Advanced Green Synthesis of Olanzapine Intermediates: Enhancing Purity and Scalability for Global API Manufacturing
Advanced Green Synthesis of Olanzapine Intermediates: Enhancing Purity and Scalability for Global API Manufacturing
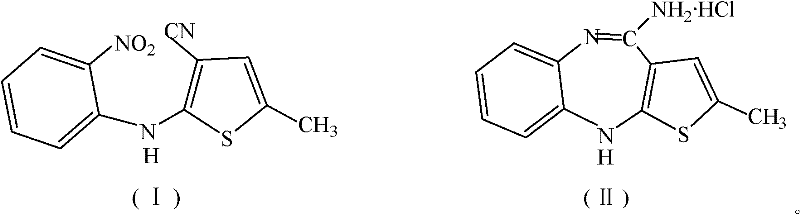
The pharmaceutical industry is constantly seeking robust, environmentally sustainable pathways for the production of critical antipsychotic agents. A pivotal advancement in this domain is detailed in patent CN102199162A, which discloses a highly efficient preparation method for the key olanzapine intermediate, 4-amino-2-methyl-10H-thiophene[2,3-b][1,5]-benzodiazepine hydrochloride. This technology represents a significant leap forward in process chemistry, addressing long-standing challenges related to toxic reagent consumption and waste management. By optimizing the reductive condensation of 2-(2-nitroanilino)-5-methyl thiophene-3-nitrile, this method ensures that manufacturers can achieve superior yields while adhering to stricter environmental regulations. For R&D directors and procurement specialists alike, understanding the nuances of this patented route is essential for securing a reliable supply chain for high-value psychiatric medications.
The core innovation lies in the strategic modification of the reduction system. Traditionally, the synthesis of this benzodiazepine scaffold relied heavily on excessive amounts of stannous chloride, a reagent known for its toxicity and the difficulty associated with removing tin residues from the final product. The methodology outlined in CN102199162A introduces sodium hypophosphite monohydrate as a clean, effective co-reducing agent. This adjustment not only maintains the high reaction efficiency required for industrial throughput but also fundamentally alters the economic and ecological footprint of the manufacturing process. As we delve deeper into the technical specifics, it becomes clear why this approach is becoming the preferred standard for modern pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those referenced in earlier patents like CN1028429C, typically employed anhydrous stannous chloride as the sole or primary reducing agent in hydrochloric acid solutions. While chemically effective, this approach suffered from severe drawbacks regarding reagent stoichiometry and environmental safety. Specifically, the conventional process required a molar ratio of stannous chloride to the nitro-substituted precursor of approximately 3.36:1. This massive excess was necessary to drive the reduction to completion but resulted in the generation of substantial quantities of tin-containing waste liquids. For a reliable pharmaceutical intermediate supplier, managing this hazardous waste stream adds significant operational costs and complexity. Furthermore, the presence of high concentrations of tin salts complicates the downstream purification process, often necessitating additional treatment steps to ensure the final API meets rigorous heavy metal limits.
The Novel Approach
In stark contrast, the novel approach described in the patent leverages a synergistic reduction system combining hydrochloric acid, stannous chloride, and sodium hypophosphite in an ethanol medium. This tripartite system allows for a dramatic reduction in the reliance on toxic tin reagents. The molar ratio of the precursor to stannous chloride is optimized to a mere 1:0.2 to 1:0.5, representing a more than tenfold decrease in tin usage compared to traditional methods. The reaction proceeds smoothly at temperatures between 85°C and 95°C over a period of 5 to 10 hours. By shifting the reducing burden to the cleaner sodium hypophosphite, the process minimizes the formation of inorganic sludge and simplifies the isolation of the product. This shift is not merely a chemical curiosity; it is a strategic enabler for cost reduction in API manufacturing, as it lowers raw material costs and waste disposal fees simultaneously.
Mechanistic Insights into Reductive Cyclization
The transformation of 2-(2-nitroanilino)-5-methyl thiophene-3-nitrile into the tricyclic benzodiazepine structure involves a complex sequence of reduction and cyclization events. Initially, the nitro group on the aniline moiety undergoes reduction to an amino group. In the presence of strong acid (6-12 mol/L HCl), this newly formed amine becomes nucleophilic enough to attack the nitrile carbon on the adjacent thiophene ring. This intramolecular cyclization forms the seven-membered diazepine ring characteristic of the olanzapine scaffold. The role of stannous chloride here is catalytic rather than stoichiometric; it likely facilitates the initial electron transfer to the nitro group or activates the nitrile towards nucleophilic attack, while sodium hypophosphite serves as the terminal reductant to regenerate the active catalytic species or directly reduce intermediate nitroso/hydroxylamine species. This mechanistic efficiency ensures that the reaction proceeds with high atom economy.
From an impurity control perspective, the use of ethanol as the solvent plays a crucial role. Ethanol acts not only as a solubilizing agent for the organic starting materials but also helps in moderating the reaction kinetics, preventing runaway exotherms that could lead to decomposition byproducts. The specific choice of sodium hypophosphite monohydrate ensures a steady release of reducing equivalents without generating gaseous byproducts that could disrupt the reaction homogeneity. Consequently, the crude product obtained after filtration exhibits exceptional purity, often exceeding 99.85% by HPLC analysis without the need for extensive chromatographic purification. This high level of chemical fidelity is paramount for R&D teams focused on minimizing genotoxic impurities and ensuring batch-to-batch consistency in the final drug substance.
How to Synthesize 4-Amino-2-methyl-10H-thiophene[2,3-b][1,5]-benzodiazepine Hydrochloride Efficiently
Implementing this synthesis requires precise control over reagent addition and thermal parameters to maximize yield and safety. The process begins with the preparation of the reaction medium, where ethanol, concentrated hydrochloric acid, sodium hypophosphite, and the reduced load of stannous chloride are combined. The substrate is then introduced, and the mixture is heated to reflux conditions specifically tailored between 85°C and 95°C. Following the reaction period, the workup involves the removal of the ethanol solvent, followed by controlled cooling to induce crystallization of the hydrochloride salt. The solid is then isolated via filtration and washed with ice-cold water to remove residual acids and inorganic salts.
- Prepare the reaction medium by mixing ethanol, concentrated hydrochloric acid, sodium hypophosphite monohydrate, and a catalytic amount of stannous chloride dihydrate.
- Add 2-(2-nitroanilino)-5-methyl thiophene-3-nitrile to the mixture and heat to 85-95°C for 5-10 hours to facilitate reductive cyclization.
- Remove ethanol via distillation, cool the solution to 10°C, filter the precipitate, wash with ice water to pH 1-2, and dry to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By reducing the dependency on stannous chloride, a reagent subject to fluctuating market prices and strict handling regulations, manufacturers can stabilize their input costs. Furthermore, the reduction in hazardous waste generation translates directly into lower environmental compliance costs. Disposing of tin-heavy waste streams is expensive and logistically challenging; minimizing this output streamlines the entire production lifecycle. This efficiency makes the process highly attractive for commercial scale-up of complex pharmaceutical intermediates, ensuring that production timelines are not delayed by waste treatment bottlenecks.
- Cost Reduction in Manufacturing: The most significant economic driver is the reduction in reagent consumption. By lowering the stannous chloride ratio from 3.36 equivalents to just 0.2 equivalents, the direct material cost for the reduction step is significantly lowered. Additionally, the use of sodium hypophosphite, a relatively inexpensive and stable solid, replaces a portion of the more costly and hazardous tin reagent. This substitution eliminates the need for expensive heavy metal scavengers or complex extraction protocols often required to meet residual tin specifications in the final API. Consequently, the overall cost of goods sold (COGS) for the intermediate is optimized, providing a competitive edge in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply security. The reagents involved—ethanol, hydrochloric acid, and sodium hypophosphite—are commodity chemicals available from multiple global sources, reducing the risk of supply disruption associated with specialized or single-source catalysts. The simplified workup procedure, which relies on straightforward filtration and washing rather than complex distillation or chromatography, reduces the equipment footprint and maintenance requirements. This operational simplicity ensures that production facilities can maintain high uptime and consistent output, crucial for reducing lead time for high-purity pharmaceutical intermediates in a just-in-time manufacturing environment.
- Scalability and Environmental Compliance: As regulatory pressures regarding green chemistry intensify, this process positions manufacturers favorably. The significant decrease in heavy metal usage aligns with global initiatives to reduce toxic emissions and waste. The process generates less acidic and metallic wastewater, easing the burden on effluent treatment plants. This environmental compatibility facilitates easier permitting for new production lines and expansion of existing facilities. Moreover, the high purity of the crude product means fewer recrystallization cycles are needed, saving energy and solvents. This scalability ensures that the transition from pilot plant to multi-ton commercial production is seamless and compliant with international environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers are derived directly from the experimental data and beneficial effects reported in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: How does the new method reduce environmental impact compared to traditional synthesis?
A: The novel process utilizes sodium hypophosphite as a primary reducing agent, drastically lowering the molar ratio of toxic stannous chloride from 1:3.36 to 1:0.2, thereby minimizing heavy metal waste generation.
Q: What are the purity specifications achievable with this reductive cyclization method?
A: Experimental data indicates that the process consistently yields products with HPLC purity exceeding 99.85%, meeting stringent requirements for downstream pharmaceutical applications.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the use of common solvents like ethanol and the simplified workup procedure involving filtration and washing make the process highly scalable and operationally safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-2-methyl-10H-thiophene[2,3-b][1,5]-benzodiazepine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of the pharmaceutical industry. Our team of expert chemists has extensively analyzed the methodology described in CN102199162A and possesses the technical capability to execute this process with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate we deliver meets the highest global standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced technology for your olanzapine production needs. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and achieve your commercial goals with confidence and reliability.
