Advanced Industrial Synthesis of Repaglinide Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antidiabetic agents, and patent CN1571769A presents a transformative approach to the synthesis of repaglinide, a potent meglitinide class drug. This specific intellectual property addresses the longstanding economic and technical bottlenecks associated with traditional amide coupling strategies used in the production of this vital API. By shifting away from expensive and hazardous reagents towards a mixed anhydride methodology, the disclosed process offers a pathway that is not only chemically elegant but also industrially pragmatic for large-scale operations. For global procurement teams and R&D directors, understanding this technological pivot is essential for securing a reliable repaglinide intermediate supplier capable of meeting stringent quality and volume demands. The core innovation lies in the strategic activation of the carboxylic acid component, which circumvents the purification nightmares inherent in older methods, thereby ensuring a more consistent supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of repaglinide has relied heavily on coupling agents such as N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-carbonyldiimidazole (CDI), both of which present severe drawbacks for industrial application. The use of DCC, while effective in small-scale laboratory settings, generates dicyclohexylurea as a stoichiometric byproduct, which is notoriously difficult to remove completely from the reaction mixture without resorting to multiple, yield-reducing recrystallization steps. Furthermore, alternative methods employing triphenylphosphine and carbon tetrachloride suffer from abysmal yields, often hovering around 50-55%, and necessitate chromatographic purification, a technique that is economically prohibitive and operationally cumbersome at the multi-ton scale required for commercial API manufacturing. These legacy processes also introduce significant safety and environmental liabilities due to the toxicity of reagents like carbon tetrachloride and the allergenic potential of carbodiimides, creating substantial barriers to cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN1571769A utilizes a mixed anhydride strategy mediated by trimethylacetyl chloride (pivaloyl chloride) and a tertiary amine base, representing a significant leap forward in process efficiency. This approach activates the protected benzoic acid derivative in situ, creating a highly reactive species that couples rapidly with the chiral amine intermediate under mild thermal conditions. By eliminating the formation of insoluble urea byproducts and avoiding the need for chromatographic separation, this novel route streamlines the downstream processing significantly, allowing for simple filtration and crystallization techniques to achieve exceptional purity levels. The transition to this chemistry not only enhances the overall yield profile but also drastically simplifies the waste stream management, making it an ideal candidate for the commercial scale-up of complex antidiabetic intermediates where operational simplicity translates directly to margin improvement.
Mechanistic Insights into Mixed Anhydride-Mediated Amidation
The core chemical transformation in this improved synthesis involves the generation of a mixed anhydride intermediate, which serves as the electrophilic partner for the nucleophilic attack by the chiral amine. Initially, the protected carboxylic acid reacts with trimethylacetyl chloride in the presence of a base such as triethylamine to form the mixed anhydride species, a reaction that proceeds efficiently at low temperatures ranging from -25 to 40°C. This activation step is crucial as it avoids the racemization risks often associated with harsher activation methods, thereby preserving the stereochemical integrity of the S-enantiomer which is critical for the biological activity of the final drug product. The subsequent addition of the amine component leads to the formation of the amide bond with high regioselectivity, driven by the steric and electronic properties of the mixed anhydride which favor attack at the less hindered carbonyl center.
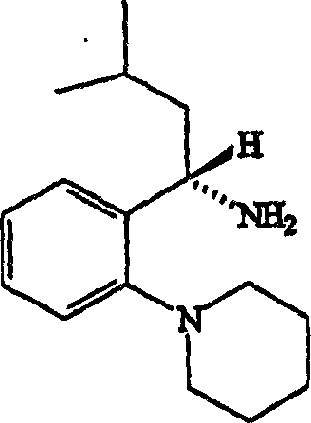
Following the coupling event, the process incorporates a robust deprotection strategy to reveal the free carboxylic acid functionality of repaglinide. The patent describes the use of standard hydrolysis conditions, either acidic or basic, or potentially hydrogenolysis depending on the specific protecting group employed, such as ethyl or benzyl esters. This flexibility in deprotection allows manufacturers to tailor the final steps to their existing infrastructure, ensuring that the removal of the protecting group does not compromise the purity of the sensitive amide linkage formed in the previous step. The ability to control impurities through crystallization rather than chromatography is a key mechanistic advantage, as it ensures that trace metal contaminants or organic impurities are effectively excluded from the final crystal lattice, resulting in a product that meets the rigorous standards required for high-purity pharmaceutical intermediates.
How to Synthesize Repaglinide Efficiently
The execution of this synthesis protocol requires precise control over reaction parameters to maximize the benefits of the mixed anhydride pathway. Operators must maintain strict temperature control during the activation phase to prevent side reactions, followed by a controlled warm-up to facilitate the coupling completion. The workup procedure is designed to be telescoped where possible, minimizing solvent swaps and handling times to enhance throughput. While the general principles are straightforward, the detailed standardized synthetic steps see the guide below for specific molar ratios and isolation techniques that guarantee reproducibility.
- Activation of the protected benzoic acid derivative using trimethylacetyl chloride and a tertiary amine base in a non-polar solvent like toluene at low temperatures.
- Coupling reaction with the chiral (S)-amine intermediate under controlled thermal conditions to form the protected amide bond with high stereoselectivity.
- Final deprotection step utilizing hydrolysis or hydrogenolysis to remove the ester protecting group, yielding the final active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic advantages that extend far beyond simple chemical yield improvements. By replacing exotic and hazardous reagents with commodity chemicals like pivaloyl chloride and triethylamine, the supply chain becomes significantly more resilient to market fluctuations and raw material shortages. The elimination of chromatographic purification steps reduces the consumption of vast quantities of solvents and silica gel, leading to a drastic simplification of the manufacturing workflow and a corresponding decrease in production cycle times. This operational efficiency allows suppliers to respond more agilely to demand spikes, effectively reducing lead time for high-purity pharmaceutical intermediates and ensuring continuity of supply for downstream formulation partners.
- Cost Reduction in Manufacturing: The economic impact of switching to this mixed anhydride method is profound, primarily driven by the removal of expensive coupling agents like CDI and the avoidance of yield losses associated with difficult purifications. Without the need to dispose of toxic dicyclohexylurea byproducts or manage hazardous carbon tetrachloride waste streams, the overall cost of goods sold (COGS) is substantially lowered through reduced waste treatment fees and raw material expenses. Furthermore, the higher yields achieved in both the coupling and deprotection steps mean that less starting material is required to produce the same amount of final API, compounding the savings across the entire production batch.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is often compromised by complex processes that are prone to failure or delay, but this streamlined approach mitigates those risks by utilizing robust and well-understood unit operations. The reliance on common solvents like toluene and standard bases ensures that raw material sourcing is not a bottleneck, as these commodities are widely available from multiple global vendors. This diversification of the supply base protects against single-source failures and allows for more flexible inventory management, ensuring that production schedules can be maintained even during periods of regional logistical disruption.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process is inherently greener and safer, aligning with the increasing global pressure for sustainable manufacturing practices. The absence of heavy metals and toxic halogenated solvents simplifies the environmental permitting process and reduces the liability associated with long-term storage and disposal of hazardous waste. Scalability is further enhanced because the exothermic nature of the mixed anhydride formation can be easily managed in large reactors using standard cooling jackets, removing the heat transfer limitations that often plague scale-up efforts for more exotic coupling chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and industry best practices. These insights are intended to clarify the feasibility of adopting this technology for commercial production and to highlight the specific quality attributes that define this manufacturing approach. Understanding these details is critical for technical teams evaluating potential technology transfers or capacity expansions.
Q: Why is the mixed anhydride method superior to DCC coupling for repaglinide production?
A: The mixed anhydride method avoids the formation of dicyclohexylurea (DCU), a difficult-to-remove byproduct associated with DCC coupling that often necessitates repeated crystallizations and reduces overall throughput.
Q: What are the purity specifications achievable with this novel process?
A: The patent data indicates that this process can achieve HPLC purities exceeding 99% for the intermediate and 99.5% for the final repaglinide product without requiring complex chromatographic purification.
Q: Is this process suitable for multi-ton commercial scale-up?
A: Yes, the process utilizes common industrial solvents like toluene and avoids hazardous reagents like carbon tetrachloride or expensive coupling agents, making it highly scalable and economically viable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Repaglinide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthesis route requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1571769A are fully realized in a GMP-compliant environment. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of repaglinide intermediate meets the exacting standards required by global regulatory authorities, providing our partners with absolute confidence in product quality.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing process can optimize your supply chain dynamics. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations.
