Advanced Synthetic Route for Cefotiam Intermediates Enhancing Commercial Scalability and Purity
Advanced Synthetic Route for Cefotiam Intermediates Enhancing Commercial Scalability and Purity
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with economic feasibility, particularly for complex beta-lactam antibiotics. Patent CN1195761C introduces a transformative methodology for the production of Cefotiam, a potent second-generation cephalosporin, by leveraging a novel N-formyl protected intermediate. This intellectual property details a sophisticated two-step sequence that begins with the acylation of a specific cephalosporin nucleus using an activated side chain, followed by a controlled deformylation process. The significance of this patent lies in its ability to generate an isolable, crystalline intermediate, which serves as a critical quality control checkpoint before the final drug substance is formed. By shifting away from traditional one-pot syntheses that often struggle with impurity profiles, this approach offers a distinct strategic advantage for manufacturers aiming to secure a reliable cephalosporin intermediate supplier status in the global market.
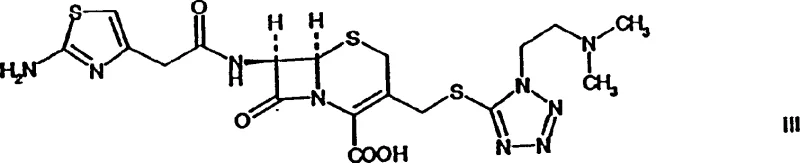
This structural innovation is not merely academic; it addresses fundamental challenges in process chemistry regarding scalability and reproducibility. The patent explicitly outlines the preparation of Cefotiam hexetil, an oral prodrug, highlighting the versatility of the intermediate for various dosage forms. For R&D directors and process chemists, the introduction of the formyl protecting group on the aminothiazole side chain represents a clever maneuver to modulate reactivity and solubility during the coupling phase. This level of molecular engineering ensures that the final active pharmaceutical ingredient meets the stringent regulatory standards required for parenteral and oral administration, thereby reducing the risk of costly late-stage development failures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cephalosporin derivatives like Cefotiam has been plagued by the difficulties associated with direct acylation of the 7-amino group on the cephem nucleus. Conventional methods often involve the use of unprotected side chain acids or their activated esters, which can lead to uncontrolled reactions and the formation of difficult-to-remove regioisomers. Furthermore, traditional processes frequently rely on thiourea-mediated cyclization steps that require harsh conditions, potentially compromising the stability of the sensitive beta-lactam ring. These legacy approaches often result in oily residues that are challenging to purify, necessitating extensive chromatographic separations that are impractical for large-scale manufacturing. The lack of a solid, isolable intermediate in these older routes means that impurities carry through to the final step, drastically affecting the overall yield and requiring expensive remediation strategies to meet pharmacopeial specifications.
The Novel Approach
In stark contrast, the methodology described in the patent utilizes a pre-formed, activated acylating agent bearing a formyl-protected amine, which fundamentally alters the reaction landscape. By employing this specific activated species, the process achieves a highly selective acylation at the 7-position of the cephem nucleus without significant side reactions. The resulting product, an N-formyl cefotiam derivative, possesses the remarkable property of forming stable crystalline salts, particularly when paired with specific organic bases. This crystallinity is a game-changer for process engineering, as it allows for purification via simple recrystallization rather than complex chromatography. The ability to isolate this key intermediate ensures that only material of defined quality proceeds to the final deprotection step, thereby guaranteeing a superior impurity profile for the final Cefotiam product.
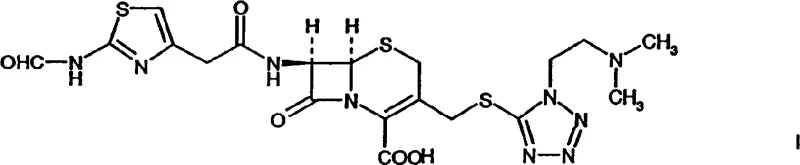
The novelty of this approach is further underscored by the specific activation strategy employed for the side chain. Instead of generic activation methods, the patent advocates for the use of acid halides, specifically acid chlorides derived from the formylated aminothiazole acetic acid. This high-energy electrophile reacts rapidly and efficiently with the nucleophilic amine of the cephem nucleus under mild conditions. The subsequent deformylation step is equally elegant, utilizing mild aqueous acidic conditions to reveal the free amine without degrading the sensitive beta-lactam core. This modular approach decouples the coupling and deprotection events, providing process chemists with greater control over each unit operation and facilitating easier troubleshooting during technology transfer.
Mechanistic Insights into Acylation and Deformylation Chemistry
The core of this synthetic strategy relies on the precise manipulation of nucleophilicity and electrophilicity within a sterically crowded environment. The reaction initiates with the activation of 2-(2-formylaminothiazol-4-yl)acetic acid, typically converting it into the corresponding acid chloride (Formula V) using reagents such as phosphorus pentachloride. This activation generates a highly reactive acyl chloride species that is primed for nucleophilic attack. When introduced to the 7-aminocephalosporanic acid derivative (7-ACMT), the primary amine at the 7-position acts as the nucleophile. The presence of a tertiary amine base, such as tri-n-butylamine, is mechanistically critical; it serves not only to scavenge the hydrochloric acid generated during the amide bond formation but also to stabilize the transition state. The base interacts with the developing positive charge, lowering the activation energy and driving the equilibrium towards the desired amide product.
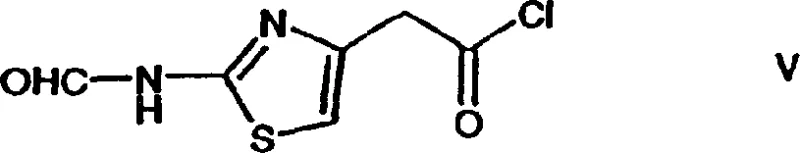
Following the acylation, the mechanism shifts to the stabilization of the product through salt formation. The patent highlights that the N-formyl cefotiam intermediate can be isolated as a salt with various amines, with tri-n-butylamine salts being particularly favorable due to their crystalline nature. This phenomenon suggests a specific lattice energy arrangement that favors solid-state formation over remaining in solution, a property that is exploited for purification. The final step involves the hydrolytic removal of the formyl group. Under aqueous acidic conditions, the amide bond of the formyl group is protonated, making the carbonyl carbon susceptible to nucleophilic attack by water. This cleavage regenerates the free primary amine on the thiazole ring, yielding the biologically active Cefotiam structure. The mildness of this acid hydrolysis is crucial, as stronger conditions could open the beta-lactam ring, rendering the molecule inactive.
Impurity control is intrinsically built into this mechanistic pathway. The formyl group acts as a protecting group that prevents self-polymerization or unwanted side reactions of the aminothiazole moiety during the coupling phase. In conventional unprotected routes, the free amine on the side chain can sometimes react with other electrophilic sites or catalyze degradation of the beta-lactam ring. By keeping this amine masked until the very end, the process minimizes the generation of polymeric impurities and dimers. Furthermore, the crystallization of the intermediate acts as a physical barrier to impurities; non-crystalline byproducts remain in the mother liquor and are discarded during filtration. This dual mechanism of chemical protection and physical purification ensures that the final API meets the rigorous standards expected of a high-purity pharmaceutical intermediate.
How to Synthesize Cefotiam Efficiently
The practical execution of this synthesis requires careful attention to temperature control and stoichiometry to maximize the benefits of the patented route. The process begins with the preparation of the activated acid chloride, which must be handled under anhydrous conditions to prevent premature hydrolysis. Subsequent coupling with the cephem nucleus is performed at low temperatures to maintain stereochemical integrity and prevent thermal degradation. The isolation of the intermediate salt is a critical unit operation where solvent selection and anti-solvent addition rates determine the crystal habit and purity. Finally, the deformylation step requires precise pH and temperature monitoring to ensure complete deprotection without compromising the beta-lactam stability. For a detailed breakdown of the specific operational parameters, reagent grades, and workup procedures, please refer to the standardized synthesis guide below.
- Acylation of 7-ACMT with activated 2-formylaminothiazolylacetyl chloride in the presence of a tertiary amine base to form the crystalline N-formyl cefotiam salt.
- Isolation of the intermediate salt through precipitation using anti-solvents, ensuring high purity before the final deprotection step.
- Deformylation of the isolated intermediate using aqueous acidic media at controlled temperatures to yield the final Cefotiam product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthetic route offers profound advantages for procurement managers and supply chain leaders who are tasked with minimizing costs while ensuring continuity of supply. The primary economic driver here is the drastic simplification of the purification process. Traditional methods that yield oily or amorphous intermediates often require resource-intensive purification techniques such as preparative HPLC or multiple extraction cycles, which consume vast amounts of solvents and time. By contrast, the ability to isolate a crystalline intermediate allows for purification via simple filtration and washing, significantly reducing solvent consumption and waste disposal costs. This efficiency translates directly into a lower cost of goods sold (COGS), allowing manufacturers to offer more competitive pricing without sacrificing margin.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps represents a major operational saving. In traditional synthesis, the inability to crystallize intermediates often forces manufacturers to use expensive silica gel columns or specialized resin beds, which are slow and have limited throughput. The crystalline nature of the N-formyl intermediate described in this patent enables the use of standard centrifuges and dryers, equipment that is ubiquitous in chemical plants and far cheaper to operate. Additionally, the high selectivity of the acylation step reduces the formation of byproducts, meaning less raw material is wasted on generating impurities that must later be removed. This atom economy, combined with reduced solvent usage for purification, creates a leaner manufacturing process that is inherently more cost-effective.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the robustness of the chemical process. Processes that rely on sensitive catalysts or unstable intermediates are prone to batch failures, which can disrupt delivery schedules and damage customer relationships. The route described in CN1195761C utilizes stable, commercially available reagents and avoids the use of precious metal catalysts that are subject to volatile market pricing and supply shortages. The crystalline intermediate acts as a buffer in the production schedule; it can be manufactured in large batches, tested for quality, and stored for extended periods before being converted to the final API. This decoupling of steps allows for better inventory management and ensures that downstream formulation teams have a consistent supply of high-quality material, regardless of upstream fluctuations.
- Scalability and Environmental Compliance: Scaling a chemical process from the laboratory to commercial production often reveals hidden bottlenecks, particularly regarding heat transfer and mixing in viscous solutions. The precipitation of a solid intermediate improves the rheology of the reaction mixture, making heat removal and agitation more efficient on a large scale. Furthermore, the reduction in solvent volume and the avoidance of heavy metal catalysts align perfectly with modern environmental, health, and safety (EHS) regulations. A process that generates less hazardous waste and consumes fewer resources is easier to permit and operate in regulated jurisdictions. This environmental compliance reduces the risk of regulatory shutdowns and enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their supply chains. The following questions address common concerns regarding the implementation of this technology, focusing on the practical implications of the crystalline intermediate strategy and the specific reaction conditions required. These insights are derived directly from the technical disclosures within the patent documentation, providing a factual basis for decision-making regarding technology adoption and vendor qualification.
Q: What is the primary advantage of the N-formyl intermediate route?
A: The primary advantage is the ability to isolate the intermediate as a stable, crystalline salt, which significantly simplifies purification and ensures higher final product purity compared to direct synthesis methods.
Q: Why is tri-n-butylamine preferred as the base in this process?
A: Tri-n-butylamine is preferred because it effectively neutralizes the acid byproducts while facilitating the formation of a crystalline salt of the intermediate, which improves handling and filtration properties on an industrial scale.
Q: How does this method impact supply chain stability for cephalosporins?
A: By utilizing robust reaction conditions and avoiding complex transition metal catalysts, this method reduces the risk of batch failures and heavy metal contamination, leading to more consistent production timelines and reliable supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Intermediate Supplier
The technical sophistication of the route described in CN1195761C demands a manufacturing partner with deep expertise in beta-lactam chemistry and a commitment to quality excellence. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors capable of maintaining the precise low-temperature conditions required for the acylation step, alongside advanced filtration systems designed to handle crystalline intermediates efficiently. We understand that consistency is key in pharmaceutical manufacturing, which is why our operations are governed by stringent purity specifications and supported by rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest international standards.
We invite procurement leaders and R&D directors to explore how our capabilities can optimize your supply chain for cephalosporin intermediates. By leveraging our technical expertise, you can achieve significant process efficiencies and secure a stable source of high-quality materials. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you need specific COA data to validate our quality or route feasibility assessments to determine the best fit for your portfolio, our team is ready to provide the detailed support necessary to drive your projects forward successfully.
