Advanced Purification of Cefixime Intermediates Using Novel Tert-Octylamine Salt Technology
Advanced Purification of Cefixime Intermediates Using Novel Tert-Octylamine Salt Technology
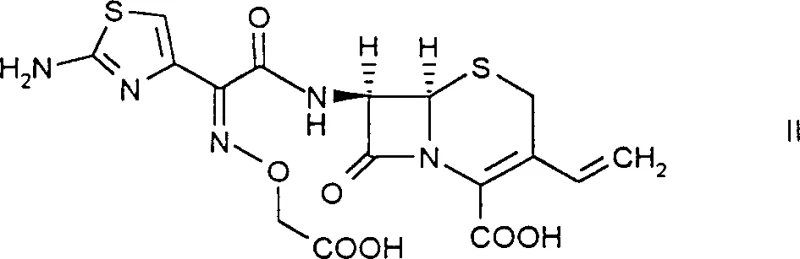
The pharmaceutical industry continuously seeks robust methodologies to enhance the purity and yield of critical antibiotic intermediates, particularly for third-generation cephalosporins like cefixime. Patent CN1134447C introduces a groundbreaking approach to overcoming the historical purification bottlenecks associated with 7-[2-(aminothiazol-4-yl)-2-((aryl or alkyl)oxycarbonylmethoxyimino)acetamido]-3-vinyl-3-ceph-3-ene-4-carboxylic acid derivatives. Traditionally, these intermediates exist in a free acid form that demonstrates notoriously poor solubility in standard organic solvents such as halogenated hydrocarbons, ketones, or esters, rendering conventional recrystallization techniques ineffective and often leading to significant yield losses during downstream processing. This patent discloses the formation of a novel, crystalline tert-octylamine salt which serves as a pivotal purification handle, allowing manufacturers to isolate the intermediate in high purity before final conversion to the active pharmaceutical ingredient. By leveraging this specific salt formation strategy, producers can achieve purity levels exceeding 98.9%, effectively addressing the stringent regulatory requirements for beta-lactam antibiotics while streamlining the overall production workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefixime and its precursors has been plagued by significant downstream processing challenges, primarily stemming from the physicochemical properties of the free acid intermediates. In standard synthetic routes, the coupling of the side chain to the 7-aminocephalosporanic acid nucleus often results in a crude reaction mixture containing various isomers, unreacted starting materials, and polymeric by-products. Because the target intermediate in its free acid form lacks favorable crystallization kinetics in common industrial solvents, manufacturers were previously forced to rely on complex chromatographic separations or multiple precipitation steps that offered limited resolution. These traditional methods not only incurred substantial operational costs due to high solvent consumption and extended processing times but also resulted in variable product quality, with purity often fluctuating below the desired thresholds for direct conversion to the final API. Furthermore, the instability of the beta-lactam ring under prolonged exposure to harsh purification conditions posed a constant risk of degradation, necessitating rapid and often inefficient workup procedures that compromised overall process yield and economic viability.
The Novel Approach
The innovative methodology described in the patent fundamentally shifts the purification paradigm by introducing a selective salt formation step using tert-octylamine. Instead of attempting to purify the difficult-to-handle free acid directly, the process converts the crude intermediate into a tert-octylamine salt, which exhibits superior crystallinity and solubility characteristics tailored for efficient isolation. This transformation allows for the effective exclusion of impurities that remain soluble in the mother liquor or fail to co-crystallize with the bulky tert-octylamine cation. The procedure can be executed as a one-pot reaction, where the intermediate is generated in situ and immediately treated with the amine base, thereby eliminating the need for isolating the unstable free acid. This seamless integration of synthesis and purification not only simplifies the equipment requirements but also drastically reduces the exposure of the sensitive beta-lactam core to potentially degrading conditions. Consequently, this approach facilitates the production of high-purity intermediates that can be directly saponified and converted into cefixime trihydrate with minimal additional cleanup, representing a significant leap forward in process efficiency and product consistency.
Mechanistic Insights into Tert-Octylamine Salt Formation and Crystallization
The core of this technological advancement lies in the specific interaction between the carboxylic acid moiety of the cephalosporin intermediate and the sterically hindered tert-octylamine base. When the reaction mixture containing the 7-acylated intermediate is treated with tert-octylamine in a suitable solvent system, typically involving alcohols like methanol mixed with non-solvents such as dichloromethane or esters, an acid-base neutralization occurs rapidly. The resulting ammonium carboxylate salt possesses a unique crystal lattice energy that favors ordered precipitation over the disordered aggregation seen in the free acid form. The bulky tert-octyl group provides significant steric bulk that prevents the close packing of impurities within the crystal structure, effectively acting as a molecular sieve during the crystallization phase. This phenomenon is critical for removing closely related structural analogs and geometric isomers that often co-elute in chromatographic systems. The patent highlights that this salt formation can be driven to completion even in the presence of residual reaction by-products, as the solubility product of the tert-octylamine salt is sufficiently low to ensure quantitative precipitation from the reaction matrix.
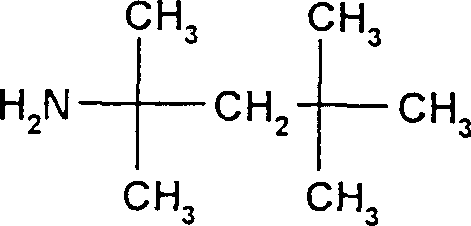
Furthermore, the mechanistic pathway ensures robust impurity control throughout the synthesis lifecycle. The initial acylation step, whether performed via activated esters or Vilsmeier-activated acids, generates the crude intermediate which is then subjected to the salting-out process. By adjusting parameters such as solvent polarity and temperature, operators can fine-tune the supersaturation level to optimize crystal growth and morphology. The patent data indicates that the resulting crystals are stable and can be filtered and dried without significant decomposition, preserving the integrity of the vinyl group and the aminothiazole side chain. Subsequent conversion to the final cefixime API involves a straightforward saponification of the ester protecting groups followed by pH adjustment, a process that is rendered more efficient because the starting material (the salt) is already of high purity. This mechanistic elegance minimizes the generation of new impurities during the final deprotection steps, ensuring that the final API meets the rigorous specifications required for global pharmaceutical markets without the need for extensive reprocessing.
How to Synthesize Cefixime Intermediate Efficiently
The synthesis of this high-value intermediate relies on a carefully orchestrated sequence of acylation and salt formation steps that maximize yield while minimizing waste. The process begins with the activation of the side-chain acid, followed by coupling with the cephalosporin nucleus, and concludes with the critical addition of tert-octylamine to induce crystallization. Detailed operational parameters regarding solvent ratios, temperature profiles, and stoichiometry are essential for reproducibility at scale. For a comprehensive guide on the specific reaction conditions and workup procedures validated by the patent data, please refer to the standardized synthesis protocol outlined below.
- React 7-amino-3-vinyl-ceph-3-em-4-carboxylic acid derivatives with activated side-chain esters in organic solvents.
- Introduce tert-octylamine to the reaction mixture to induce crystallization of the intermediate salt.
- Isolate the crystalline salt via filtration and convert to final cefixime through saponification and pH adjustment.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this tert-octylamine salt technology translates into tangible operational efficiencies and risk mitigation strategies. The primary advantage lies in the drastic simplification of the purification workflow, which eliminates the need for expensive and time-consuming chromatographic columns or multiple recrystallization cycles that typically bottleneck production capacity. By enabling a one-pot synthesis where the intermediate is purified via simple filtration, manufacturers can significantly reduce solvent consumption and waste disposal costs, aligning with modern green chemistry initiatives. This streamlined approach also enhances batch-to-batch consistency, reducing the variability that often leads to production delays and quality investigations. The ability to produce high-purity intermediates reliably ensures a steady flow of material for final API synthesis, thereby stabilizing the supply chain against fluctuations in raw material quality or processing errors.
- Cost Reduction in Manufacturing: The implementation of this crystallization-based purification method offers substantial cost savings by removing the dependency on complex separation technologies. Traditional methods often require large volumes of high-grade solvents and specialized equipment for chromatography, both of which represent significant capital and operational expenditures. By shifting to a precipitation-based workflow, facilities can utilize standard reactor vessels and filtration units, lowering the barrier to entry for production and reducing the overall cost of goods sold. Additionally, the higher yields associated with avoiding degradative purification steps mean that less raw material is required to produce the same amount of final product, further driving down the unit cost of the cefixime intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the robustness of the manufacturing process, and this technology provides a more forgiving and stable production route. The crystalline nature of the tert-octylamine salt allows for easier storage and handling compared to oily or amorphous free acid intermediates, which may degrade over time. This stability extends the shelf-life of the intermediate, providing greater flexibility in inventory management and reducing the risk of stockouts due to material expiration. Furthermore, the simplified process reduces the number of unit operations, thereby decreasing the probability of mechanical failures or operator errors that could disrupt production schedules and delay deliveries to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often exposes hidden inefficiencies, but this salt formation technique is inherently scalable due to its reliance on fundamental thermodynamic principles rather than kinetic separations. The use of common organic solvents and the absence of exotic catalysts or reagents facilitate easy technology transfer between different manufacturing sites. From an environmental perspective, the reduction in solvent usage and the elimination of silica gel or resin waste from chromatography contribute to a lower environmental footprint. This alignment with sustainability goals not only aids in regulatory compliance but also enhances the corporate social responsibility profile of the manufacturing entity, making it a more attractive partner for global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process changes and procurement officers assessing supplier capabilities.
Q: Why is the tert-octylamine salt preferred for cefixime intermediate purification?
A: The free acid form of the cefixime intermediate exhibits poor solubility in common organic solvents, making traditional recrystallization difficult. The tert-octylamine salt forms highly crystalline structures that can be easily isolated via filtration, significantly enhancing purity to over 98.9%.
Q: Can this purification method be integrated into a one-pot synthesis?
A: Yes, the patent describes a one-pot methodology where the intermediate is generated and immediately converted into the tert-octylamine salt without isolation. This reduces solvent usage and processing time compared to multi-step isolation protocols.
Q: What represents the critical quality attribute in this synthesis route?
A: The critical quality attribute is the crystalline nature of the salt, which ensures the removal of impurities and isomers. The process achieves HPLC purity levels exceeding 99% for the final cefixime product after conversion from the salt.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefixime Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the production of life-saving antibiotics like cefixime. Our technical team has extensively analyzed advanced purification technologies, including the tert-octylamine salt method, to ensure our manufacturing processes meet the highest global standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of cefixime intermediate adheres to the precise requirements necessary for successful API synthesis.
We invite pharmaceutical partners to collaborate with us to leverage these advanced manufacturing capabilities for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that details how our optimized processes can reduce your overall production costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition from development to commercial supply.
