Advanced Synthesis of Protected Olanzapine Intermediates for Commercial Scale Production
Advanced Synthesis of Protected Olanzapine Intermediates for Commercial Scale Production
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for high-volume antipsychotic agents like Olanzapine. Patent CN100383144C introduces a transformative approach to synthesizing key intermediates, specifically 2-methyl-4-[4-formate-piperazinyl]-10H-thieno[2,3-b][1,5]benzodiazepine derivatives. This technology addresses critical bottlenecks in the traditional manufacturing of Olanzapine by replacing hazardous methylation steps with a sophisticated N-protection strategy using diester dicarbonates. For R&D directors and procurement specialists, this represents a shift towards greener chemistry that significantly mitigates the risks associated with toxic alkylating agents while enhancing the overall yield profile. The method leverages mild reaction conditions, typically ranging from -10°C to 60°C, which contrasts sharply with the harsh thermal requirements of legacy processes. By integrating this intermediate into the supply chain, manufacturers can achieve a more consistent quality profile, ensuring that the final active pharmaceutical ingredient meets stringent regulatory standards without the need for excessive recrystallization cycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Olanzapine has relied heavily on direct methylation strategies that pose substantial safety and efficiency challenges. As illustrated in prior art such as European Patent EP0454436 and US20060035887, traditional routes often employ virulent methylating agents like methyl iodide or dimethyl sulfate. 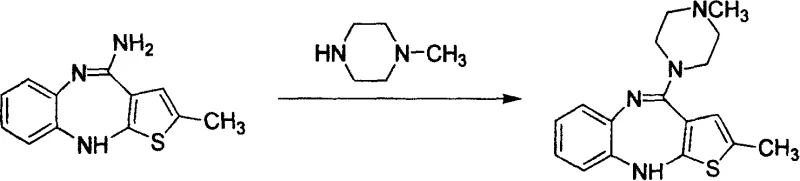 These reagents are not only highly toxic and carcinogenic but also generate significant amounts of hazardous waste, complicating environmental compliance and disposal protocols. Furthermore, comparative data indicates that these direct methylation pathways frequently suffer from suboptimal yields, often hovering around 51%, necessitating extensive downstream purification to remove unreacted starting materials and side products. The reliance on high-boiling solvents such as DMSO or DMF in some variations further exacerbates the issue, as removing these solvents requires energy-intensive distillation processes that increase the carbon footprint and operational costs of the manufacturing facility. Consequently, the impurity profile of the crude product is often poor, leading to batch-to-batch variability that is unacceptable for modern GMP production environments.
These reagents are not only highly toxic and carcinogenic but also generate significant amounts of hazardous waste, complicating environmental compliance and disposal protocols. Furthermore, comparative data indicates that these direct methylation pathways frequently suffer from suboptimal yields, often hovering around 51%, necessitating extensive downstream purification to remove unreacted starting materials and side products. The reliance on high-boiling solvents such as DMSO or DMF in some variations further exacerbates the issue, as removing these solvents requires energy-intensive distillation processes that increase the carbon footprint and operational costs of the manufacturing facility. Consequently, the impurity profile of the crude product is often poor, leading to batch-to-batch variability that is unacceptable for modern GMP production environments.
The Novel Approach
In stark contrast, the methodology disclosed in CN100383144C utilizes a protective group strategy that fundamentally alters the reaction landscape. Instead of forcing a methyl group onto the nitrogen atom directly, the process first converts N-demethylolanzapine into a stable carbamate intermediate using diester dicarbonates, such as di-tert-butyl dicarbonate (Boc2O) or dibenzyl dicarbonate. 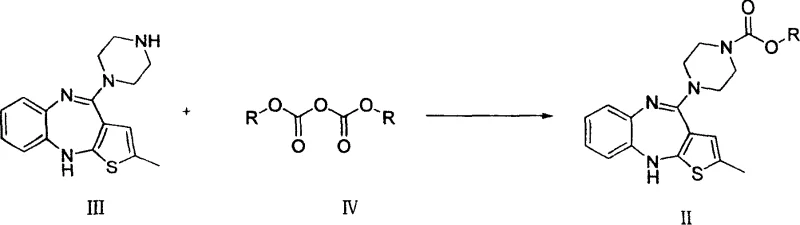 This transformation proceeds under remarkably mild conditions, often initiated at temperatures as low as -10°C and maintained between 10°C to 60°C, which drastically reduces the thermal load on the reactor system. The use of common organic solvents like tetrahydrofuran (THF), dichloromethane, or toluene simplifies the workup procedure, allowing for efficient extraction and solvent recovery. Experimental examples within the patent demonstrate that this route consistently achieves yields exceeding 70%, with HPLC purity levels surpassing 99.5%. This high level of purity at the intermediate stage significantly reduces the burden on the final purification steps, streamlining the entire production workflow. By avoiding toxic methylating agents, the process also eliminates the need for specialized containment equipment required for handling carcinogens, thereby lowering capital expenditure and improving worker safety.
This transformation proceeds under remarkably mild conditions, often initiated at temperatures as low as -10°C and maintained between 10°C to 60°C, which drastically reduces the thermal load on the reactor system. The use of common organic solvents like tetrahydrofuran (THF), dichloromethane, or toluene simplifies the workup procedure, allowing for efficient extraction and solvent recovery. Experimental examples within the patent demonstrate that this route consistently achieves yields exceeding 70%, with HPLC purity levels surpassing 99.5%. This high level of purity at the intermediate stage significantly reduces the burden on the final purification steps, streamlining the entire production workflow. By avoiding toxic methylating agents, the process also eliminates the need for specialized containment equipment required for handling carcinogens, thereby lowering capital expenditure and improving worker safety.
Mechanistic Insights into N-Protection and Reduction Strategy
The core chemical innovation lies in the nucleophilic attack of the secondary amine of N-demethylolanzapine on the electrophilic carbonyl carbon of the diester dicarbonate. In the presence of a base, such as sodium hydroxide, potassium carbonate, or triethylamine, the nitrogen lone pair becomes sufficiently nucleophilic to displace the leaving group, forming a stable urethane (carbamate) linkage. This protection step is highly selective and prevents unwanted side reactions that typically plague direct alkylation methods. The resulting intermediate, characterized by the formula (II) where R is a tert-butyl or benzyl group, serves as a robust precursor that can be stored or transported with greater stability than the free amine. The subsequent conversion to the final API involves a reduction step where the carbamate protecting group is cleaved. 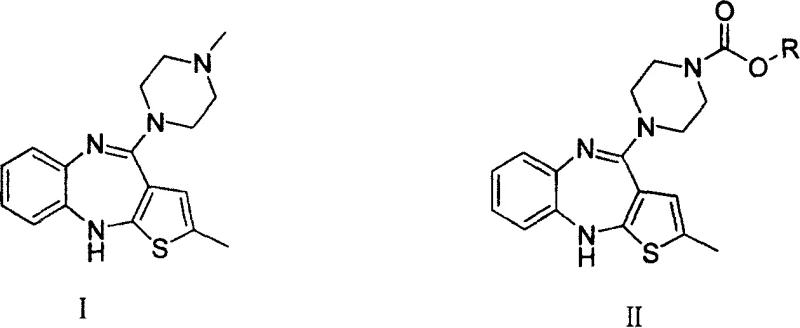 This is achieved using powerful reducing agents like lithium aluminum hydride (LiAlH4) or borane complexes. The mechanism involves the hydride attacking the carbonyl of the carbamate, eventually leading to the expulsion of the protecting group as an alcohol and the regeneration of the N-methyl functionality (after subsequent methylation logic or if the intermediate was designed to mimic the final state, though here it acts as a purified precursor for final assembly). The patent specifically notes that this reduction can be conducted at temperatures ranging from -10°C to 100°C, providing flexibility in process control. The ability to tune the reaction temperature allows chemists to optimize the rate of reduction while minimizing the formation of degradation byproducts, ensuring a clean impurity profile.
This is achieved using powerful reducing agents like lithium aluminum hydride (LiAlH4) or borane complexes. The mechanism involves the hydride attacking the carbonyl of the carbamate, eventually leading to the expulsion of the protecting group as an alcohol and the regeneration of the N-methyl functionality (after subsequent methylation logic or if the intermediate was designed to mimic the final state, though here it acts as a purified precursor for final assembly). The patent specifically notes that this reduction can be conducted at temperatures ranging from -10°C to 100°C, providing flexibility in process control. The ability to tune the reaction temperature allows chemists to optimize the rate of reduction while minimizing the formation of degradation byproducts, ensuring a clean impurity profile.
Controlling impurities is paramount in the synthesis of psychotropic medications, and this pathway offers distinct advantages in that regard. Traditional methods often generate quaternary ammonium salts or over-alkylated byproducts that are structurally similar to the target molecule and difficult to separate. The protection-deprotection sequence effectively masks the reactive amine, preventing these specific side reactions during the intermediate handling phases. Furthermore, the crystallization properties of the protected intermediate (Formula II) are superior to the free base, allowing for a purification step via recrystallization from ethanol or other alcohols before the final reduction. This "purify-then-reduce" strategy ensures that any impurities carried forward are minimized, resulting in a final Olanzapine product that meets rigorous pharmacopeial standards with minimal additional processing. The use of inorganic bases like sodium bicarbonate or potassium carbonate also helps in maintaining a neutral to slightly alkaline pH, preventing acid-catalyzed degradation of the sensitive thienobenzodiazepine ring system during the reaction.
How to Synthesize 2-Methyl-4-[4-formate-piperazinyl]-10H-thieno[2,3-b][1,5]benzodiazepine Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits of the novel chemistry. The process begins with the dissolution of N-demethylolanzapine in a suitable solvent system, followed by the controlled addition of the dicarbonate reagent. The reaction mixture is then allowed to stir for a defined period, typically between 0.5 to 3 hours, depending on the specific scale and reagents used. Following the isolation of the intermediate, the reduction step is performed under an inert atmosphere to prevent moisture sensitivity issues associated with hydride reagents. This structured approach ensures reproducibility and safety, making it an ideal candidate for technology transfer from the laboratory to pilot and commercial scales.
- Dissolve N-demethylolanzapine in an organic solvent such as THF or dichloromethane, cool to -10°C to 0°C, and add di-tert-butyl dicarbonate with a base like sodium hydroxide to form the protected intermediate.
- Maintain the reaction at room temperature or slightly elevated temperatures (10-60°C) for 0.5 to 3 hours to ensure complete conversion to the carbamate intermediate.
- Reduce the isolated intermediate using lithium aluminum hydride or borane in THF at controlled temperatures (-10°C to room temperature) to yield pure Olanzapine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented intermediate synthesis offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of hazardous methylating agents like methyl iodide removes a significant regulatory burden and reduces the costs associated with hazardous waste disposal and worker safety monitoring. Cost Reduction in Manufacturing: By utilizing readily available reagents such as di-tert-butyl dicarbonate and common solvents like THF and toluene, the raw material costs are stabilized and often lower than those of specialized alkylating agents. The higher yields observed in the examples (70% vs 51% in comparative examples) directly translate to a lower cost of goods sold (COGS), as less starting material is required to produce the same amount of final API. Additionally, the simplified purification process reduces the consumption of auxiliary materials and energy, further driving down operational expenses. Enhanced Supply Chain Reliability: The reagents required for this process are commodity chemicals with robust global supply chains, reducing the risk of shortages that can occur with niche or highly regulated toxic substances. The milder reaction conditions also mean that the process can be run in a wider variety of standard chemical reactors without the need for exotic metallurgy or extreme pressure ratings, increasing the number of potential contract manufacturing organizations (CMOs) capable of producing the material. Scalability and Environmental Compliance: The process generates fewer toxic byproducts and avoids the use of heavy metals or persistent organic pollutants, aligning with modern green chemistry principles. This facilitates smoother environmental permitting and reduces the long-term liability associated with chemical manufacturing. The ability to recrystallize the intermediate ensures a high-quality feedstock for the final step, reducing the risk of batch failures and ensuring a continuous, reliable supply of high-purity Olanzapine for the market.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common concerns regarding safety, scalability, and compatibility with existing infrastructure. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: Why is the diester dicarbonate method superior to direct methylation for Olanzapine production?
A: Direct methylation often utilizes toxic agents like methyl iodide, resulting in lower yields (around 51%) and significant impurity profiles requiring complex purification. The diester dicarbonate route operates under milder conditions, avoids virulent reagents, and achieves HPLC purity greater than 99.5%.
Q: What reducing agents are compatible with this intermediate for the final step?
A: The patent specifies that strong reducing agents such as lithium aluminum hydride (LiAlH4), lithium borohydride, or borane gas are effective. These agents successfully cleave the formate/carbamate protecting group to regenerate the secondary amine without degrading the thienobenzodiazepine core.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial viability. It utilizes common solvents like THF, toluene, and dichloromethane, and avoids high-boiling solvents that are difficult to remove. The reaction temperatures are moderate (-10°C to 60°C), facilitating safer scale-up compared to prior art methods requiring over 100°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olanzapine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN100383144C and is fully prepared to execute this protected intermediate strategy at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. Our facilities are equipped with state-of-the-art reactors capable of handling the mild thermal profiles required for this synthesis, and our stringent purity specifications guarantee that every batch of intermediate meets the highest quality standards. With our rigorous QC labs, we perform comprehensive testing at every stage of the process to verify identity, potency, and impurity levels, providing our partners with the confidence they need to move forward.
We invite you to collaborate with us to leverage this innovative chemistry for your Olanzapine supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your production costs and secure a sustainable supply of high-quality pharmaceutical intermediates.
