Revolutionizing Antibiotic Synthesis: Scalable Production of Novel C-3 Substituted Cephalosporin Intermediates
Revolutionizing Antibiotic Synthesis: Scalable Production of Novel C-3 Substituted Cephalosporin Intermediates
The escalating crisis of antibiotic resistance, particularly among Gram-positive pathogens like Methicillin-resistant Staphylococcus aureus (MRSA) and Penicillin-resistant Streptococcus pneumoniae (PRSP), demands urgent innovation in cephalosporin chemistry. Patent CN1727347A introduces a groundbreaking class of C-3-methylene-azacyclo substituted-guanylthioacetylamino cephalosporins that address these critical therapeutic gaps. This technology represents a significant leap forward for pharmaceutical manufacturers seeking to expand their antibiotic portfolios with high-efficacy agents. By modifying both the C-3 and C-7 positions of the cephem nucleus, this invention achieves a synergistic enhancement in antimicrobial spectrum and metabolic stability. For R&D directors and procurement strategists, understanding the synthetic accessibility and structural novelty of these intermediates is paramount for securing a competitive edge in the global API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional third-generation cephalosporins, while effective against many Gram-negative organisms, often exhibit diminished potency against resistant Gram-positive bacteria. Conventional modification strategies typically focus on either the C-3 or C-7 position in isolation, which limits the potential for broad-spectrum optimization. For instance, standard C-3 modifications involving sulfydryl groups, such as those found in cefoperazone or cefmetazole, improve stability but may not sufficiently overcome resistance mechanisms in evolving bacterial strains. Furthermore, existing synthetic routes often suffer from complex purification steps, low yields due to side reactions, or the use of hazardous reagents that complicate environmental compliance. These limitations create bottlenecks in the supply chain, leading to higher production costs and inconsistent availability of high-purity active pharmaceutical ingredients.
The Novel Approach
The novel approach detailed in this patent overcomes these hurdles by introducing a unique combination of substituents: a nitrogen heterocyclic ring at the C-3 position and an amidine thioacetamide group at the C-7 position. This dual-modification strategy creates a molecular architecture that not only penetrates bacterial cell walls more effectively but also resists enzymatic degradation by beta-lactamases. 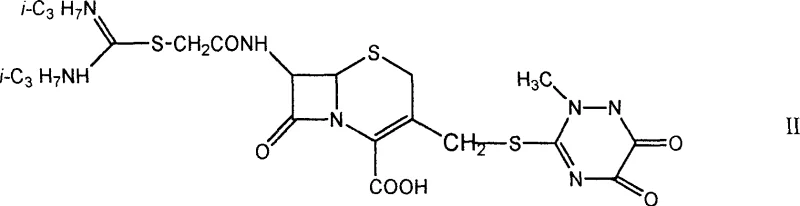 As illustrated in the structural representation, the specific arrangement of the triazine ring at C-3 and the di-isopropyl amidine group at C-7 provides steric and electronic properties that enhance binding affinity to penicillin-binding proteins. This structural innovation translates directly into superior clinical performance, offering a robust solution for treating infections caused by resistant pathogens while maintaining a favorable safety profile for mammalian systems.
As illustrated in the structural representation, the specific arrangement of the triazine ring at C-3 and the di-isopropyl amidine group at C-7 provides steric and electronic properties that enhance binding affinity to penicillin-binding proteins. This structural innovation translates directly into superior clinical performance, offering a robust solution for treating infections caused by resistant pathogens while maintaining a favorable safety profile for mammalian systems.
Mechanistic Insights into Nucleophilic Substitution and Amidination
The core of this synthesis lies in a precise two-step functionalization of the 7-amino cephalosporanic acid derivative. Initially, the free amine group at the C-7 position undergoes acylation with bromoacetyl bromide. This reaction is highly sensitive to pH and temperature, requiring careful control to prevent hydrolysis of the beta-lactam ring. The use of organic bases like triethylamine or mineral alkalis such as sodium bicarbonate ensures the amine remains nucleophilic enough to attack the carbonyl carbon of the bromoacetyl bromide without degrading the sensitive cephem core. Following this, the resulting bromoacetyl intermediate serves as an electrophile for the subsequent reaction with N,N-disubstituted thioureas. 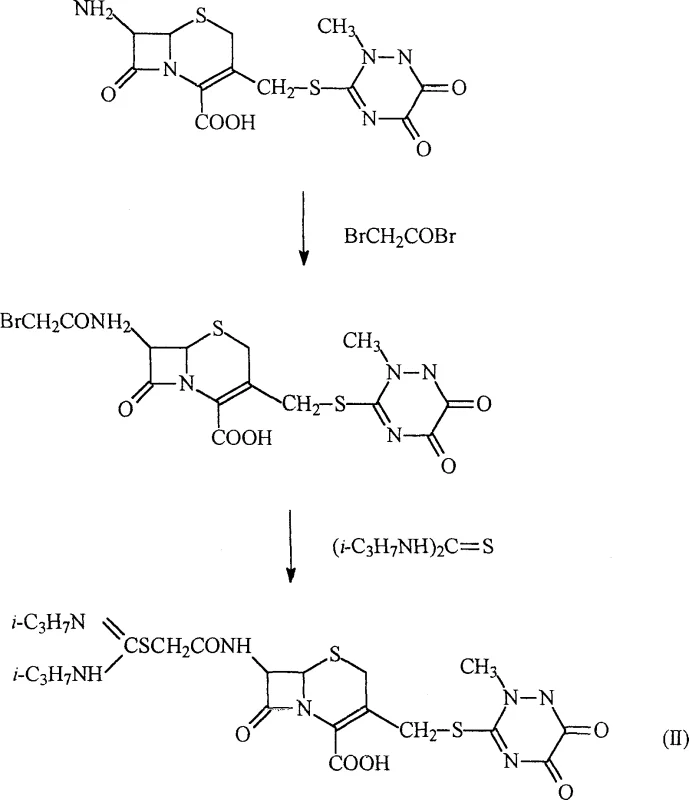 This nucleophilic substitution displaces the bromide ion, forming the stable amidine thioacetamide linkage. The mechanism is driven by the strong nucleophilicity of the sulfur atom in the thiourea, facilitated by the electron-withdrawing nature of the adjacent carbonyl group.
This nucleophilic substitution displaces the bromide ion, forming the stable amidine thioacetamide linkage. The mechanism is driven by the strong nucleophilicity of the sulfur atom in the thiourea, facilitated by the electron-withdrawing nature of the adjacent carbonyl group.
Impurity control is critical in this process, particularly regarding the stereochemistry at the C-6 and C-7 positions and the integrity of the double bond in the dihydrothiazine ring. The patent specifies reaction temperatures between 0°C and 50°C, with a preferred range of 10°C to 40°C, to minimize epimerization and ring-opening side reactions. Solvent selection plays a pivotal role; mixtures of ketones (acetone, MIBK) and water, or halogenated alkanes like dichloromethane, provide the optimal polarity balance to keep intermediates in solution while allowing for easy precipitation of the final product upon acidification. This meticulous control over reaction parameters ensures a final product purity exceeding 90%, meeting the rigorous standards required for pharmaceutical intermediates intended for human use.
How to Synthesize Cephalo Piperazine Amidine Efficiently
The synthesis of this high-value intermediate follows a streamlined protocol designed for reproducibility and scale-up. The process begins with the dissolution of the starting material, 7-amino-3-((2,5-dihydro-6-hydroxy-2-methyl-5-oxo-as-triazine-3-yl) methyl)-Cephalosporanic acid (7-ACT), in a suitable solvent system. pH adjustment is performed to ensure complete solubilization before the addition of bromoacetyl bromide. After the formation of the bromoacetyl intermediate, the mixture is acidified to precipitate the solid, which is then filtered and dried. The second stage involves reacting this intermediate with N,N-di-isopropyl thiourea in a solvent like dichloromethane or acetone, followed by reflux and cooling to crystallize the final product.
- Dissolve 7-amino-3-methylene nitrogen heterocyclic substituted cephalosporanic acid in a solvent system and adjust pH with organic base.
- React the solution with bromoacetyl bromide at controlled temperatures (0-50°C) to form the bromoacetyl intermediate.
- Acidify the mixture, separate the intermediate, and react with N,N-disubstituted thiourea to obtain the final amidine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical novelty. The reliance on commodity chemicals such as bromoacetyl bromide and di-isopropyl thiourea eliminates the dependency on exotic or scarce reagents, thereby stabilizing raw material costs and reducing supply chain volatility. The process operates under relatively mild conditions, avoiding the need for high-pressure reactors or cryogenic cooling, which significantly lowers capital expenditure and energy consumption during manufacturing. Furthermore, the workup procedure relies on simple pH adjustments and filtration, removing the need for complex chromatographic purifications that often bottleneck production throughput.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of inexpensive, widely available solvents like acetone and dichloromethane drastically simplify the cost structure of the synthesis. By avoiding expensive chiral auxiliaries or specialized reagents, the overall cost of goods sold (COGS) is substantially reduced, allowing for more competitive pricing in the generic antibiotic market. The high yield and purity achieved through straightforward crystallization steps further minimize waste disposal costs and maximize material efficiency.
- Enhanced Supply Chain Reliability: The starting materials, specifically the 7-ACT derivative and the thiourea reagents, are commercially established intermediates with robust global supply networks. This ensures a continuous flow of raw materials, mitigating the risk of production stoppages due to supplier shortages. The simplicity of the reaction setup also means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand without extensive requalification of equipment.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods that might utilize heavy metals or toxic organometallic reagents. The aqueous workup and ability to recycle solvents like acetone align with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility. This compliance is increasingly critical for maintaining operational licenses and meeting the sustainability goals of multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel cephalosporin intermediates. Understanding these details is essential for integrating this technology into existing production lines and ensuring regulatory compliance.
Q: What is the primary advantage of the C-3 methylene nitrogen heterocyclic substitution in this cephalosporin derivative?
A: The C-3 methylene nitrogen heterocyclic substitution significantly enhances antimicrobial activity against Gram-positive bacteria, including resistant strains like MRSA, while maintaining stability against beta-lactamases.
Q: Which solvents are preferred for the bromoacetylation step in this synthesis?
A: The patent specifies alkyl ketones such as acetone and methyl isobutyl ketone (MIBK), alkyl alcohols like ethanol and isopropanol, or halogenated alkanes such as dichloromethane as effective solvent systems.
Q: How does this synthesis method impact the scalability of antibiotic production?
A: The method utilizes readily available reagents like bromoacetyl bromide and N,N-diisopropyl thiourea under mild conditions, allowing for straightforward workup via pH adjustment and filtration, which facilitates large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalosporin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cephalosporin intermediate meets the highest international standards. Our commitment to quality assurance ensures that your downstream API synthesis proceeds without interruption or compromise.
We invite you to collaborate with us to leverage this advanced synthetic technology for your antibiotic portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing objectives, ensuring a seamless transition from laboratory discovery to commercial success.
