Optimizing Repaglinide Intermediate Production: A Technical Breakthrough in Amide Coupling
Optimizing Repaglinide Intermediate Production: A Technical Breakthrough in Amide Coupling
The global demand for effective type II diabetes treatments continues to drive innovation in the synthesis of key pharmaceutical intermediates. Among these, Repaglinide stands out as a critical meglitinide class drug, necessitating robust and scalable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN103012319A, which introduces a superior synthetic route for Repaglinide intermediates. This technology addresses long-standing challenges in heterocyclic chemistry, specifically focusing on the efficient construction of the amide bond between complex aromatic acids and chiral amines. By shifting away from traditional, problematic coupling reagents, this method offers a pathway to higher yields and exceptional stereochemical integrity.
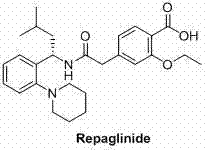
The significance of this patent lies in its ability to streamline the production of the Repaglinide precursor, specifically the ethyl ester intermediate. In the competitive landscape of reliable pharmaceutical intermediate supplier networks, the ability to deliver high-purity materials with consistent batch-to-batch reproducibility is paramount. The disclosed methodology not only enhances the chemical efficiency of the coupling step but also aligns with modern green chemistry principles by reducing the reliance on toxic solvents and difficult-to-remove byproducts. For R&D directors and process chemists, this represents a viable strategy to optimize the overall synthetic tree for this vital antidiabetic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Repaglinide and its precursors has relied on several established coupling strategies, each fraught with significant industrial drawbacks. Early methods utilized reagents such as N,N'-carbonyldiimidazole (CDI), which, while effective in small-scale laboratory settings, presents severe challenges for commercial scale-up due to its high cost and extreme sensitivity to moisture. Furthermore, alternative protocols employing triphenylphosphine in conjunction with carbon tetrachloride introduce substantial environmental and safety liabilities, as carbon tetrachloride is a known toxin with strict regulatory controls. Perhaps most critically, the widespread use of dicyclohexylcarbodiimide (DCC) generates dicyclohexylurea (DCU) as a stoichiometric byproduct.
The removal of DCU is a notorious bottleneck in process chemistry; its poor solubility in most organic solvents necessitates rigorous filtration and repeated recrystallization steps to achieve the required purity levels. These additional unit operations not only extend the production cycle time but also lead to significant material losses, thereby depressing the overall yield. Moreover, DCC-mediated reactions are prone to inducing racemization at the chiral center, compromising the optical purity of the final active pharmaceutical ingredient (API). Such impurities are unacceptable in modern pharmacopeia standards, forcing manufacturers to invest heavily in downstream purification technologies like preparative chromatography, which further escalates the cost of goods sold (COGS).
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN103012319A leverages advanced phosphonium and azide-based condensing agents, specifically 3-(diethoxyphosphoryloxy)-1,2,3-benzotriazin-4-one (DEPBT) or diphenylphosphoryl azide (DPPA). This strategic substitution fundamentally alters the reaction profile, eliminating the formation of insoluble urea byproducts and mitigating the risk of racemization. The new approach facilitates a cleaner reaction matrix where the desired amide product can be isolated through simple aqueous workups, such as washing with sodium bicarbonate and brine, rather than complex chromatographic separations.
This methodological shift enables a dramatic improvement in process efficiency, with reported total yields for the two-step sequence exceeding 90%. More importantly, the optical purity of the resulting intermediate is consistently maintained above 97%, often reaching 100% enantiomeric excess in optimized embodiments. By avoiding the use of hazardous reagents like carbon tetrachloride and eliminating the need for extensive purification cycles, this novel approach not only enhances the economic viability of the synthesis but also significantly reduces the environmental footprint. For procurement managers, this translates to a more stable supply chain with reduced dependency on expensive, moisture-sensitive reagents and lower waste disposal costs.
Mechanistic Insights into DEPBT-Mediated Amide Coupling
The core of this technological advancement lies in the unique activation mechanism provided by DEPBT and DPPA. Unlike carbodiimides which form O-acylisourea intermediates susceptible to rearrangement and racemization, phosphonium salts like DEPBT generate highly reactive acyloxyphosphonium species. This activated complex reacts rapidly with the nucleophilic amine component (Compound c) to form the peptide bond. The presence of the benzotriazine moiety in DEPBT acts as an excellent leaving group, facilitating the attack of the amine while suppressing side reactions. This mechanistic pathway is particularly advantageous for substrates containing sensitive chiral centers, such as the (S)-3-methyl-2-[2-(piperidino)phenyl]butylamine moiety found in Repaglinide.
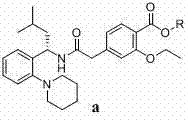
Furthermore, the reaction conditions specified in the patent allow for precise control over the stereochemical outcome. By conducting the reaction in non-protic solvents like toluene or ethyl acetate at moderate temperatures (e.g., 20°C to 60°C), the thermal energy is sufficient to drive the coupling forward without providing enough activation energy for epimerization. The choice of base, ranging from tertiary amines like triethylamine to inorganic bases like potassium carbonate, plays a crucial role in scavenging the acid byproducts generated during the activation step. This careful balancing of reactivity and selectivity ensures that the final product retains its biological activity, a critical parameter for high-purity Repaglinide intermediate manufacturing.
From an impurity control perspective, the absence of DCU and the minimal formation of other coupling-related byproducts simplify the impurity profile significantly. This cleanliness allows for direct telescoping of the reaction; the intermediate ester (Compound a) does not necessarily need to be isolated before hydrolysis to the final acid. This "one-pot" capability is a hallmark of mature process chemistry, reducing the number of isolation steps and minimizing the exposure of the chiral intermediate to potentially degrading conditions. For quality assurance teams, this means a more predictable impurity spectrum and easier validation of the cleaning procedures between batches.
How to Synthesize Repaglinide Ethyl Ester Efficiently
Implementing this improved synthesis route requires attention to specific stoichiometric ratios and solvent choices to maximize the benefits of the DEPBT or DPPA system. The patent outlines a robust protocol where the benzoic acid derivative (Compound b) and the condensing agent are dissolved in a solvent such as toluene, followed by the addition of a base. The chiral amine is then introduced dropwise to manage the exotherm and ensure complete conversion. This section provides a high-level overview of the operational parameters derived from the patent examples, serving as a guide for process engineers looking to adapt this chemistry for pilot or commercial plant operations.
- Dissolve the benzoic acid derivative (Compound b) and the condensing agent (DEPBT or DPPA) in a suitable organic solvent such as toluene or ethyl acetate under inert atmosphere.
- Add an organic base like triethylamine or pyridine at controlled temperatures (e.g., 20°C) to activate the carboxylic acid species.
- Introduce the chiral amine component (Compound c) dropwise, maintaining reaction conditions to prevent racemization, followed by aqueous workup to isolate the high-purity ester.
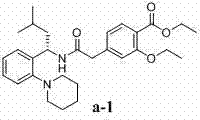
The successful execution of this synthesis hinges on the precise molar ratios of the reactants. The patent suggests a molar ratio of Compound b to Compound c of approximately 1:1 to 1:1.2, ensuring that the valuable chiral amine is fully consumed without a massive excess of the acid component. Similarly, the base and condensing agent are typically used in slight excess (1:1 to 1:2 relative to the acid) to drive the equilibrium towards product formation. Post-reaction workup involves standard liquid-liquid extraction techniques, utilizing aqueous sodium bicarbonate to remove unreacted acid and phosphorous byproducts, followed by brine washes to reduce water content before concentration. The resulting solid is of sufficient purity (>95%) to proceed directly to the next stage or to be used as a high-quality reference standard.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and logistical flow of API production, the transition to this DEPBT/DPPA-based methodology offers compelling economic arguments beyond mere chemical elegance. The primary driver for cost optimization in this context is the drastic simplification of the downstream processing train. By eliminating the formation of dicyclohexylurea (DCU), manufacturers can remove the energy-intensive and time-consuming recrystallization or column chromatography steps that were previously mandatory. This reduction in unit operations directly correlates to lower utility consumption, reduced solvent usage, and decreased labor hours per kilogram of product, leading to substantial cost savings in API manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive and moisture-sensitive reagents like CDI, alongside the removal of toxic carbon tetrachloride, stabilizes raw material costs and reduces hazard management expenses. Furthermore, the high yield (>90% over two steps) implies less starting material is required to produce the same amount of final API, effectively lowering the material cost basis. The ability to avoid chromatographic purification is perhaps the most significant financial benefit, as preparative chromatography is often the most expensive step in small molecule manufacturing due to resin costs and solvent recovery requirements.
- Enhanced Supply Chain Reliability: The reagents employed in this novel process, such as DEPBT, DPPA, and common organic bases, are widely available from multiple global chemical suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions—tolerating a range of solvents like toluene, ethyl acetate, and acetonitrile—provides flexibility in sourcing. If a specific solvent faces supply constraints, the process can be adapted to an alternative without compromising the reaction outcome, ensuring continuous production schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From a regulatory and sustainability standpoint, replacing carbon tetrachloride and minimizing waste generation aligns perfectly with increasingly stringent environmental regulations. The process generates fewer hazardous wastes, simplifying disposal and lowering compliance costs. The scalability is evidenced by the straightforward nature of the reaction, which does not require specialized equipment for handling extremely sensitive reagents or cryogenic temperatures. This ease of commercial scale-up of complex heterocycles ensures that supply can be ramped up quickly to meet market demand without extensive re-engineering of the production line.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, aiming to clarify the operational benefits for potential partners and licensees.
Q: How does the DEPBT method improve upon traditional DCC coupling for Repaglinide?
A: Traditional DCC coupling generates dicyclohexylurea (DCU), a byproduct that is notoriously difficult to remove due to low solubility, often requiring repeated crystallizations. The DEPBT method avoids DCU formation entirely, eliminating the need for complex purification steps and significantly reducing production time.
Q: What is the impact of this synthesis route on optical purity?
A: The novel process utilizing DEPBT or DPPA minimizes racemization risks associated with harsher coupling agents. Experimental data indicates optical purity can reach up to 100%, ensuring the final API meets stringent stereochemical requirements for antidiabetic efficacy.
Q: Can this intermediate synthesis be scaled for industrial production?
A: Yes, the process is designed for scalability. It utilizes common solvents like toluene and avoids hazardous reagents like carbon tetrachloride. Furthermore, the potential for a 'one-pot' telescoped reaction directly to Repaglinide enhances throughput and operational safety for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Repaglinide Intermediate Supplier
The technical advantages outlined in patent CN103012319A represent a significant leap forward in the manufacturing of antidiabetic intermediates, yet realizing this potential requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry to deliver superior value to our global clients. As a dedicated CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are translated into tangible supply chain reliability. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of every batch.
We invite R&D leaders and procurement executives to engage with us to explore how this optimized route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation can drive efficiency and quality in your Repaglinide production.
