Advanced Synthesis of 7β-Amino-3-Acephalosporin-4-Carboxylic Acid Intermediates for Commercial Scale-Up
Advanced Synthesis of 7β-Amino-3-Acephalosporin-4-Carboxylic Acid Intermediates for Commercial Scale-Up
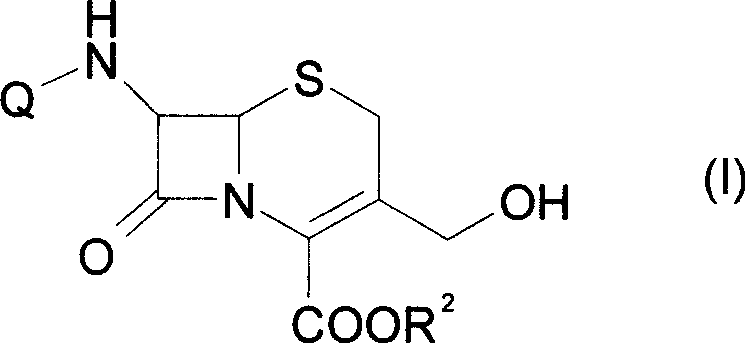
The global pharmaceutical landscape is continuously driven by the demand for high-quality beta-lactam antibiotics, particularly third and fourth-generation cephalosporins which rely heavily on robust intermediate supply chains. Patent CN101463039A introduces a transformative methodology for the preparation of 7β-(alkanoylamino)-3-hydroxymethylcephalosporin-4-carboxylic acid, a critical precursor in the synthesis of 7β-amino-3-acephalosporin-4-carboxylic acid (7-ANCA). This intermediate serves as the foundational scaffold for manufacturing vital antibiotics such as Cefotetan, Cefadroxil, and Ceftizoxime. The disclosed technology represents a significant leap forward in process chemistry, moving away from unstable enzymatic or harsh alkaline hydrolysis towards a controlled, mild organic base-mediated deacetylation. By leveraging tetrabutylammonium hydroxide (TBAH) and triethylamine (TEA), the process achieves exceptional stability of the sensitive beta-lactam ring, addressing a long-standing pain point in cephalosporin manufacturing. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent offers a blueprint for cost-effective, high-purity production that aligns with modern GMP standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of 7-aminocephalosporanic acid (7-ACA) to 7-amino-3-hydroxymethylcephalosporanic acid (7-AHCA) has been fraught with technical challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional approaches often relied on biological enzymes such as Rhodotorula rubra or harsh inorganic bases like sodium hydroxide and potassium hydroxide. These conventional methods suffer from severe operational drawbacks, primarily the extreme sensitivity of the 7-AHCA molecule to pH and temperature fluctuations. Specifically, the beta-lactam ring is highly prone to opening under alkaline conditions, leading to significant degradation and the formation of unwanted polymeric byproducts. Furthermore, to mitigate this instability, prior art processes frequently necessitated cryogenic reaction conditions, often requiring temperatures below -40°C. Such extreme thermal requirements impose a massive burden on energy consumption and require specialized, expensive refrigeration infrastructure, drastically inflating the cost of goods sold (COGS) and complicating the supply chain logistics for high-purity API intermediates.
The Novel Approach
In stark contrast, the novel process detailed in CN101463039A utilizes a sophisticated combination of organic phase transfer catalysis and mild buffering to overcome these thermodynamic barriers. By employing a mixture of 40% TBAH and triethylamine in an alcohol-water solvent system, the invention creates a reaction environment that is sufficiently basic to effect deacetylation yet buffered enough to preserve the integrity of the beta-lactam nucleus. This approach allows the reaction to proceed efficiently within a much broader and more manageable temperature range of -20°C to 40°C. This shift from cryogenic to moderate temperatures is not merely a convenience; it is a fundamental economic driver that enables cost reduction in API manufacturing by eliminating the need for deep-freeze reactors. Additionally, the subsequent steps involving acylation with acid chlorides and protection with diphenyldiazomethane are streamlined to occur at ambient or slightly elevated temperatures (0-60°C), further simplifying the operational workflow and enhancing the overall yield reliability compared to the erratic outputs of enzymatic hydrolysis.
Mechanistic Insights into TBAH-Mediated Deacetylation and Oxidation
The core innovation of this synthesis lies in the precise mechanistic control exerted during the initial deacetylation step. The use of TBAH acts as a phase transfer catalyst that facilitates the nucleophilic attack on the acetyl group at the C-3 position of the cephem ring, while the concurrent presence of triethylamine serves to neutralize acidic byproducts and maintain a stable pH microenvironment. This dual-base system prevents the localized high pH spikes that typically trigger beta-lactam ring scission. Following the formation of the 7β-amino-3-hydroxymethyl intermediate, the process employs a strategic protection-deprotection sequence. The carboxylic acid at the C-4 position is esterified using diphenyldiazomethane to form the benzhydryl ester, which provides steric bulk and electronic stabilization during subsequent oxidative transformations. The oxidation of the C-3 hydroxymethyl group to the corresponding aldehyde is achieved using activated manganese dioxide (MnO2) in dichloromethane. This specific oxidant is chosen for its chemoselectivity, ensuring that the sensitive double bond in the dihydrothiazine ring and the amide side chain remain untouched, thereby preserving the stereochemical integrity required for biological activity in the final antibiotic product.
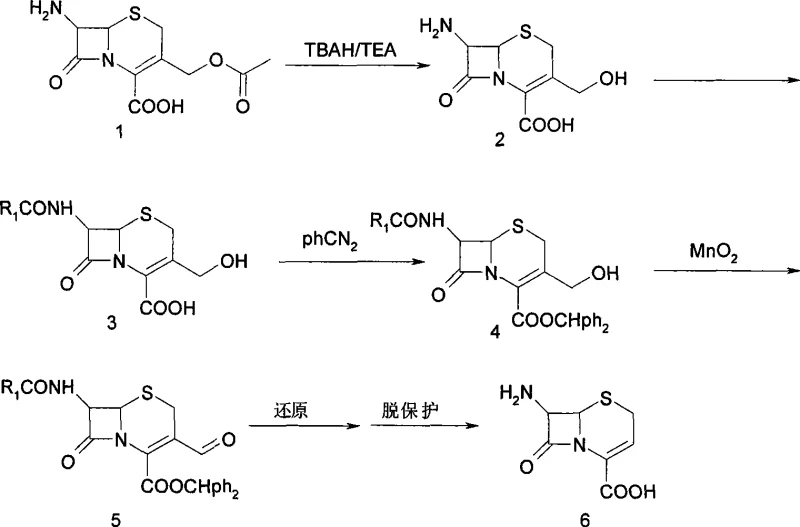
Furthermore, the impurity profile is rigorously controlled through the crystallization and washing steps described in the embodiments. For instance, the isolation of the intermediate 7β-phenylacetamide-3-hydroxymethylcephalosporin-4-carboxylic acid involves precipitation at pH 3-4 followed by washing with cold methanol-water mixtures. This specific pH control ensures that zwitterionic impurities are removed while the desired product precipitates in high purity. The final conversion to 7-ANCA involves a decarbonylation and deprotection sequence that removes the benzhydryl group and the formyl moiety. The mechanistic elegance of this route ensures that the final product, 7β-amino-3-acephalosporin-4-carboxylic acid, is obtained with minimal racemization and high structural fidelity, which is critical for downstream coupling reactions in the synthesis of semi-synthetic cephalosporins.
How to Synthesize 7-ANCA Efficiently
The synthesis of 7β-amino-3-acephalosporin-4-carboxylic acid (7-ANCA) via this patented route involves a sequential four-step transformation starting from commercially available 7-ACA. The process begins with the mild deacetylation to generate the hydroxymethyl intermediate, followed by N-acylation to introduce the desired side chain functionality. Subsequent protection of the carboxylic acid as a benzhydryl ester allows for the selective oxidation of the C-3 alcohol to an aldehyde using manganese dioxide. The final stage involves catalytic hydrogenation and acid-mediated deprotection to yield the target acephalosporin nucleus. This standardized protocol is designed for reproducibility and safety, avoiding hazardous reagents where possible. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the comprehensive technical guide below.
- Deacetylation of 7-ACA using TBAH and TEA at -20 to 40°C to form 7β-amino-3-hydroxymethylcephalosporanic acid.
- Acylation with acid chloride followed by esterification with diphenyldiazomethane to protect the carboxylic acid group.
- Oxidation of the hydroxymethyl group to an aldehyde using active MnO2, followed by decarbonylation and deprotection to yield the final 7-ANCA product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the reaction conditions, which directly translates to enhanced supply chain reliability and reduced operational risk. By eliminating the dependency on expensive and potentially variable enzymatic catalysts, manufacturers can secure a more consistent supply of raw materials, mitigating the risks associated with biological sourcing variability. Furthermore, the ability to run reactions at near-ambient temperatures significantly lowers the energy footprint of the manufacturing process, contributing to both cost efficiency and environmental sustainability goals. The robustness of the chemical reagents used, such as TBAH and MnO2, ensures that the supply chain is less vulnerable to disruptions compared to processes relying on specialized biocatalysts or cryogenic infrastructure.
- Cost Reduction in Manufacturing: The elimination of deep-freeze requirements (below -40°C) represents a massive saving in capital expenditure (CAPEX) and operational expenditure (OPEX). Traditional methods require specialized low-temperature reactors and significant energy input for cooling, whereas this novel process operates efficiently between -20°C and 40°C. Additionally, the avoidance of costly cephalosporin enzymes removes a major variable cost component. The use of standard organic solvents and inorganic oxidants like manganese dioxide further drives down the cost of goods, making the final intermediate more price-competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as triethylamine, tetrabutylammonium hydroxide, and diphenyldiazomethane ensures a stable and diversified supply base. Unlike enzymatic processes that may suffer from batch-to-batch potency variations or shelf-life limitations, these chemical reagents offer long-term stability and consistent performance. This consistency is crucial for maintaining uninterrupted production schedules and meeting tight delivery windows for downstream API manufacturers. The simplified workup procedures, involving standard filtration and crystallization, also reduce the turnaround time between batches, allowing for faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale industrial production, as evidenced by the straightforward isolation techniques like precipitation and filtration. The mild reaction conditions reduce the formation of hazardous byproducts and simplify waste stream management. The use of manganese dioxide, while requiring proper disposal, is a well-understood reagent in industrial chemistry with established recycling or neutralization protocols. This alignment with green chemistry principles facilitates easier regulatory approval and environmental compliance, ensuring long-term viability of the manufacturing site and reducing the risk of production halts due to environmental audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 7-ANCA intermediates. These answers are derived directly from the experimental data and claims presented in patent CN101463039A, providing clarity on the process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Why is the TBAH/TEA method superior to traditional alkaline hydrolysis for 7-ACA?
A: Traditional alkaline hydrolysis often requires extremely low temperatures (below -40°C) to prevent β-lactam ring opening, whereas the TBAH/TEA method operates effectively between -20°C and 40°C, significantly reducing energy costs and equipment complexity while maintaining high product stability.
Q: What are the key impurities controlled in this synthesis route?
A: The process specifically addresses the instability of the β-lactam ring under basic conditions. By using mild organic bases like triethylamine alongside TBAH, the method minimizes ring-opening byproducts, ensuring a cleaner impurity profile suitable for downstream antibiotic synthesis.
Q: Is this process scalable for industrial production of cephalosporin intermediates?
A: Yes, the patent explicitly highlights the route's suitability for large-scale industrial production due to its mild reaction conditions, avoidance of expensive enzymatic catalysts, and the use of readily available reagents like manganese dioxide and diphenyldiazomethane.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-ANCA Intermediate Supplier
The technological advancements outlined in this patent underscore the critical importance of having a manufacturing partner with deep expertise in beta-lactam chemistry. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of cephalosporin synthesis, including the precise temperature control and containment necessary for handling potent intermediates. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of 7-ANCA or related cephem nuclei meets the highest international pharmacopoeial standards, guaranteeing the safety and efficacy of the final antibiotic formulations.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
