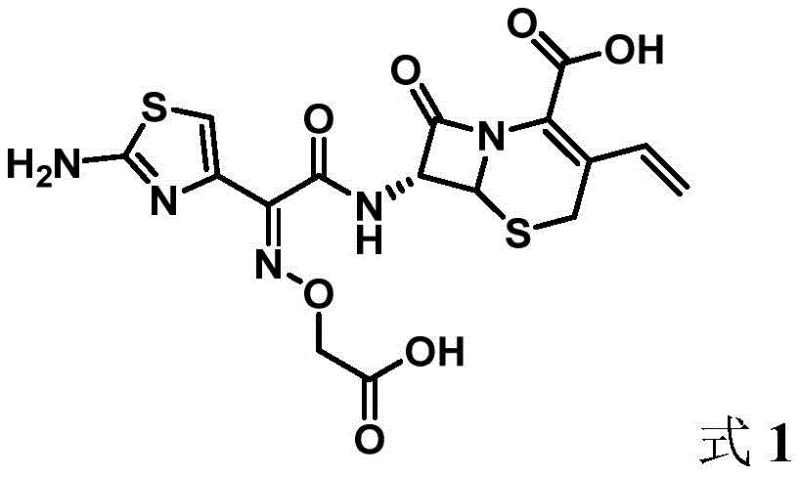
Revolutionizing Cefixime Production: Advanced One-Pot Synthesis for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics, and the recent disclosure in patent CN114805394A presents a significant advancement in the production of cefixime. This third-generation cephalosporin is vital for treating resistant bacterial infections, yet its traditional synthesis has long been plagued by complex multi-step procedures and environmental concerns. The new methodology described in this patent introduces a streamlined one-pot condensation strategy that utilizes a novel active intermediate to couple 7-AVNA with aminothiazole acetoxamic acid derivatives. This breakthrough not only simplifies the operational workflow but also drastically enhances the overall reaction efficiency and product quality. By shifting away from cumbersome activation steps, this technology offers a compelling value proposition for manufacturers aiming to optimize their antibiotic production lines while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of cefixime has relied heavily on the activation of the side chain carboxylic acid using reagents such as MICA activated thioesters or dinines activated thioesters. These conventional pathways necessitate the use of organophosphorus reagents to facilitate the coupling reaction, which introduces significant complexity and cost into the manufacturing process. The reliance on these specific activators often results in low reaction yields and generates substantial amounts of sulfide by-products that are notoriously difficult to separate from the final product. Furthermore, the traditional route typically requires at least two distinct steps for side chain introduction, involving intermediate isolation and purification operations that increase solvent consumption and processing time. These inefficiencies not only drive up production costs but also pose considerable challenges for waste management and environmental compliance in large-scale facilities.
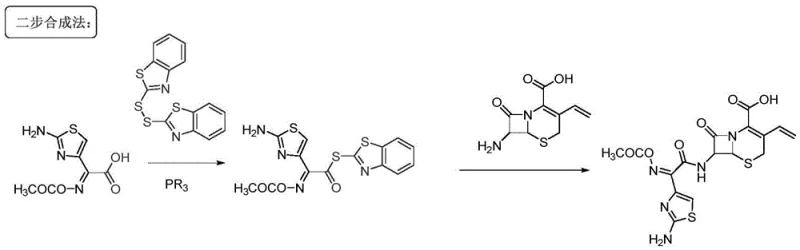
The Novel Approach
In stark contrast to the legacy methods, the innovative approach detailed in the patent utilizes N-ethynyl-N,4-dimethylbenzenesulfonamide as a key reagent to form a highly reactive intermediate in situ. This strategy allows for a direct nucleophilic substitution reaction with 7-AVNA without the need for isolating the intermediate species, effectively collapsing the synthesis into a single pot. The elimination of organophosphorus reagents is a critical improvement, as it removes the need for expensive and hazardous chemicals while simultaneously simplifying the downstream purification process. By operating under mild conditions, typically between 20°C and 35°C, this method reduces energy consumption and minimizes the risk of thermal degradation of the sensitive beta-lactam ring. The result is a process that is not only chemically superior in terms of yield and purity but also operationally more efficient for commercial scale-up of complex pharmaceutical intermediates.
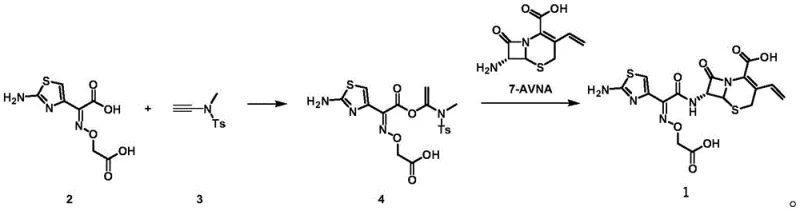
Mechanistic Insights into N-Ethynyl-N,4-Dimethylbenzenesulfonamide Mediated Coupling
The core of this technological advancement lies in the unique reactivity of the N-ethynyl-N,4-dimethylbenzenesulfonamide reagent, which facilitates a highly efficient addition reaction with the aminothiazole acetoxamic acid analog. This initial step generates a transient active intermediate that possesses superior electrophilic character compared to traditional thioester activators, thereby accelerating the subsequent nucleophilic attack by the 7-AVNA amine group. The mechanism avoids the formation of stable phosphorus-containing by-products, which are often the source of contamination and purification difficulties in older synthetic routes. Instead, the by-products generated are primarily sulfonamide derivatives that are significantly easier to separate via standard chromatographic or crystallization techniques. This mechanistic shift ensures that the final cefixime product achieves high-purity specifications with minimal impurity profiles, a critical factor for meeting regulatory requirements in global pharmaceutical markets.
Furthermore, the control of impurities is inherently built into the reaction design through the use of mild temperatures and specific solvent systems such as dichloromethane or ethyl acetate. The reaction conditions are optimized to prevent the epimerization of the chiral centers within the cefixime molecule, which is a common degradation pathway in harsher synthetic environments. By maintaining the reaction temperature within a narrow range of 0-50°C, preferably at 25°C, the process ensures the stereochemical integrity of the final API. The high conversion rate observed in the first step means that the reaction mixture can proceed directly to the second step without intermediate workup, reducing the exposure of the reactive species to potential degradative conditions. This level of process control is essential for R&D directors focused on impurity谱 analysis and ensuring the long-term stability of the drug product.
How to Synthesize Cefixime Efficiently
Implementing this synthesis route requires precise control over reagent stoichiometry and solvent selection to maximize the benefits of the one-pot design. The process begins with the dissolution of the aminothiazole acetoxamic acid analog in a suitable organic solvent, followed by the addition of the sulfonamide reagent to generate the active species. Once the formation of the intermediate is confirmed, typically via TLC or HPLC monitoring, the 7-AVNA is introduced directly to the same vessel to complete the coupling. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- React aminothiazole acetoxamic acid analog with N-ethynyl-N,4-dimethylbenzenesulfonamide in an organic solvent like dichloromethane at 25°C to form the active intermediate.
- Without isolating the intermediate, directly add 7-AVNA to the reaction mixture to proceed with nucleophilic substitution.
- Monitor reaction completion via HPLC, then concentrate and purify the mixture using silica gel column chromatography to obtain high-purity cefixime.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates into tangible operational improvements and risk mitigation strategies. The simplification of the chemical process directly addresses several pain points associated with the sourcing and manufacturing of high-purity cefixime. By reducing the number of unit operations and eliminating the need for specialized phosphorus reagents, manufacturers can achieve significant cost savings in raw material procurement and waste disposal. The robustness of the reaction conditions also enhances supply chain reliability, as the process is less susceptible to variations in raw material quality or environmental fluctuations. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive organophosphorus activators and the reduction of separation steps lead to a substantial decrease in overall production costs. By avoiding the purchase of specialized sulfur reagents and reducing solvent usage through a one-pot design, the economic efficiency of the manufacturing process is significantly enhanced. Additionally, the higher reaction yield means that less raw material is required to produce the same amount of final product, further driving down the cost per kilogram. These qualitative improvements in process economics allow for more competitive pricing strategies without compromising on product quality or margin.
- Enhanced Supply Chain Reliability: The use of common organic solvents such as dichloromethane and ethyl acetate ensures that raw material sourcing is not constrained by the availability of exotic or regulated chemicals. This accessibility reduces the risk of supply disruptions and allows for more flexible inventory management strategies. Furthermore, the mild reaction conditions reduce the energy load on production facilities, making the process more resilient to energy price volatility. The simplified workflow also shortens the overall production cycle time, enabling faster response to market demand fluctuations and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis significantly reduces the generation of hazardous waste, particularly sulfide by-products that are difficult to treat in traditional methods. This reduction in environmental impact simplifies the regulatory compliance process and lowers the costs associated with waste treatment and disposal. The process is inherently safer due to the absence of highly reactive phosphorus compounds, making it easier to scale from pilot plant to commercial production volumes. This scalability ensures that the technology can meet the growing global demand for cefixime while adhering to increasingly stringent environmental protection standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced cefixime synthesis technology. These answers are derived directly from the patent data and practical manufacturing considerations to provide clarity for decision-makers. Understanding these aspects is crucial for evaluating the feasibility of adopting this route for your specific production needs.
Q: How does this new synthesis method improve yield compared to traditional routes?
A: The novel one-pot method eliminates the need for cumbersome separation steps and expensive organophosphorus reagents used in conventional thioester activation, resulting in yields up to 94.0% and purity exceeding 99.4%.
Q: What are the environmental benefits of avoiding organophosphorus reagents?
A: By utilizing N-ethynyl-N,4-dimethylbenzenesulfonamide instead of phosphorus-based activators, the process significantly reduces hazardous waste generation and simplifies the removal of sulfide by-products, enhancing environmental compliance.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (0-50°C) and uses common organic solvents like dichloromethane or ethyl acetate, making it highly scalable and safer for commercial manufacturing compared to multi-step traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefixime Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing processes for essential antibiotics like cefixime. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial operations. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch conforms to the highest international standards. Our capability to implement complex synthetic routes allows us to offer reliable solutions for clients seeking to optimize their supply chains.
We invite you to collaborate with us to explore how this advanced synthesis technology can benefit your organization. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how we can support your goal of reducing lead time for high-purity pharmaceutical intermediates while enhancing overall operational efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
