Advanced Manufacturing of Perindopril Intermediates via Activated Sulfinyl Coupling
Introduction to Next-Generation Perindopril Manufacturing
The global demand for antihypertensive agents continues to drive the need for robust, scalable, and high-purity synthetic routes for Angiotensin-Converting Enzyme (ACE) inhibitors. Patent CN1802384B introduces a transformative methodology for the synthesis of Perindopril and its pharmaceutically acceptable salts, specifically addressing the critical bottlenecks found in legacy manufacturing processes. This intellectual property outlines a sophisticated chemical strategy that replaces problematic coupling reagents with a novel sulfinyl-chloride activation system, fundamentally altering the impurity profile of the final Active Pharmaceutical Ingredient (API). For R&D directors and supply chain stakeholders, this patent represents a significant opportunity to optimize production costs while ensuring stringent quality standards required by international regulatory bodies. The technical breakthrough lies in the precise control of stereochemistry and the elimination of tenacious by-products that have historically plagued Perindopril synthesis.
By leveraging the specific reactivity of 1H-imidazole-1-sulfinyl chloride, the inventors have established a pathway that not only improves overall yield but also simplifies downstream processing. This is particularly vital for a reliable pharmaceutical intermediates supplier aiming to deliver consistent quality in a competitive market. The process moves away from the traditional reliance on carbodiimides, which generate insoluble ureas, towards a cleaner activation mechanism that facilitates easier purification. As we delve into the mechanistic details and commercial implications, it becomes clear that this technology offers a viable solution for cost reduction in API manufacturing without compromising the structural integrity or pharmacological efficacy of the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Perindopril, as described in earlier patents like EP 0308341, relied heavily on peptide coupling strategies utilizing dicyclohexylcarbodiimide (DCC). While chemically effective for bond formation, the use of DCC introduces severe downstream processing challenges that impact both cost and quality. The primary issue is the formation of dicyclohexylurea (DCU) as a stoichiometric by-product. DCU is notoriously difficult to remove completely from the reaction mixture due to its low solubility in many organic solvents and its tendency to occlude within the crystal lattice of the product. This necessitates extensive washing, recrystallization, or even chromatographic purification steps, all of which erode overall yield and increase solvent consumption. Furthermore, DCC coupling can lead to racemization at the chiral centers, generating diastereomeric impurities that are structurally similar to the target molecule and difficult to separate. These impurities pose a significant risk to patient safety and require rigorous analytical control, adding time and expense to the quality assurance workflow.
The Novel Approach
The methodology disclosed in CN1802384B circumvents these issues by employing a unique activation strategy involving sulfinyl chlorides. Instead of forming a urea by-product, the reaction proceeds through the formation of a cyclic oxathiazolidinone intermediate. This activated species is highly reactive towards nucleophilic attack by the amine component but decomposes into soluble, easily removable by-products upon reaction completion. 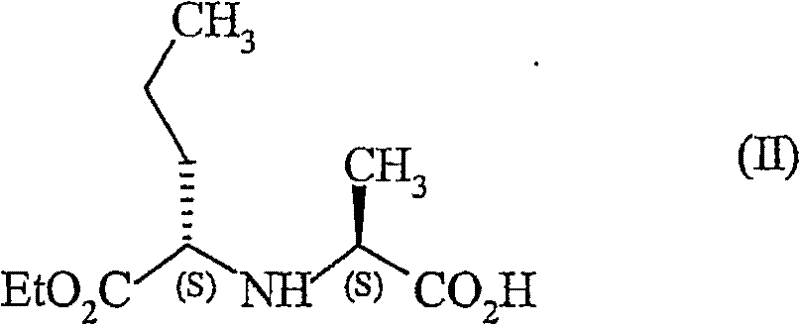 This shift in chemistry effectively eliminates the DCU filtration bottleneck, allowing for a more streamlined isolation of the coupled product. The process begins with the activation of the N-[(S)-1-carboxybutyl]-(S)-alanine ethyl ester derivative. By reacting this starting material with 1H-imidazole-1-sulfinyl chloride, the carboxylic acid is converted into a highly electrophilic intermediate ready for coupling. This approach ensures that the stereochemical integrity of the chiral centers is maintained throughout the synthesis, which is critical for the biological activity of Perindopril. The result is a cleaner reaction profile that supports higher throughput and reduced waste generation.
This shift in chemistry effectively eliminates the DCU filtration bottleneck, allowing for a more streamlined isolation of the coupled product. The process begins with the activation of the N-[(S)-1-carboxybutyl]-(S)-alanine ethyl ester derivative. By reacting this starting material with 1H-imidazole-1-sulfinyl chloride, the carboxylic acid is converted into a highly electrophilic intermediate ready for coupling. This approach ensures that the stereochemical integrity of the chiral centers is maintained throughout the synthesis, which is critical for the biological activity of Perindopril. The result is a cleaner reaction profile that supports higher throughput and reduced waste generation.
Mechanistic Insights into Sulfinyl-Mediated Peptide Coupling
The core innovation of this patent lies in the formation and reactivity of the oxathiazolidinone ring system. When the starting amino acid derivative reacts with the sulfinyl chloride, it undergoes a cyclization to form the intermediate shown in the figure below. 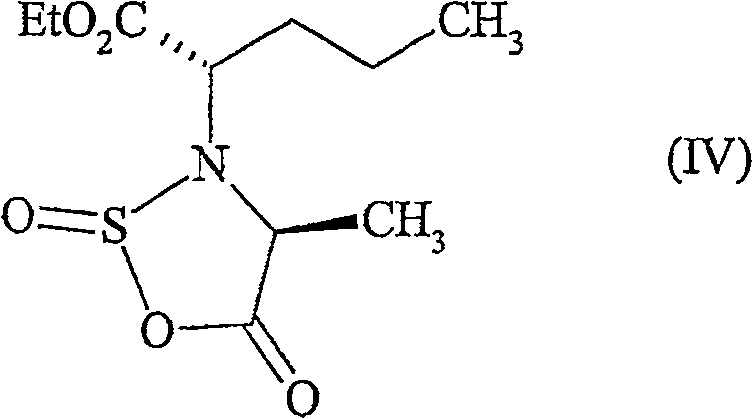 This five-membered ring containing sulfur, nitrogen, and oxygen atoms serves as a potent acylating agent. The electron-withdrawing nature of the sulfinyl group activates the carbonyl carbon, making it susceptible to nucleophilic attack by the secondary amine of the hexahydroindole moiety. Unlike mixed anhydrides or active esters which might suffer from hydrolysis or slow reaction rates, this sulfinyl-activated species reacts rapidly and selectively at ambient temperatures. The mechanism avoids the formation of stable urea linkages, instead releasing imidazole and sulfur dioxide derivatives that are either volatile or water-soluble, facilitating their removal during the aqueous workup phase. This mechanistic elegance translates directly to operational simplicity in a plant setting.
This five-membered ring containing sulfur, nitrogen, and oxygen atoms serves as a potent acylating agent. The electron-withdrawing nature of the sulfinyl group activates the carbonyl carbon, making it susceptible to nucleophilic attack by the secondary amine of the hexahydroindole moiety. Unlike mixed anhydrides or active esters which might suffer from hydrolysis or slow reaction rates, this sulfinyl-activated species reacts rapidly and selectively at ambient temperatures. The mechanism avoids the formation of stable urea linkages, instead releasing imidazole and sulfur dioxide derivatives that are either volatile or water-soluble, facilitating their removal during the aqueous workup phase. This mechanistic elegance translates directly to operational simplicity in a plant setting.
Following the coupling step, the synthesis requires the saturation of the indole double bond to form the octahydroindole core characteristic of Perindopril. This is achieved through a catalytic hydrogenation step using noble metal catalysts such as platinum on carbon (Pt/C). The patent specifies mild conditions, typically around 5 bar of hydrogen pressure, which is sufficient to reduce the C=C bond without affecting the ester or amide functionalities. 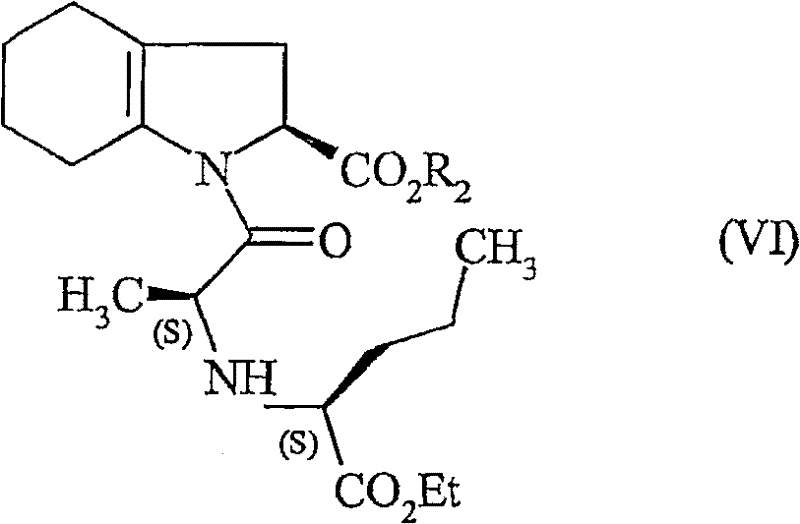 The stereochemical outcome of this hydrogenation is crucial; the catalyst surface directs the addition of hydrogen to ensure the correct cis-fusion of the ring system (3aS, 7aS configuration). The ability to control this stereochemistry under mild pressure conditions minimizes the risk of over-reduction or epimerization, ensuring that the final impurity profile remains within tight specifications. This level of control is essential for meeting the rigorous purity standards demanded by global health authorities for cardiovascular medications.
The stereochemical outcome of this hydrogenation is crucial; the catalyst surface directs the addition of hydrogen to ensure the correct cis-fusion of the ring system (3aS, 7aS configuration). The ability to control this stereochemistry under mild pressure conditions minimizes the risk of over-reduction or epimerization, ensuring that the final impurity profile remains within tight specifications. This level of control is essential for meeting the rigorous purity standards demanded by global health authorities for cardiovascular medications.
How to Synthesize Perindopril Intermediates Efficiently
The practical execution of this synthesis involves a sequence of well-defined unit operations that are compatible with standard multipurpose pharmaceutical manufacturing facilities. The process initiates with the activation of the amino acid component in a chlorinated solvent like dichloromethane at controlled low temperatures to manage exotherms. Following the formation of the activated intermediate, the hexahydroindole acid is introduced in the presence of a base such as triethylamine to scavenge the generated acid. The subsequent hydrogenation step is performed in acetic acid, a solvent choice that enhances the solubility of the polar intermediate and stabilizes the catalyst. Finally, the free acid is converted to the stable tert-butylamine salt through a crystallization process in ethyl acetate.
- Activate N-[(S)-1-carboxybutyl]-(S)-alanine ethyl ester using 1H-imidazole-1-sulfinyl chloride to form the oxathiazolidinone intermediate.
- Couple the activated intermediate with (2S)-2,3,4,5,6,7-hexahydro-1H-indole-2-carboxylic acid in the presence of triethylamine.
- Perform catalytic hydrogenation using Pt/C at mild pressure (5 bar) to saturate the indole ring, followed by salt formation with tert-butylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond simple chemistry. The elimination of DCC removes a significant cost driver associated with expensive coupling reagents and the disposal of hazardous urea waste. Furthermore, the simplified purification protocol reduces the consumption of solvents and the time required for isolation, directly impacting the cost of goods sold (COGS). The reliance on standard hydrogenation equipment operating at low pressures means that this process can be scaled up in existing facilities without the need for significant capital expenditure on high-pressure reactors. This flexibility ensures a more resilient supply chain capable of responding to market fluctuations.
- Cost Reduction in Manufacturing: The replacement of DCC with sulfinyl chloride activation eliminates the formation of dicyclohexylurea, a solid by-product that traditionally requires energy-intensive filtration and washing steps to remove. By avoiding this bottleneck, the process significantly reduces labor hours and solvent usage associated with purification. Additionally, the high yield reported in the salt formation step (up to 95% in examples) indicates minimal material loss, maximizing the efficiency of raw material utilization. This logical deduction suggests a substantial decrease in production costs per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as 1H-imidazole-1-sulfinyl chloride and platinum on carbon, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate ambient temperatures and mild pressures, minimizes the likelihood of batch failures due to equipment malfunction or operational deviations. This reliability ensures consistent delivery schedules for downstream formulation partners, securing the continuity of supply for finished dosage forms.
- Scalability and Environmental Compliance: The process generates fewer hazardous solid wastes compared to conventional methods, aligning with modern green chemistry principles and reducing the burden on waste treatment facilities. The use of catalytic hydrogenation rather than stoichiometric reducing agents further minimizes the environmental footprint of the manufacturing process. The scalability is evidenced by the straightforward workup procedures involving pH adjustments and crystallizations, which are easily transferred from pilot scale to multi-ton commercial production without complex engineering modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities.
Q: How does this new method improve impurity profiles compared to DCC coupling?
A: The novel method eliminates the use of dicyclohexylcarbodiimide (DCC), thereby preventing the formation of dicyclohexylurea (DCU), a by-product known for its difficult removal and tendency to co-crystallize with the API, ensuring higher purity.
Q: What are the safety advantages of the hydrogenation conditions described?
A: The process utilizes mild hydrogen pressures ranging from 1 to 10 bar (specifically 5 bar in examples), which significantly reduces high-pressure safety risks and allows for the use of standard industrial hydrogenation reactors rather than specialized high-pressure vessels.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route avoids chromatographic purifications and relies on crystallization and filtration steps, such as the final salt formation which achieved high yields, making it highly amenable to multi-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perindopril Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity Perindopril intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. Our infrastructure is designed to handle complex chemistries, including sensitive hydrogenation steps and stereocontrolled couplings, with the utmost precision and safety.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to this DCC-free methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a secure and efficient supply of this vital cardiovascular intermediate.
