceutical ingredients. Patent CN102010428A introduces a transformative approach to the preparation of Cefathiamidine, a critical antibiotic used extensively for treating respiratory, biliary, and urinary tract infections caused by susceptible organisms such as Staphylococcus aureus and Streptococcus pneumoniae. This proprietary technology addresses the inherent instability of the Cefathiamidine molecule, which exists as a zwitterionic inner salt prone to decomposition under heat, light, and moisture. By shifting away from traditional purification bottlenecks, this method leverages a sophisticated hydrolysis and esterification sequence that not only simplifies the operational workflow but also drastically enhances the final quality of the drug substance, ensuring it meets the rigorous standards required for modern clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of Cefathiamidine has been plagued by significant technical hurdles that compromise both yield and safety. Conventional synthetic routes often rely on the use of bromoacetyl bromide during the build-up process, which poses a severe risk of residual bromide anions remaining in the final product if not managed with extreme precision. Furthermore, traditional purification strategies frequently involve adjusting the solution to the isoelectric point using acids or alkalis, a delicate operation that can easily lead to the destructive opening of the sensitive beta-lactam ring. Such degradation not only lowers the assay content of the crude product but also generates complex impurity profiles that are notoriously difficult to separate. Additionally, older methods often necessitate the use of large volumes of expensive solvents or energy-intensive distillation crystallization processes, resulting in inflated production costs and operational complexity that strain manufacturing efficiency.
The Novel Approach
In stark contrast, the methodology outlined in the patent data presents a robust alternative that circumvents these structural vulnerabilities through a clever two-step transformation. Instead of attempting to purify the unstable final molecule directly, the process first converts the crude material into a more stable intermediate via controlled alkaline hydrolysis. This intermediate can then be subjected to rigorous purification techniques, such as charcoal adsorption in organic solvents, without the fear of immediate degradation. The final step involves a mild re-esterification catalyzed by specific strong acid salts, which restores the active molecular structure with high fidelity. This strategic detour allows for the effective removal of color bodies and trace impurities while preserving the integrity of the core cephalosporin scaffold, ultimately delivering a product with superior stability and therapeutic potential.
Mechanistic Insights into Hydrolysis and Acid Salt-Catalyzed Esterification
The core of this technological advancement lies in the precise manipulation of the C-3' substituent on the cephem nucleus. The process initiates with the dispersion of the crude Cefathiamidine in an aqueous medium, where an alkaline catalyst, preferably sodium hydroxide, facilitates the hydrolysis of the acetoxymethyl group. This reaction converts the ester functionality into a hydroxymethyl group, yielding Intermediate (II), which exhibits different solubility characteristics that facilitate its isolation as a solid. This phase is critical because it breaks the cycle of impurity entrapment; by converting the molecule into a form that can be easily filtered and washed, the process effectively sheds the bulk of non-polar and colored contaminants that typically co-crystallize with the final API.
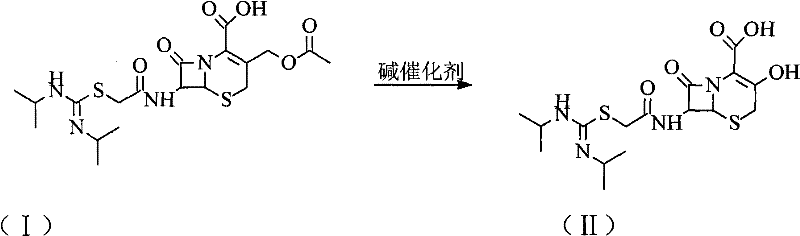
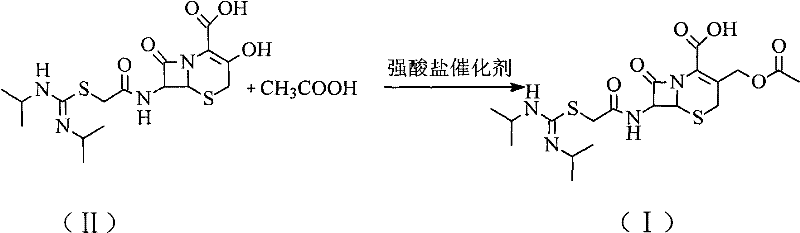
Following the isolation and charcoal treatment of Intermediate (II), the synthesis proceeds to the regeneration of the active drug through a targeted esterification. In this stage, glacial acetic acid serves as both the reactant and the solvent component, while a strong acid salt catalyst, such as ferric sulfate (Fe2(SO4)3·9H2O), drives the reaction forward. The inclusion of a water-carrying agent like cyclohexane is mechanistically vital, as it forms an azeotrope to continuously remove water generated during the esterification, thereby shifting the equilibrium towards product formation according to Le Chatelier's principle. This catalytic system is remarkably gentle compared to traditional strong mineral acids, preventing the protonation-induced ring opening of the beta-lactam moiety while ensuring rapid conversion rates at moderate temperatures.
How to Synthesize Cefathiamidine Efficiently
The implementation of this purification protocol requires strict adherence to temperature controls and reagent stoichiometry to maximize the yield of the high-purity final product. The process is designed to be scalable, moving seamlessly from laboratory validation to industrial production by utilizing standard unit operations such as heated stirring reactors, filtration units, and vacuum drying systems. Operators must ensure that the alkaline hydrolysis step is monitored closely to prevent over-reaction, while the subsequent esterification benefits from the continuous removal of water to drive completion. For a comprehensive breakdown of the specific operating parameters, reagent grades, and safety protocols required to execute this synthesis successfully, please refer to the detailed technical guide provided below.
- Disperse crude Cefathiamidine in water and add an alkaline catalyst such as sodium hydroxide, then heat to 70-80°C to induce hydrolysis and precipitate Intermediate (II).
- Dissolve the isolated Intermediate (II) in an organic solvent like benzyl alcohol and perform charcoal adsorption at 50-60°C to remove impurities, followed by filtration.
- Add glacial acetic acid and a strong acid salt catalyst (e.g., ferric sulfate) to the filtrate along with a water-carrying agent like cyclohexane, reacting at 40-50°C to regenerate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel purification route offers substantial strategic benefits that extend far beyond simple chemical yield improvements. By eliminating the need for complex and hazardous reagents like bromoacetyl bromide in the final stages, the process inherently reduces the regulatory burden associated with controlling genotoxic impurities and heavy metal residues. This simplification of the impurity profile translates directly into a more reliable supply of raw materials, as the risk of batch rejection due to out-of-specification purity results is significantly minimized. Furthermore, the use of inexpensive and readily available catalysts like ferric sulfate replaces costly proprietary reagents, creating a leaner cost structure that enhances overall margin potential without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the elimination of expensive solvent recovery steps and the reduction of waste generation associated with failed crystallizations. By utilizing a hydrolysis-esterification loop, the method avoids the high energy consumption typical of distillation-based purification, leading to a drastically simplified utility profile. The ability to use common industrial solvents like benzyl alcohol and cyclohexane, combined with the high efficiency of the acid salt catalyst, ensures that the cost of goods sold is optimized through material efficiency and reduced processing time.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route ensures consistent lead times and reduces the volatility often seen in API manufacturing. The process is less sensitive to minor fluctuations in reaction conditions compared to traditional isoelectric point adjustments, meaning that production schedules are more predictable and less prone to delays caused by re-processing. This stability allows supply chain planners to maintain lower safety stock levels while still guaranteeing continuity of supply for downstream formulation partners who depend on timely deliveries of high-quality Cefathiamidine.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing method is considerably lower, aligning with modern green chemistry principles and stringent global regulatory requirements. The avoidance of harsh mineral acids reduces the load on wastewater treatment facilities, while the high selectivity of the reaction minimizes the formation of by-products that require disposal. This eco-friendly profile not only facilitates easier permitting for new production lines but also future-proofs the supply chain against tightening environmental regulations, making it a sustainable choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific Cefathiamidine purification technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, offering clarity on how this method outperforms legacy techniques. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What purity levels can be achieved with this new purification method?
A: The patented process consistently achieves a purity greater than 99.2%, significantly reducing toxic side effects associated with residual impurities like bromide ions found in conventional synthesis routes.
Q: Why is a strong acid salt catalyst preferred over traditional acids?
A: Strong acid salts like ferric sulfate act as effective catalysts for the esterification step while minimizing the risk of beta-lactam ring opening, which is a common degradation pathway when using harsh mineral acids.
Q: How does this process address the issue of residual bromide?
A: By utilizing a hydrolysis and re-esterification strategy rather than direct condensation with bromoacetyl bromide in the final steps, the method effectively eliminates the carryover of hazardous bromide anions into the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefathiamidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced purification technologies requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Cefathiamidine we deliver meets the highest international pharmacopoeia standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this superior purification method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your product competitiveness in the global market.
