Advanced Cefdinir Manufacturing: Leveraging Stable Active Ester Technology for Industrial Scale-Up
Advanced Cefdinir Manufacturing: Leveraging Stable Active Ester Technology for Industrial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for third-generation cephalosporins that balance high purity with operational safety. Patent CN101798313B introduces a transformative approach to Cefdinir production, specifically addressing the instability issues plaguing previous active ester methodologies. This innovation centers on the utilization of a novel active ester intermediate, 1-[(Z)-2-(2-amino-4-thiazolyl)-2-(acetoxyimino) acetoxy] benzotriazole, abbreviated as AB-AE. By shifting away from corrosive acid chloride pathways and unstable thioesters, this method achieves a remarkable overall yield exceeding 90% while maintaining exceptional product integrity. The structural complexity of Cefdinir, characterized by its vinyl group at the 3-position and the specific oxime configuration at the 7-position, demands precise chemical control to ensure bioactivity. 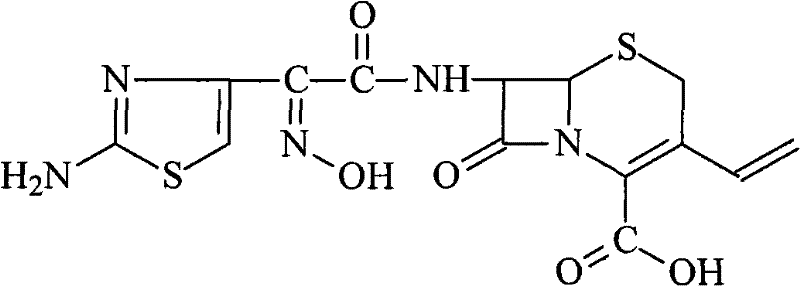
For R&D directors and process chemists, the significance of this patent lies in its ability to streamline the acylation step without compromising stereochemistry. Traditional methods often struggle with racemization or require cumbersome protection-deprotection sequences that erode overall efficiency. The AB-AE route utilizes 1-hydroxybenzotriazole (HOBT) to activate the side chain acid, creating a leaving group that is both reactive enough for efficient coupling with 7-AVCA and stable enough for practical storage. This dual characteristic solves a critical bottleneck in cephalosporin manufacturing, where intermediate degradation often leads to batch failures and increased waste. The process is designed to be scalable, moving seamlessly from laboratory optimization to commercial production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis of Cefdinir has largely relied on two problematic strategies: the acid chloride method and earlier active ester variations. The acid chloride route, while chemically direct, necessitates the handling of highly corrosive reagents and generates hazardous hydrogen chloride gas, imposing severe constraints on equipment material selection and operator safety. Furthermore, this method typically requires low-temperature reactions around -40°C and the use of silyl protecting groups like BSA, which adds cost and complexity to the downstream processing. Yields in these conventional chloride processes often stagnate between 60% and 70%, with purity profiles that demand extensive recrystallization. 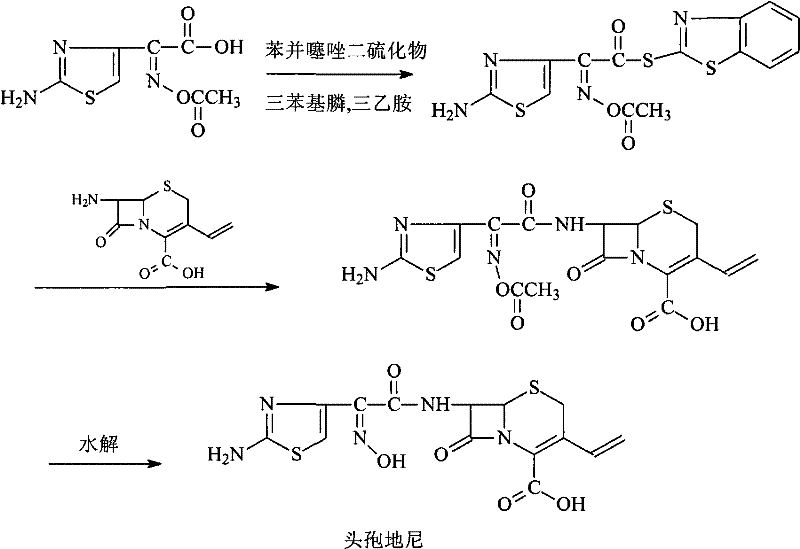
Alternatively, previous active ester methods, such as those utilizing benzothiazole thioesters (AT-AE), attempted to mitigate the harshness of acid chlorides but introduced significant supply chain vulnerabilities due to poor stability. Data indicates that these earlier esters degrade substantially over time, losing over 5% of their content and suffering noticeable purity drops within six months of storage. This instability forces manufacturers to produce intermediates just-in-time, preventing the strategic stockpiling of key materials and increasing the risk of production stoppages. The presence of trityl protecting groups in some variations further complicates the purification landscape, requiring acidic removal steps that can jeopardize the sensitive beta-lactam ring integrity.
The Novel Approach
The methodology disclosed in CN101798313B fundamentally reengineers the acylation landscape by introducing the AB-AE active ester. This benzotriazole-based ester is synthesized through the dehydration condensation of (Z)-2-(2-aminothiazole-4-yl)-2-acetoxyiminoacetic acid (ATAA) with HOBT. The resulting intermediate exhibits superior thermodynamic stability, retaining over 98% of its content even after six months of storage under ambient conditions. This robustness allows for the decoupling of intermediate synthesis from the final coupling reaction, granting procurement teams the flexibility to manufacture AB-AE in large batches during optimal conditions. The subsequent condensation with 7-amino-vinyl-3-cephalosporin-4-carboxylic acid (7-AVCA) proceeds smoothly in mild solvent systems like acetone or methylene chloride, eliminating the need for cryogenic temperatures.
Mechanistic Insights into Benzotriazole-Mediated Active Ester Formation
The core chemical innovation involves the activation of the carboxylic acid moiety of the side chain using HOBT and a carbodiimide dehydrating agent such as DCC or DIC. Mechanistically, the carbodiimide first reacts with the carboxylic acid to form an O-acylisourea intermediate, which is highly reactive but prone to rearrangement. The immediate intervention of HOBT captures this intermediate to form the AB-AE ester, a species where the benzotriazole ring acts as an excellent leaving group. This transformation is crucial because the electron-deficient nature of the benzotriazole nitrogen facilitates nucleophilic attack by the amine group of 7-AVCA, driving the amide bond formation forward with high kinetics. 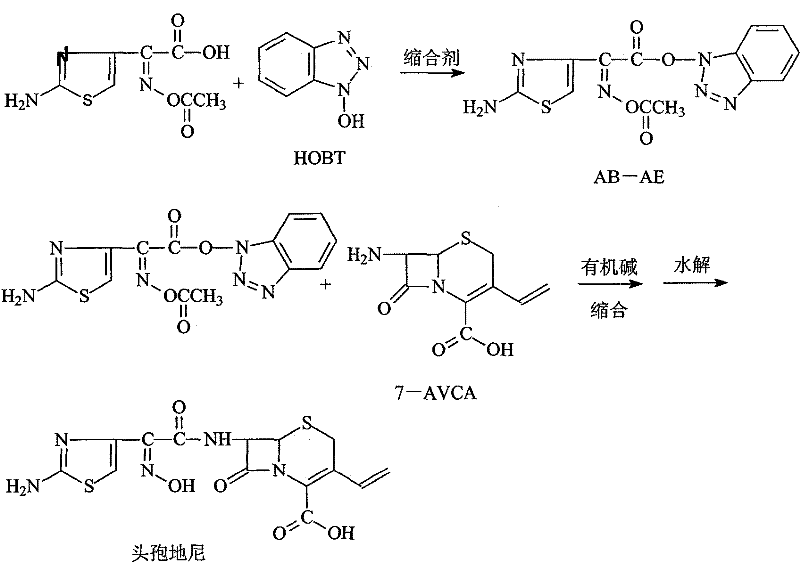
Furthermore, the use of the acetoxyimino protecting group on the oxime nitrogen serves a dual purpose: it prevents unwanted side reactions at the oxime functionality during the coupling phase and is easily removed in the final hydrolysis step. The hydrolysis is conducted under controlled alkaline conditions (pH 8.5) using mineral bases like sodium carbonate, which selectively cleaves the acetyl ester without opening the beta-lactam ring. This selectivity is paramount for maintaining the antibiotic potency of the final API. The entire sequence minimizes the formation of difficult-to-remove impurities, such as N-acylureas or racemized byproducts, resulting in a crude product profile that is exceptionally clean and ready for final crystallization.
How to Synthesize Cefdinir Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage pathway suitable for industrial implementation. The process begins with the activation of the side chain acid, followed by the coupling with the cephalosporin nucleus, and concludes with deprotection. Each step is optimized for yield and safety, utilizing common organic solvents and reagents that are readily available in the global chemical market. The detailed standard operating procedures for temperature control, stoichiometry, and workup are critical for replicating the high purity reported in the examples.
- Condense ATAA with HOBT using a dehydrating agent like DCC to form the stable AB-AE intermediate.
- React the purified AB-AE with 7-AVCA in the presence of an organic base to form the protected cephalosporin intermediate.
- Perform alkaline hydrolysis to remove the acetyl protecting group and isolate high-purity Cefdinir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the AB-AE route translates directly into enhanced operational resilience and cost efficiency. The primary economic driver is the dramatic improvement in intermediate stability. Unlike previous active esters that degraded rapidly, forcing costly just-in-time logistics, the AB-AE intermediate can be stored for extended periods without significant loss of potency. This capability allows manufacturers to build strategic inventory buffers, mitigating the risk of supply disruptions caused by raw material shortages or equipment downtime. The ability to stockpile a key intermediate effectively smooths out production scheduling and reduces the premium costs associated with rush orders or expedited shipping of unstable precursors.
- Cost Reduction in Manufacturing: The elimination of corrosive acid chlorides and the associated need for specialized glass-lined or Hastelloy reactors significantly lowers capital expenditure requirements. Furthermore, the high yield exceeding 90% means that less raw material is wasted per kilogram of final API produced. By avoiding the use of expensive silyl protecting agents and the complex workups required to remove them, the process reduces both reagent costs and solvent consumption. The simplified purification train also shortens cycle times, allowing for more batches to be produced within the same timeframe, thereby maximizing asset utilization and lowering the fixed cost per unit.
- Enhanced Supply Chain Reliability: The robustness of the AB-AE intermediate ensures a consistent supply of high-quality material, which is critical for maintaining regulatory compliance and batch-to-batch consistency. Since the intermediate does not require cold chain logistics for storage, transportation costs are significantly reduced, and the risk of spoilage during transit is minimized. This stability extends the shelf life of the material, reducing the frequency of disposal for expired stocks and aligning perfectly with lean manufacturing principles. Suppliers can offer more reliable lead times as the production process is less susceptible to the variability inherent in unstable chemical intermediates.
- Scalability and Environmental Compliance: The process operates under mild conditions without the generation of hazardous gaseous byproducts like HCl, simplifying waste gas treatment and scrubbing requirements. The use of standard organic solvents like acetone and ethyl acetate facilitates easier solvent recovery and recycling, contributing to a lower environmental footprint. The high purity of the crude product reduces the load on wastewater treatment systems by minimizing the volume of mother liquors and washings required for purification. This green chemistry profile not only meets stringent environmental regulations but also enhances the corporate sustainability metrics valued by modern pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Cefdinir synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of the AB-AE technology. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or process optimization projects.
Q: Why is the AB-AE active ester superior to traditional acid chloride methods for Cefdinir?
A: The AB-AE active ester method avoids the use of corrosive acid chlorides and harsh low-temperature conditions required in chloride methods. It offers significantly higher yields (>90%) and eliminates the need for complex silyl protection groups, simplifying the purification process and reducing equipment corrosion risks.
Q: How does the stability of the AB-AE intermediate compare to other active esters like AT-AE?
A: The AB-AE intermediate demonstrates exceptional storage stability compared to prior art esters like AT-AE. While AT-AE suffers from significant content reduction (over 5%) and purity loss after six months, AB-AE maintains its integrity with minimal degradation, ensuring consistent quality for long-term inventory management.
Q: What are the critical reaction conditions for the condensation step in this new process?
A: The condensation of AB-AE with 7-AVCA is optimally performed in solvents like acetone or methylene chloride with water, using organic bases such as triethylamine. The reaction proceeds efficiently at mild temperatures (0-40°C), avoiding the extreme cold required by older methods and facilitating easier thermal control during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the AB-AE method requires a partner with deep technical expertise and proven scale-up capabilities. Our facility is equipped to handle complex cephalosporin chemistry with precision, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of Cefdinir meets the highest international pharmacopoeia standards. Our commitment to quality assurance ensures that the theoretical advantages of this patent are fully realized in the commercial product delivered to your doorstep.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential efficiencies specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply security.
