Revolutionizing Irbesartan Production: A Deep Dive into Patented Reductive Alkylation and Scalable Manufacturing
Introduction to Advanced Irbesartan Manufacturing Technologies
The global demand for effective antihypertensive agents continues to drive innovation in the synthesis of Angiotensin II receptor blockers, with Irbesartan standing out as a critical therapeutic molecule. As detailed in the seminal patent CN101006064B, filed in 2011, a groundbreaking methodology has been established that fundamentally alters the economic and technical landscape of producing this vital cardiovascular drug. This patent introduces a highly efficient route that bypasses traditional bottlenecks associated with biphenyl tetrazole synthesis, specifically addressing the challenges of halogenation selectivity and intermediate purification. For R&D directors and procurement strategists, understanding this technology is paramount, as it represents a shift towards more sustainable and cost-effective pharmaceutical intermediates manufacturing. The core innovation lies in the ability to utilize crude mixtures of brominated precursors directly, thereby streamlining the supply chain and reducing the environmental footprint of the production process.
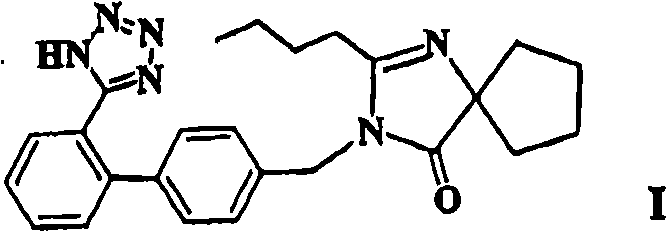
Furthermore, the structural complexity of Irbesartan, characterized by its biphenyl moiety and spiro-fused imidazolone ring, demands precise synthetic control to ensure high purity and consistent bioavailability. The patented approach not only secures the integrity of the final active pharmaceutical ingredient (API) but also offers a scalable framework that aligns with modern Good Manufacturing Practice (GMP) standards. By leveraging this advanced chemistry, manufacturers can achieve superior impurity profiles, which is a critical metric for regulatory approval in major markets such as the US and EU. Consequently, this technology serves as a cornerstone for any organization aiming to establish itself as a reliable API intermediate supplier in the competitive cardiovascular sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Irbesartan and its precursors have long been plagued by inefficiencies related to the bromination of the biphenyl starting material. In standard protocols, achieving high selectivity for the monobrominated species is notoriously difficult, often resulting in significant quantities of dibrominated and tribrominated byproducts. Historically, these over-brominated impurities were considered waste, necessitating expensive and time-consuming purification steps such as column chromatography or repeated recrystallization to isolate the pure monobromide required for the subsequent alkylation. This not only drives up the cost of goods sold (COGS) due to material loss but also creates substantial chemical waste, posing challenges for environmental compliance and waste management. Moreover, the strict requirement for high-purity monobromide limits the overall throughput of the manufacturing line, creating bottlenecks that can disrupt supply continuity for downstream API production.
The Novel Approach
The methodology disclosed in patent CN101006064B presents a paradigm shift by transforming these former liabilities into assets through a clever telescoping strategy. Instead of discarding the dibrominated byproducts, the novel process employs a simultaneous reduction and alkylation step. As illustrated in the comprehensive reaction scheme below, the crude mixture containing both monobromo (IVa) and dibromo (IVb) compounds is reacted directly with the spiro-imidazolone precursor (Compound V). This eliminates the need for isolating the pure monobromide, effectively turning a multi-step purification nightmare into a single, streamlined operation. This approach drastically simplifies the workflow, allowing for the direct conversion of a crude bromination mixture into the key intermediate (Compound II) with high efficiency.
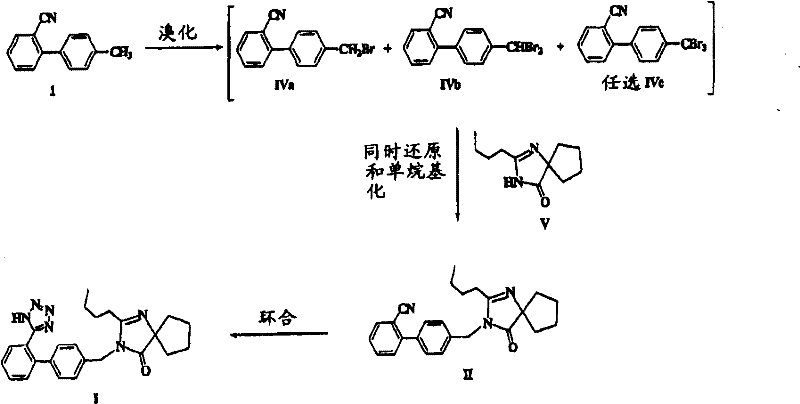
By integrating the reduction of the dibromide directly into the alkylation phase, the process maximizes atom economy and minimizes solvent usage. This is particularly advantageous for cost reduction in pharmaceutical intermediates manufacturing, as it reduces the number of unit operations and the associated labor and energy costs. The ability to tolerate a mixture of brominated species (with ratios ranging from 1:99 to 99:1) provides immense flexibility in the upstream bromination step, allowing operators to optimize for speed and yield rather than obsessing over perfect selectivity. This robustness is a key differentiator for commercial scale-up, ensuring that minor fluctuations in the initial reaction conditions do not compromise the quality or quantity of the final product.
Mechanistic Insights into Reductive Alkylation and Impurity Control
At the heart of this technological breakthrough is the sophisticated interplay between the alkylating agent, the base, and the reducing agent during the formation of Compound II. The mechanism relies on the use of a dialkyl phosphite, such as diethyl phosphite, which acts as a mild yet effective reducing agent in the presence of a strong base like sodium hydride or aqueous potassium hydroxide. When the mixture of brominated biphenyls encounters the deprotonated spiro-imidazolone (Compound V), the monobromide (IVa) undergoes a standard nucleophilic substitution to form the C-N bond. Simultaneously, the dibromide (IVb), which would traditionally be an unreactive impurity in this context, is selectively reduced by the phosphite species back to the monobromide state in situ. This regenerated monobromide then immediately participates in the alkylation reaction, ensuring that nearly all carbon feedstock is converted into the desired product rather than being lost as waste.
This mechanistic elegance extends to impurity control, a primary concern for R&D directors focused on regulatory compliance. The use of phase transfer catalysts, such as methyltri-n-butylammonium chloride (MTBAC), further enhances the reaction kinetics and selectivity, particularly when using aqueous base systems. This facilitates the efficient mass transfer of reactants across phase boundaries, minimizing side reactions that could lead to hard-to-remove impurities. Furthermore, the subsequent crystallization of Compound II using solvents like methyl tert-butyl ether (MTBE) or isopropanol (IPA) acts as a powerful purification gate. The patent highlights that these solvents can be recycled from the washing steps of previous batches, creating a closed-loop system that maintains high purity specifications while reducing solvent procurement costs. This level of control over the impurity profile is essential for ensuring the high-purity pharmaceutical intermediates required for final API synthesis.
How to Synthesize Irbesartan Efficiently
The implementation of this patented route requires careful attention to reaction conditions, particularly temperature control and reagent stoichiometry, to maximize the benefits of the reductive alkylation strategy. The process begins with the bromination of 4'-methylbiphenyl-2-carbonitrile, followed by the critical telescoping step where reduction and alkylation occur concurrently. Detailed operational parameters, including specific molar ratios and temperature ranges, are crucial for replicating the high yields reported in the patent examples. For process chemists looking to adopt this methodology, understanding the nuances of the workup and crystallization phases is equally important to ensure the isolation of Compound II in its substantially pure form. The following guide outlines the standardized synthesis steps derived from the patent data to facilitate technology transfer and scale-up.
- Bromination of 4'-methylbiphenyl-2-carbonitrile using NaBrO3/HBr to generate a mixture of monobromo (IVa) and dibromo (IVb) intermediates without strict separation.
- Simultaneous reductive alkylation where the bromo-mixture reacts with the spiro-imidazolone (Compound V) in the presence of a base and dialkyl phosphite reducing agent.
- Cyclization of the nitrile intermediate (Compound II) with sodium azide to form the final tetrazole ring of Irbesartan (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic advantages that extend beyond simple chemistry. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly translates to enhanced supply chain reliability and reduced operational risk. By eliminating the need for intermediate purification of the monobromide, the process reduces the number of processing days and the dependency on specialized separation equipment. This streamlining effect allows for faster batch turnover times, enabling manufacturers to respond more agilely to market demand fluctuations. Additionally, the robustness of the reaction against variations in the bromination mixture means that raw material specifications can be relaxed slightly without impacting final quality, potentially widening the pool of qualified suppliers for the starting materials.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the elimination of wasteful purification steps and the efficient utilization of raw materials. By converting what was previously waste (dibromides) into valuable product, the overall material yield is significantly improved, lowering the effective cost per kilogram of the intermediate. Furthermore, the ability to recycle solvents like MTBE and IPA from the crystallization and washing steps creates a circular economy within the plant, drastically cutting down on solvent purchase and disposal costs. This holistic approach to cost optimization ensures that the production of complex pharmaceutical intermediates remains economically viable even in the face of rising raw material prices.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that have multiple failure points; this simplified route mitigates those risks by reducing the total number of unit operations. The tolerance for mixed bromination products means that the upstream supply of the brominated intermediate does not require ultra-high purity, making sourcing easier and more resilient. Moreover, the use of common, commercially available reagents such as diethyl phosphite and standard bases ensures that the supply chain is not dependent on exotic or single-source catalysts. This diversification of input materials strengthens the overall supply network, reducing lead time for high-purity intermediates and ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process is exceptionally well-suited for commercial scale-up of complex APIs. The reduction in solvent usage and chemical waste aligns with increasingly stringent global environmental regulations, reducing the burden on waste treatment facilities. The crystallization protocol, which allows for solvent recycling, further minimizes the environmental footprint of the operation. Scalability is enhanced because the reaction conditions (0-20°C for bromination, ambient for alkylation) are easily manageable in large-scale reactors without requiring extreme cryogenic or high-pressure equipment. This ease of scale-up ensures that production capacity can be expanded rapidly to meet growing global demand for cardiovascular medications without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Irbesartan synthesis route. These insights are derived directly from the experimental data and claims within patent CN101006064B, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement teams assessing the quality of suppliers utilizing this technology.
Q: How does the new process handle over-brominated impurities?
A: Unlike conventional methods that require costly purification of monobromides, this patented process utilizes a reducing agent (dialkyl phosphite) to selectively convert dibrominated byproducts back into reactive monobromides in situ during the alkylation step.
Q: What are the yield expectations for the key intermediate Compound II?
A: The patent data indicates robust yields typically ranging from 85% to 88% for Compound II, achieved through efficient crystallization using solvents like MTBE or IPA, which can be recycled for subsequent batches.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is designed for scalability by eliminating complex chromatographic separations and enabling solvent recycling, significantly reducing waste generation and operational costs associated with large-volume API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Irbesartan Supplier
As the pharmaceutical industry continues to evolve, the need for partners who can bridge the gap between innovative patent chemistry and commercial reality has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging deep technical expertise to deliver high-quality intermediates and APIs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101006064B are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Irbesartan intermediate meets the exacting standards required by global regulatory bodies.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains and reduce manufacturing costs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of high-quality Irbesartan intermediates, empowering your organization to lead in the competitive cardiovascular therapeutics market.
