Scalable Single-Solvent Synthesis of Cefathiamidine for Commercial API Manufacturing
Introduction to Advanced Cefathiamidine Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with economic efficiency, particularly for first-generation cephalosporin antibiotics like cefathiamidine. Patent CN102070654A introduces a transformative preparation method that addresses longstanding challenges in the synthesis of this critical antibiotic. By shifting away from complex multi-solvent systems towards a streamlined single-solvent approach, this technology offers a compelling value proposition for reliable cefathiamidine suppliers aiming to optimize their production lines. The core innovation lies in the precise manipulation of crystallization dynamics through pH and temperature control within the reaction matrix, effectively eliminating the need for traditional anti-solvent precipitation steps. This technical advancement not only simplifies the operational workflow but also significantly enhances the purity profile of the final active pharmaceutical ingredient, meeting stringent pharmacopoeia requirements without the need for energy-intensive recrystallization processes.
For R&D directors and process engineers, the implications of this patent extend beyond mere procedural changes; it represents a fundamental improvement in process chemistry that mitigates the risks associated with thermal instability and amorphous powder formation. The method utilizes a sophisticated mixed base catalyst system, combining organic and inorganic bases to fine-tune the reaction environment. This dual-catalyst approach facilitates the generation of trace inorganic salts that serve as nucleation agents, promoting the growth of stable, uniform crystals rather than the coarse, impurity-laden particles typical of older methods. As global demand for high-quality antibiotics remains steady, adopting such efficient synthesis routes becomes a strategic imperative for maintaining competitiveness in the generic pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefathiamidine, as detailed in prior art such as USP 3,646,025, has relied heavily on the addition of large volumes of crystallization solvents to induce precipitation after the initial condensation reaction. This conventional approach suffers from two major defects that severely impact both economic viability and product quality. Firstly, the requirement for multiple solvent systems complicates the recovery process, leading to increased operational expenditures and a larger environmental footprint due to the energy required for distillation and separation. Secondly, the rapid precipitation caused by adding anti-solvents often results in the co-precipitation of impurities, yielding coarse crystals that fail to meet purity standards without further processing. Consequently, manufacturers are forced to implement additional recrystallization steps, which further reduce overall yield and extend production lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
Furthermore, the zwitterionic nature of cefathiamidine makes it particularly susceptible to forming hydrogen bonds with solvents, leading to the formation of amorphous powders when separated out rapidly. These amorphous forms are thermally unstable and prone to degradation during storage, posing significant risks for inventory management and shelf-life stability. The reliance on external crystallization solvents exacerbates this issue by causing sudden changes in solubility that do not allow for the orderly arrangement of molecules into a stable crystal lattice. For procurement managers, these inefficiencies translate into higher raw material costs and unpredictable batch consistency, making it difficult to secure a stable supply of compliant API materials for downstream formulation.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in CN102070654A utilizes a single solvent system, preferably methylene dichloride, to carry out both the reaction and the subsequent crystallization. By eliminating the addition of a separate devitrification or anti-solvent, the process achieves a controlled crystallization environment where the product precipitates solely through the regulation of pH value and temperature. This method allows for the gradual formation of crystal seeds, ensuring that the resulting particles are homogeneous and free from trapped impurities. The reaction equation illustrates the direct condensation of 7-bromoacetyl ACA with N,N-diisopropyl thiourea, a transformation that is now rendered more efficient and cleaner through this optimized protocol.
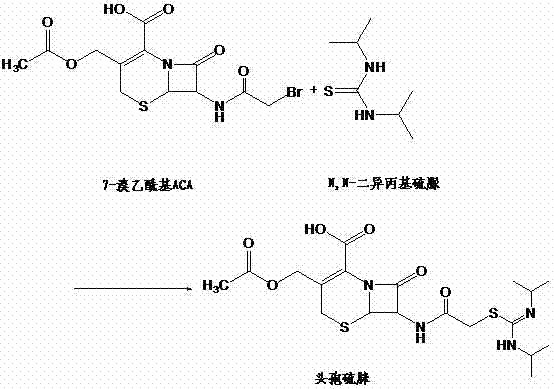
The implementation of this single-solvent strategy drastically simplifies the post-reaction workup, as there is no need to recover and recycle a secondary crystallization solvent. This reduction in solvent usage directly correlates to cost reduction in antibiotic manufacturing, as it lowers both the purchase price of raw materials and the utility costs associated with solvent recovery. Moreover, the absence of rapid anti-solvent precipitation prevents the mechanical entrapment of mother liquor impurities within the crystal lattice, resulting in a product with purity levels reaching 98.4% to 98.6% directly from the reactor. This level of purity is achieved without the need for recrystallization, representing a significant leap forward in process intensification for commercial scale-up of complex pharmaceutical compounds.
Mechanistic Insights into Mixed Base Catalyzed Condensation
The success of this synthesis route hinges on the intricate interplay between the mixed base catalyst and the physicochemical properties of the reaction mixture. The catalyst system comprises a specific ratio of organic bases, such as triethylamine, and inorganic bases, such as ammonia water. While the organic base serves the traditional role of scavenging the hydrobromic acid generated during the condensation, the inorganic base plays a subtler yet critical role in the crystallization phase. As the reaction proceeds, trace amounts of inorganic bromides are generated which, being insoluble in the organic solvent, act as heterogeneous nucleation sites. These microscopic nuclei provide a template for the cefathiamidine molecules to align and grow, facilitating the transition from a supersaturated solution to a stable crystalline solid without the shock of anti-solvent addition.
Equally critical is the precise control of the solution pH during the crystallization phase, which must be maintained strictly between 3 and 6. The solubility of cefathiamidine is highly sensitive to pH changes due to its zwitterionic character; at a pH above 6.0, the compound remains too soluble to crystallize effectively, while a pH below 3.0 causes excessively rapid precipitation. Rapid precipitation at low pH leads to agglomeration and the encapsulation of impurities, defeating the purpose of the purification. By carefully titrating the pH to an optimal range, preferably around 4, the process ensures a controlled supersaturation level that favors the growth of large, pure crystals over the nucleation of fine, impure particles. This mechanistic understanding allows for the reproducible production of high-purity cefathiamidine, addressing the thermal instability issues inherent to the molecule's structure.
How to Synthesize Cefathiamidine Efficiently
The synthesis of cefathiamidine via this patented method involves a sequence of carefully controlled unit operations designed to maximize yield and purity while minimizing waste. The process begins with the dissolution of the key starting materials, 7-bromoacetyl ACA and N,N-diisopropyl thiourea, in a single organic solvent, followed by the addition of the mixed base catalyst to initiate the condensation. Once the reaction is complete, the mixture undergoes a specialized crystallization protocol where pH and temperature are modulated to induce precipitation without external solvents. The detailed standardized synthetic steps for implementing this laboratory-to-pilot scale procedure are outlined in the guide below.
- Dissolve 7-bromoacetyl ACA and N,N-diisopropyl thiourea in a single solvent such as methylene dichloride, adding a mixed base catalyst comprising organic and inorganic bases.
- Conduct the condensation reaction at a controlled temperature between 15°C and 35°C while adding activated carbon for decolorization.
- Filter the solution, adjust the pH to between 3 and 6, and lower the temperature to 5°C to 30°C to induce crystallization without adding anti-solvents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this single-solvent synthesis method offers tangible benefits that extend well beyond the laboratory bench. The primary economic driver is the drastic simplification of the solvent recovery infrastructure. By utilizing only one main solvent throughout the reaction and crystallization phases, manufacturers can significantly reduce the capital expenditure required for distillation columns and solvent storage tanks. This consolidation of the solvent system not only lowers the initial investment but also reduces the ongoing operational costs associated with energy consumption and solvent loss, leading to substantial cost savings in the overall manufacturing budget. Furthermore, the elimination of the recrystallization step shortens the total batch cycle time, allowing for increased throughput and better responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The removal of the recrystallization step and the reduction in solvent variety directly lower the cost of goods sold (COGS). By avoiding the use of anti-solvents like acetone for precipitation, the process eliminates the need to recover and purify a second solvent stream, which is often energy-intensive. Additionally, the higher yield obtained directly from the reactor means that less raw material is required per kilogram of finished API, further driving down the variable costs associated with production. This efficiency makes the process highly attractive for generic drug manufacturers operating on thin margins who need to optimize every aspect of their production line.
- Enhanced Supply Chain Reliability: Simplifying the chemical process inherently reduces the number of potential failure points in the supply chain. With fewer solvents to source and manage, the risk of supply disruptions due to raw material shortages is minimized. The robustness of the crystallization control, which relies on pH and temperature rather than the precise addition rate of an anti-solvent, makes the process more forgiving and easier to scale. This reliability ensures a consistent supply of high-purity cefathiamidine, reducing the risk of stockouts and enabling more accurate forecasting for downstream formulation partners.
- Scalability and Environmental Compliance: The reduction in solvent usage aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. Less solvent consumption translates to lower volatile organic compound (VOC) emissions and reduced hazardous waste generation, simplifying the permitting process for new manufacturing facilities. The ability to produce pharmacopoeia-grade material without recrystallization also means less wastewater is generated from washing steps, lowering the burden on effluent treatment plants. This environmental compliance is a crucial factor for long-term sustainability and corporate social responsibility goals in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cefathiamidine synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on how this method compares to traditional approaches. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of suppliers utilizing this advanced process.
Q: What is the primary advantage of the single-solvent method for cefathiamidine production?
A: The primary advantage is the elimination of a separate crystallization solvent, which simplifies solvent recovery, reduces production costs, and prevents the co-precipitation of impurities often caused by anti-solvents.
Q: How does the mixed base catalyst improve the crystallization process?
A: The mixture of organic and inorganic bases generates trace inorganic bromides during the reaction, which act as nucleation sites to facilitate uniform crystal growth and prevent the formation of amorphous powders.
Q: Why is strict pH control critical during the crystallization of cefathiamidine?
A: Cefathiamidine is a zwitterion with solubility highly dependent on pH; maintaining a pH between 3 and 6 ensures optimal solubility balance, preventing rapid agglomeration that traps impurities or excessive solubility that hinders crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefathiamidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively evaluated the single-solvent methodology described in CN102070654A and possesses the technical capability to implement this process at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of cefathiamidine meets the highest international standards, providing our partners with the confidence they need to proceed with their own formulation development.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By leveraging our expertise in process optimization, we can offer a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how the elimination of recrystallization and solvent reduction translates to bottom-line savings. We encourage you to request specific COA data and route feasibility assessments to verify the superior quality and consistency of our cefathiamidine offerings. Let us collaborate to streamline your antibiotic supply chain with a partner dedicated to technical excellence and commercial reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
