Advanced Deprotection Technology for High-Purity Cefdinir Intermediates
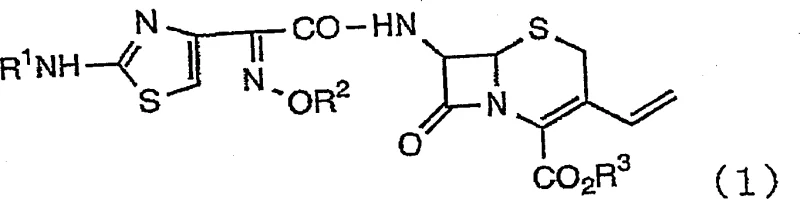
The pharmaceutical industry continuously seeks robust methodologies for the synthesis of complex beta-lactam antibiotics, particularly third-generation cephalosporins like cefdinir. Patent CN1134446C introduces a groundbreaking process for the preparation of 3-vinylcephem compounds, specifically addressing the critical deprotection step that has historically plagued manufacturers with low yields and safety concerns. This technology leverages a sophisticated dual-acid system comprising a perhalogenic acid and an organic protonic acid to achieve unprecedented efficiency. By shifting away from hazardous Lewis acids and volatile strong acids, this innovation offers a pathway to high-purity intermediates essential for modern oral antibiotic production. The strategic application of this chemistry allows for the precise removal of protecting groups without compromising the delicate beta-lactam ring or the vinyl side chain. For global supply chains, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for key active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the deprotection of protected 3-vinylcephem derivatives has been a bottleneck in cefdinir production, characterized by harsh conditions and suboptimal outcomes. Traditional methodologies, such as those disclosed in Japanese Patent Publication No. 1-49273, rely heavily on boron trifluoride etherate in anisole and acetic acid mixtures. These processes are fundamentally flawed for industrial scale-up due to reaction yields stagnating at merely 35%, coupled with the necessity of handling large quantities of dangerous boron trifluoride compounds. Furthermore, alternative approaches utilizing trifluoroacetic acid in the presence of anisole, as seen in JP-A-62-294687, suffer from volatility issues, complex handling procedures, and exorbitant costs. These conventional routes often necessitate the use of excessive amounts of industrially difficult reagents, leading to yields as low as 28%. Additionally, methods employing 99% formic acid as a solvent or aluminum chloride in anisole introduce severe risks of beta-lactam ring decomposition and are unsuitable due to the excessive acidity which destabilizes the cefdinir skeleton. The prevalence of strong protic or Lewis acids in these legacy methods frequently induces unwanted cis/trans isomerization of the oxime moiety, generating unacceptable levels of impurities that complicate downstream purification.
The Novel Approach
In stark contrast to these inefficient legacy protocols, the technology outlined in patent CN1134446C presents a refined deprotection strategy that harmonizes reactivity with stability. This novel approach utilizes a controlled mixture of a perhalogenic acid, such as perchloric acid, and an organic protonic acid with a pKa between 3 and 5, such as formic or acetic acid. By carefully tuning the acidity of the reaction medium, the process effectively cleaves protecting groups like trityl or benzyl derivatives while preserving the integrity of the acid-labile cefdinir core. The method operates under mild thermal conditions, typically ranging from 0°C to 50°C, which significantly reduces energy consumption and thermal stress on the molecule. Crucially, this system requires only a catalytic or near-stoichiometric amount of the strong perhalic acid (approximately 0.15 molar equivalents), drastically reducing the corrosive load on reactor equipment. The result is a dramatic improvement in process efficiency, transforming a previously low-yielding, hazardous step into a high-performance unit operation capable of delivering yields exceeding 95%.
Mechanistic Insights into Perhalic Acid-Catalyzed Deprotection
The success of this deprotection methodology lies in its unique mechanistic interplay between the organic protonic acid and the perhalogenic acid within the organic solvent matrix. The organic protonic acid, present in a molar excess of 3 to 5 equivalents relative to the substrate, serves a dual function: it acts as a proton source for the initial activation of the protecting group and, more importantly, forms hydrogen bonds with the amide and amino groups on the cefdinir skeleton. This hydrogen bonding network effectively shields the sensitive functional groups from the harsh effects of the strong acid, preventing the degradation of the beta-lactam ring that typically occurs in strongly acidic environments. Simultaneously, the perhalogenic acid, used in minimal quantities, provides the necessary protonating power to cleave the robust carbon-oxygen or carbon-nitrogen bonds of the protecting groups, such as trityl or diphenylmethyl moieties. This synergistic effect ensures that the deprotection proceeds rapidly and selectively without inducing the structural collapse of the antibiotic core. The reaction kinetics are further optimized by the choice of solvent, with halogenated hydrocarbons like dichloromethane and aromatic hydrocarbons like toluene providing ideal polarity for stabilizing the transition states involved in the cleavage mechanism.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the stereochemical integrity of the oxime side chain. In conventional strong acid hydrolysis, the high concentration of protons facilitates the equilibration of the oxime group, leading to a mixture of Z and E isomers, where the E-isomer is an undesirable impurity. The buffered acidity provided by the organic protonic acid in this novel system maintains a proton activity level that is sufficient for deprotection but insufficient to drive significant oxime isomerization. Consequently, the Z-configuration of the oxime, which is essential for the biological activity of cefdinir, is preserved with high fidelity throughout the reaction. Furthermore, the mild conditions prevent the polymerization or degradation of the reactive vinyl group at the 3-position of the cephem nucleus. Post-reaction workup is simplified as the minimal acid usage allows for straightforward neutralization and extraction, avoiding the formation of intractable emulsions or salt precipitates that often trap product in traditional workflows. This precise control over the reaction environment ensures a clean impurity profile, reducing the burden on subsequent crystallization and purification steps.
How to Synthesize 3-Vinylcephem Compounds Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the dual-acid system. The process begins with the dissolution of the protected precursor in a suitable organic solvent, followed by the sequential addition of the organic acid and the perhalic acid catalyst. Maintaining the reaction temperature within the optimal window of 0°C to 50°C is crucial to balance reaction rate with product stability. Detailed standard operating procedures for this high-yield transformation are outlined below, providing a clear roadmap for technical teams to replicate the >95% yields reported in the patent examples. Adhering to these parameters ensures consistent quality and safety during the manufacturing of this critical antibiotic intermediate.
- Dissolve the protected 3-vinylcephem derivative (Formula 1) in an organic solvent such as dichloromethane or toluene.
- Add an organic protonic acid (e.g., formic acid, acetic acid) in an amount of 3-5 molar equivalents relative to the substrate.
- Introduce a perhalogenic acid (e.g., 60% perchloric acid) at a catalytic amount of approximately 0.15 molar equivalents.
- Maintain the reaction temperature between 0°C and 50°C for 1 to 6 hours, then perform extraction and crystallization to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this deprotection technology translates directly into enhanced operational resilience and cost efficiency. The elimination of hazardous reagents like boron trifluoride and the reduction in the volume of corrosive acids significantly lower the costs associated with safety compliance, waste disposal, and specialized equipment maintenance. By replacing low-yielding processes that waste over 60% of valuable starting materials with a high-efficiency route, manufacturers can drastically reduce the cost of goods sold (COGS) for cefdinir intermediates. The simplified workup procedure, which avoids complex neutralization and filtration steps, shortens the overall batch cycle time, thereby increasing plant throughput and asset utilization. This technological upgrade addresses the chronic pain points of supply volatility and high production costs that have long affected the availability of oral cephalosporins in the global market.
- Cost Reduction in Manufacturing: The shift from expensive and dangerous reagents like boron trifluoride etherate to commodity chemicals such as formic acid and perchloric acid results in substantial raw material cost savings. The dramatic increase in reaction yield from historical averages of 30% to over 95% means that significantly less starting material is required to produce the same amount of final product, effectively tripling the material efficiency of the process. Furthermore, the reduction in acid usage minimizes the consumption of neutralizing bases and solvents during the workup phase, leading to lower utility and consumable expenses. The avoidance of specialized corrosion-resistant reactors required for handling anhydrous hydrogen fluoride or high concentrations of trifluoroacetic acid also reduces capital expenditure requirements for new production lines.
- Enhanced Supply Chain Reliability: The reliance on widely available, stable organic acids and standard perhalic acids mitigates the risk of supply disruptions often associated with specialized fluorine reagents. The robustness of the reaction across a variety of solvents, including toluene and dichloromethane, provides procurement teams with the flexibility to source materials based on regional availability and price fluctuations without compromising process performance. The high selectivity of the reaction reduces the formation of hard-to-remove impurities, ensuring a consistent supply of high-quality intermediate that meets stringent regulatory specifications without the need for reprocessing. This reliability is critical for maintaining continuous production schedules for finished dosage forms and meeting the demands of global health markets.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified isolation procedure make this process highly amenable to commercial scale-up from pilot plants to multi-ton production facilities. The reduction in hazardous waste generation, particularly the elimination of boron-containing waste streams, aligns with increasingly strict environmental regulations and sustainability goals. The ability to operate at near-ambient temperatures reduces the energy footprint of the manufacturing process, contributing to a lower carbon intensity for the final pharmaceutical product. These factors collectively enhance the long-term viability of the supply chain, ensuring that production can be scaled up rapidly to meet surges in demand for essential antibiotics without encountering environmental or safety bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced deprotection technology. These insights are derived directly from the experimental data and process descriptions found in patent CN1134446C, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: What is the primary advantage of this deprotection method over traditional boron trifluoride methods?
A: Traditional methods using boron trifluoride etherate often result in low yields (around 35%) and require hazardous reagents. The novel method described in patent CN1134446C achieves yields exceeding 95% while using safer, more manageable acid systems.
Q: Which solvents are compatible with this high-yield deprotection process?
A: The process demonstrates excellent compatibility with a wide range of organic solvents including dichloromethane, chloroform, benzene, toluene, and various esters like ethyl acetate, allowing for flexible process optimization.
Q: How does this method impact the purity of the final cefdinir intermediate?
A: By utilizing a specific combination of weak organic protonic acids and minimal strong perhalic acid, the method minimizes cis/trans isomerization of the oxime group, resulting in significantly higher purity and reduced impurity profiles compared to strong acid hydrolysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the deprotection technology described in patent CN1134446C for the production of high-purity cefdinir intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of 3-vinylcephem compound meets the highest international standards. We are committed to leveraging this efficient synthesis route to provide our partners with a reliable, cost-effective, and sustainable source of critical antibiotic building blocks.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains for cephalosporin production. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our implementation of this patented technology can drive value and security into your antibiotic portfolio.
