Advanced Manufacturing of L-Alanyl-L-Glutamine: A Technical Breakdown for Global Supply Chains
Introduction to Next-Generation Dipeptide Manufacturing
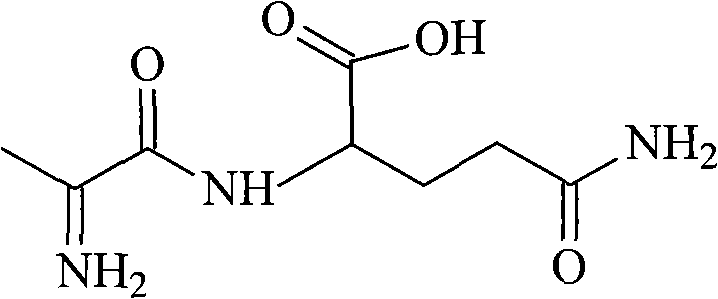
The global demand for stable, highly soluble amino acid dipeptides for parenteral nutrition has driven intense innovation in synthetic methodologies, specifically targeting the production of L-alanyl-L-glutamine (CAS: 393570-23-0). As detailed in patent CN101519428B, a novel synthetic route has been established that fundamentally alters the economic and safety profile of manufacturing this critical pharmaceutical intermediate. Unlike traditional approaches that rely on hazardous gases or expensive precious metal catalysts, this technology utilizes a mild mixed anhydride activation strategy under strictly controlled low-temperature conditions. This report provides a deep technical analysis of this process, highlighting its suitability for large-scale commercial production and its alignment with modern green chemistry principles. For procurement leaders and R&D directors, understanding the mechanistic advantages of this route is essential for securing a reliable amino acid dipeptide supplier capable of delivering consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-alanyl-L-glutamine has been plagued by significant technical and economic bottlenecks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Early methodologies often employed Z-protected alanine coupled with glutamine using dicyclohexylcarbodiimide (DCC), a process that generates dicyclohexylurea (DCU) as a stubborn byproduct that is notoriously difficult to remove, complicating purification and lowering overall yields. Alternative routes utilizing phosgene gas to generate mixed anhydrides pose severe occupational health risks due to the extreme toxicity of the reagent, requiring specialized containment infrastructure that drives up capital expenditure. Furthermore, methods relying on triphenylphosphine and hexachloroethane introduce heavy halogenated waste streams and utilize expensive N-protected starting materials, rendering the final product cost-prohibitive for high-volume applications in the body-care and clinical nutrition sectors.
The Novel Approach
The innovative process described in the patent data overcomes these legacy challenges by introducing a streamlined two-step sequence centered on N-formyl-L-alanine activation. By selecting the formyl group as the N-terminal protecting group, the synthesis bypasses the need for hydrogenation steps entirely, eliminating the requirement for expensive palladium on carbon catalysts and high-pressure reactor vessels. The core of this novelty lies in the generation of a mixed anhydride intermediate using isobutyl chloroformate and N-methylmorpholine in tetrahydrofuran at cryogenic temperatures. This approach not only ensures exceptional stereochemical control to prevent racemization but also utilizes reagents that are commodity chemicals with robust supply chains. The subsequent deprotection is achieved using mild hydrochloric acid hydrolysis, a simple and scalable unit operation that facilitates the isolation of the final dipeptide with minimal environmental impact.
Mechanistic Insights into Mixed Anhydride Activation and Coupling
The success of this synthetic route hinges on the precise kinetic control of the mixed anhydride formation and the subsequent nucleophilic attack by the amino group of glutamine. At temperatures maintained between -18°C and -15°C, the reaction between N-formyl-L-alanine and isobutyl chloroformate generates a highly reactive acylating species without inducing significant epimerization at the chiral alpha-carbon. The addition of N-methylmorpholine serves as a proton scavenger, neutralizing the hydrochloric acid byproduct and driving the equilibrium toward the anhydride. Crucially, the low thermal energy of the system suppresses the formation of oxazolone intermediates, which are the primary culprits for racemization in peptide synthesis. This mechanistic fidelity ensures that the optical purity of the final L-alanyl-L-glutamine remains above 99.8%, a critical specification for regulatory approval in injectable formulations.
Following the coupling reaction, the removal of the formyl protecting group is achieved through acid-catalyzed hydrolysis, a mechanism that is both robust and selective. The formyl amide bond is sufficiently labile under mild acidic conditions (1M HCl at 40°C) to cleave efficiently without degrading the sensitive peptide backbone or the side-chain amide of the glutamine residue. This selectivity is paramount, as harsher conditions could lead to the cyclization of glutamine into pyroglutamic acid, a common impurity that compromises product stability. The process design incorporates a recrystallization step from an ethanol-water system, which leverages the differential solubility of the dipeptide to exclude trace organic impurities and residual salts. This final purification stage guarantees a product with a sharp melting point and consistent specific rotation, meeting the stringent requirements of high-purity L-alanyl-L-glutamine for clinical use.
How to Synthesize L-Alanyl-L-Glutamine Efficiently
The operational protocol for this synthesis is designed for scalability, utilizing standard stainless steel reactors equipped with efficient cooling jackets to maintain the critical sub-zero temperatures required for the activation step. The process begins with the dissolution of N-formyl-L-alanine in anhydrous tetrahydrofuran, followed by the controlled addition of isobutyl chloroformate and N-methylmorpholine. Once the mixed anhydride is formed, L-glutamine is introduced, and the reaction mixture is allowed to warm gradually to 0°C before quenching. The workup involves solvent exchange to iso-butyl acetate, a greener alternative to chlorinated solvents, followed by aqueous washes to remove inorganic salts. The detailed standardized synthesis steps, including precise molar ratios, stirring times, and crystallization parameters, are outlined below for technical reference.
- Activate N-formyl-L-alanine with isobutyl chloroformate and N-methylmorpholine in THF at -18°C to form a mixed anhydride intermediate.
- Couple the activated intermediate with L-glutamine in THF while maintaining temperature below -15°C to prevent racemization.
- Deprotect the N-formyl group using 1M hydrochloric acid at 40°C, followed by pH adjustment and recrystallization from ethanol-water to achieve 99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing route offers profound advantages that directly address the pain points of cost volatility and supply continuity in the fine chemical sector. By eliminating the dependency on precious metal catalysts and hazardous gaseous reagents, the process significantly reduces the complexity of the supply chain and mitigates the risks associated with handling dangerous goods. The use of recyclable solvents like iso-butyl acetate and tetrahydrofuran further enhances the economic viability of the process, allowing for substantial cost savings through solvent recovery loops. For supply chain heads, the robustness of this method translates to reduced lead time for high-purity pharmaceutical intermediates, as the simplified workflow minimizes batch cycle times and equipment turnaround. The avoidance of complex filtration steps to remove urea byproducts also streamlines the production schedule, enabling higher throughput capacity without proportional increases in capital investment.
- Cost Reduction in Manufacturing: The substitution of expensive Z-protecting groups with the economical formyl group removes the necessity for hydrogenation infrastructure, drastically lowering both CAPEX and OPEX. Additionally, the avoidance of coupling reagents like HOSu and DCC, which generate difficult-to-remove solid waste, simplifies the downstream processing and reduces waste disposal costs. The overall yield improvements observed in this method, combined with the lower cost of raw materials, result in a highly competitive cost structure for the final API intermediate.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as isobutyl chloroformate and N-methylmorpholine, are widely available commodity chemicals with stable global supply networks, reducing the risk of raw material shortages. The process operates under atmospheric pressure after the initial cooling phase, removing the need for specialized high-pressure equipment that often creates bottlenecks in multi-purpose facilities. This operational simplicity ensures consistent batch-to-batch reproducibility, a key factor for maintaining long-term contracts with multinational pharmaceutical clients who require absolute supply security.
- Scalability and Environmental Compliance: The elimination of phosgene and heavy metal catalysts aligns the manufacturing process with increasingly stringent environmental regulations, facilitating easier permitting and reduced regulatory scrutiny. The aqueous workup and alcohol-based recrystallization steps generate waste streams that are easier to treat compared to the halogenated waste from older methods. This environmental compatibility supports the commercial scale-up of complex peptide intermediates, allowing manufacturers to expand capacity from pilot scales to multi-ton production without encountering significant ecological hurdles.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and quality assurance of this specific synthetic pathway. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical implications of adopting this technology. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the N-formyl protecting group preferred over Z-protection for L-alanyl-L-glutamine synthesis?
A: The N-formyl group is significantly cheaper than the benzyloxycarbonyl (Z) group and eliminates the need for hazardous hydrogenation steps involving palladium catalysts, thereby reducing both raw material costs and safety risks.
Q: How does the mixed anhydride method control optical purity during peptide coupling?
A: By strictly controlling the reaction temperature at -18°C to -15°C during the activation and coupling phases, the method minimizes the risk of racemization, ensuring an optical purity of over 99.8%.
Q: What are the primary safety advantages of this process compared to traditional phosgene methods?
A: This process completely avoids the use of phosgene gas, a highly toxic reagent used in older methods, replacing it with safer liquid reagents like isobutyl chloroformate, which greatly improves operational safety and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Alanyl-L-Glutamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthetic routes like the one described in CN101519428B are fully realized in practice. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify optical purity and impurity profiles for every batch of L-alanyl-L-glutamine we produce. Our commitment to quality assurance ensures that our clients receive a product that consistently meets the demanding standards of the global parenteral nutrition market.
We invite procurement directors and R&D leaders to engage with our technical procurement team to discuss how this optimized manufacturing process can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic impact of switching to this superior synthetic route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless integration of high-quality intermediates into your final drug products.
