Advanced Manufacturing of Broad-Spectrum Cephalosporin Intermediates for Global Pharma Supply Chains
The pharmaceutical landscape continuously demands more potent and versatile antibiotics to combat evolving bacterial resistance, and patent CN1022629C presents a significant breakthrough in this arena by detailing a sophisticated process for preparing novel cephalosporin compounds. This intellectual property outlines a robust synthetic pathway for generating cephalosporin derivatives characterized by a unique 7-position substituent containing a (Z)-2-(2-aminothiazol-4-yl)-2-(1,5-dihydroxy-4-pyridon-2-yl)alkoxyiminoacetamide group. For R&D directors and procurement specialists, this technology represents a critical advancement because it addresses the persistent challenge of treating infections caused by Gram-negative microorganisms, particularly Pseudomonas aeruginosa, which are often resistant to earlier generations of beta-lactams. The method described involves intricate organic transformations that convert readily available precursors into high-value intermediates, offering a viable route for the commercial production of next-generation antibacterial agents. By leveraging this patented methodology, manufacturers can access a new class of therapeutic compounds that combine broad-spectrum efficacy with favorable pharmacological properties, thereby securing a competitive edge in the global antibiotic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for cephalosporin antibiotics often struggle with achieving the optimal balance between broad-spectrum activity and metabolic stability, particularly when targeting resistant Gram-negative strains. Conventional side chains frequently lack the specific electronic and steric configurations necessary to penetrate the outer membrane of difficult pathogens like Pseudomonas aeruginosa effectively. Furthermore, older methodologies may rely on harsh reaction conditions or unstable intermediates that complicate purification and reduce overall yield, leading to higher production costs and inconsistent quality control. The presence of impurities in final API batches can necessitate extensive downstream processing, which not only increases manufacturing lead times but also introduces potential safety risks associated with residual solvents or reagents. Consequently, there is a pressing industry need for synthetic strategies that can introduce complex, highly functionalized side chains under mild conditions while maintaining high stereochemical purity and structural integrity throughout the multi-step sequence.
The Novel Approach
The innovative process disclosed in the patent overcomes these historical bottlenecks by introducing a specialized 1,5-dihydroxy-4-pyridone moiety at the 7-position of the cephalosporin nucleus, which dramatically enhances antibacterial potency. This approach utilizes a modular synthetic strategy where the side chain is constructed separately and then coupled to the beta-lactam core, allowing for precise control over the stereochemistry and functional group orientation. 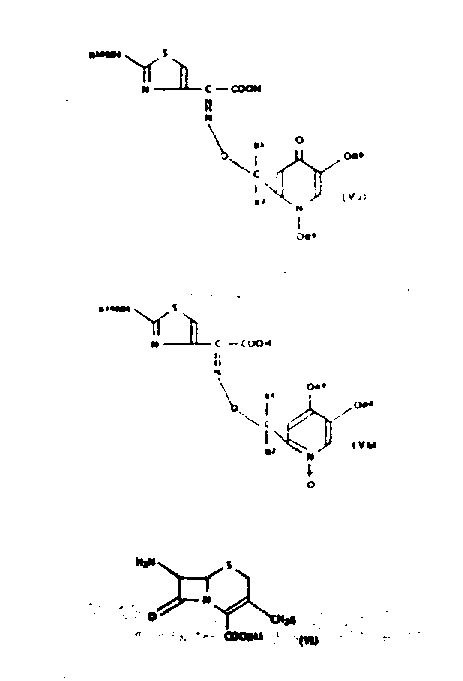 The method employs protective group chemistry, such as p-methoxybenzyl groups, to stabilize reactive hydroxyl functions during the coupling steps, ensuring that the sensitive beta-lactam ring remains intact. Additionally, the process offers flexibility through two distinct pathways: Method A involves direct acylation of the 7-amino group, while Method B utilizes nucleophilic substitution at the 3-position to introduce diverse heterocyclic groups. This dual-pathway capability allows manufacturers to tailor the synthesis based on available equipment and raw material inventory, providing significant operational flexibility. The result is a high-purity intermediate that exhibits superior water solubility, a critical factor for parenteral formulations, without compromising the stability required for long-term storage and distribution.
The method employs protective group chemistry, such as p-methoxybenzyl groups, to stabilize reactive hydroxyl functions during the coupling steps, ensuring that the sensitive beta-lactam ring remains intact. Additionally, the process offers flexibility through two distinct pathways: Method A involves direct acylation of the 7-amino group, while Method B utilizes nucleophilic substitution at the 3-position to introduce diverse heterocyclic groups. This dual-pathway capability allows manufacturers to tailor the synthesis based on available equipment and raw material inventory, providing significant operational flexibility. The result is a high-purity intermediate that exhibits superior water solubility, a critical factor for parenteral formulations, without compromising the stability required for long-term storage and distribution.
Mechanistic Insights into Pyridone Side Chain Construction and Coupling
The core of this technological advancement lies in the meticulous construction of the 1,5-dihydroxy-4-pyridone side chain, which begins with the derivatization of kojic acid. As illustrated in the reaction schemes, the process initiates with the protection of kojic acid hydroxyl groups using alkylating agents like p-methoxybenzyl chloride in the presence of a base such as potassium carbonate. 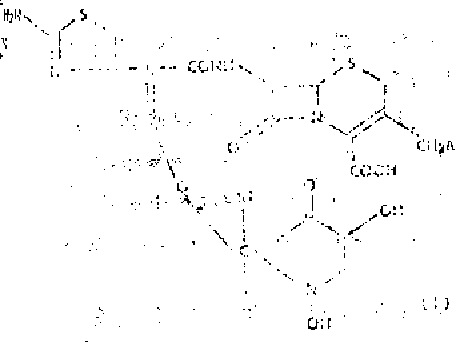 This step is crucial for preventing unwanted side reactions during subsequent transformations and ensures that the final deprotection yields the active dihydroxy species cleanly. The protected intermediate is then converted into a reactive oxime derivative through condensation with an aminothiazole glyoxylate equivalent, establishing the critical Z-configuration required for biological activity. The mechanistic precision here is vital; the formation of the syn-oxime isomer is thermodynamically favored and kinetically controlled to maximize the yield of the active pharmaceutical ingredient. Following side chain assembly, the activated acid derivative is coupled to the 7-amino cephalosporin nucleus using standard peptide coupling reagents or mixed anhydride techniques. This acylation step must be carefully monitored to prevent epimerization at the chiral centers, which could render the final product inactive. The subsequent nucleophilic displacement at the 3-position, often involving quaternization of a pyridine or thiazole ring, further diversifies the molecule's pharmacokinetic profile, allowing for fine-tuning of tissue penetration and excretion rates.
This step is crucial for preventing unwanted side reactions during subsequent transformations and ensures that the final deprotection yields the active dihydroxy species cleanly. The protected intermediate is then converted into a reactive oxime derivative through condensation with an aminothiazole glyoxylate equivalent, establishing the critical Z-configuration required for biological activity. The mechanistic precision here is vital; the formation of the syn-oxime isomer is thermodynamically favored and kinetically controlled to maximize the yield of the active pharmaceutical ingredient. Following side chain assembly, the activated acid derivative is coupled to the 7-amino cephalosporin nucleus using standard peptide coupling reagents or mixed anhydride techniques. This acylation step must be carefully monitored to prevent epimerization at the chiral centers, which could render the final product inactive. The subsequent nucleophilic displacement at the 3-position, often involving quaternization of a pyridine or thiazole ring, further diversifies the molecule's pharmacokinetic profile, allowing for fine-tuning of tissue penetration and excretion rates.
Impurity control is inherently built into this mechanism through the strategic use of crystallizable intermediates and chromatographic purification steps. The patent specifies the use of macroporous adsorption resins, such as Diaion HP-20, to isolate the final product from reaction byproducts and inorganic salts. This purification technique is highly effective for polar cephalosporin salts, removing trace metal catalysts and organic impurities that could trigger allergic reactions in patients. By understanding the mechanistic nuances of the pyridone ring formation and the subsequent coupling reactions, process chemists can identify critical control points where impurities are likely to generate. For instance, maintaining strict temperature controls during the oxime formation prevents the generation of anti-isomers, while careful pH adjustment during the final workup ensures the precipitation of the desired sodium salt form. This level of mechanistic understanding translates directly into a more robust and reproducible manufacturing process, minimizing batch-to-batch variability and ensuring that every kilogram of produced intermediate meets stringent regulatory specifications for identity, strength, and purity.
How to Synthesize Novel Cephalosporin Intermediates Efficiently
Implementing this synthesis requires a disciplined approach to reaction engineering and quality assurance to ensure the successful translation of laboratory protocols to commercial scale. The process begins with the preparation of the protected pyridone acid, followed by its activation and coupling to the cephalosporin nucleus, and concludes with the introduction of the 3-position substituent and final deprotection. Each stage demands precise stoichiometry, solvent selection, and temperature management to maintain high yields and product integrity. Detailed standardized operating procedures are essential for managing the reactive intermediates and ensuring operator safety during the handling of halogenating agents and strong acids.
- Protect kojic acid derivatives using p-methoxybenzyl groups to form stable pyridone precursors suitable for further functionalization.
- Convert the protected hydroxymethyl group into a chloromethyl or reactive leaving group to facilitate coupling with aminothiazole oxime acids.
- Perform acylation of the 7-amino cephalosporin nucleus followed by nucleophilic substitution at the 3-position to introduce the desired heterocyclic moiety.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The reliance on kojic acid as a starting material is a significant advantage, as it is a fermentation-derived product available in large quantities from established global suppliers, reducing the risk of raw material shortages. Unlike processes that depend on scarce or geopolitically sensitive reagents, this pathway leverages commodity chemicals, ensuring a stable and predictable cost structure over the long term. Furthermore, the modular nature of the synthesis allows for the decoupling of side chain production from the final cephalosporin coupling, enabling parallel manufacturing streams that can drastically reduce overall lead times. This flexibility means that inventory can be held at the intermediate stage, allowing for rapid response to fluctuating market demand without the need to restart the entire synthesis from scratch. The ability to produce multiple analogues from a common protected intermediate also maximizes asset utilization, as the same production lines can be adapted for different cephalosporin variants with minimal changeover downtime.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions, relying instead on classical organic transformations that utilize cost-effective reagents like phosphorus oxychloride and carbodiimides. This shift significantly lowers the direct material cost per kilogram of the final intermediate. Additionally, the use of standard solvents such as dichloromethane, ethyl acetate, and acetonitrile simplifies solvent recovery and recycling operations, further driving down operational expenditures. The high selectivity of the reactions minimizes the formation of difficult-to-remove impurities, reducing the burden on downstream purification units and lowering the consumption of chromatography media. By optimizing the stoichiometry and reaction conditions, manufacturers can achieve superior atom economy, ensuring that a greater proportion of raw materials end up in the final product rather than as waste. These cumulative efficiencies translate into a leaner cost structure, allowing pharmaceutical companies to maintain healthy margins even in competitive generic markets.
- Enhanced Supply Chain Reliability: The synthetic route is designed with supply chain resilience in mind, avoiding single-source dependencies for critical reagents. The protecting groups used, such as p-methoxybenzyl and benzhydryl, are derived from widely available petrochemical feedstocks, insulating the production process from volatility in specialty chemical markets. Moreover, the reaction conditions are relatively mild, typically ranging from ambient temperature to moderate heating, which reduces the energy load on manufacturing facilities and minimizes the risk of thermal runaways or equipment failures. This operational stability ensures consistent output rates, allowing supply chain planners to forecast production volumes with greater accuracy. The robustness of the chemistry also means that the process is less sensitive to minor variations in raw material quality, reducing the frequency of batch rejections and the need for costly rework. Consequently, partners can rely on a steady flow of high-quality intermediates to support their finished drug product launches and lifecycle management strategies.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is facilitated by the use of unit operations that are standard in the fine chemical industry, such as filtration, crystallization, and liquid-liquid extraction. The absence of hazardous reagents like azides or highly toxic heavy metals simplifies the environmental health and safety (EHS) profile of the manufacturing site. Waste streams generated during the process are primarily organic solvents and aqueous salt solutions, which can be treated using conventional wastewater treatment protocols or incineration with energy recovery. The implementation of resin-based purification instead of extensive silica gel chromatography reduces the volume of solid waste generated, aligning with modern green chemistry principles. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturer, meeting the increasingly stringent ESG criteria demanded by global pharmaceutical buyers. The process is inherently scalable, allowing for seamless capacity expansion to meet growing global demand for advanced antibiotics without requiring fundamental changes to the reaction engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel cephalosporin intermediates. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary antibacterial advantage of this cephalosporin structure?
A: The inclusion of the 1,5-dihydroxy-4-pyridone-2-alkoxyimino group at the 7-position significantly enhances activity against Gram-negative bacteria, including Pseudomonas aeruginosa, while maintaining low toxicity profiles.
Q: How does this process improve supply chain stability for antibiotic production?
A: By utilizing readily available starting materials like kojic acid and standard protecting group chemistry, the process reduces reliance on exotic reagents, ensuring consistent raw material availability and reducing procurement risks.
Q: Is the synthesis scalable for commercial API manufacturing?
A: Yes, the reaction conditions described, such as acylation and nucleophilic substitution, operate under moderate temperatures and use common solvents, making the pathway highly amenable to scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalosporin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a dependable supply of high-quality pharmaceutical intermediates to support the development of life-saving antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical instrumentation to verify identity and assay. Our facility is equipped to handle the specific solvent systems and reaction conditions required for beta-lactam chemistry, guaranteeing that the structural integrity of the sensitive cephalosporin nucleus is preserved throughout the manufacturing process. By partnering with us, you gain access to a supply chain that prioritizes consistency, compliance, and continuous improvement, mitigating the risks associated with outsourcing complex chemical synthesis.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs with a Customized Cost-Saving Analysis. Whether you require process optimization, alternative route scouting, or immediate supply of GMP-grade intermediates, we are prepared to provide comprehensive solutions tailored to your timeline and budget. Please contact us to request specific COA data and route feasibility assessments for the cephalosporin derivatives discussed in patent CN1022629C. Our goal is to become your strategic partner in bringing advanced antibacterial therapies to market efficiently and reliably, leveraging our deep technical expertise to overcome any manufacturing challenges you may face.
