Revolutionizing Tenofovir Production: A Green Synthetic Route via Configuration Inversion
Revolutionizing Tenofovir Production: A Green Synthetic Route via Configuration Inversion
The pharmaceutical industry is constantly seeking more sustainable and economically viable pathways for the production of critical antiviral agents like Tenofovir (PMPA). Patent CN111205326A, published in May 2020, introduces a groundbreaking green and environment-friendly preparation method that fundamentally alters the raw material economics of this essential HIV and HBV treatment. Unlike conventional routes that rely on expensive chiral resolutions or specific R-configured starting materials, this innovative process leverages S-propylene carbonate, a typically underutilized byproduct of chiral resolution processes, as the primary feedstock. By integrating a strategic Mitsunobu configuration inversion, the technology transforms what was once considered waste into a high-value intermediate, offering a compelling solution for manufacturers aiming to reduce both their carbon footprint and production costs. This report analyzes the technical feasibility, mechanistic advantages, and commercial implications of this novel synthetic strategy for global procurement and R&D stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tenofovir has been plagued by inefficiencies related to chirality management and raw material utilization. Traditional industrial routes often commence with R-propylene carbonate or require the resolution of racemic mixtures to obtain the necessary chiral building blocks. In these legacy processes, the S-enantiomer of propylene carbonate is frequently generated as a coproduct or waste stream during the resolution of the R-isomer, leading to a theoretical maximum atom economy of only 50% regarding the propylene carbonate feedstock. Furthermore, many existing patents describe multi-step sequences involving hazardous reagents or complex purification protocols that generate significant volumes of organic waste, complicating wastewater treatment and increasing the overall environmental compliance burden for manufacturing facilities. The reliance on specific R-configured starting materials also creates supply chain vulnerabilities, as the availability of high-purity R-propylene carbonate can fluctuate based on market demand for other chiral intermediates, potentially causing production bottlenecks.
The Novel Approach
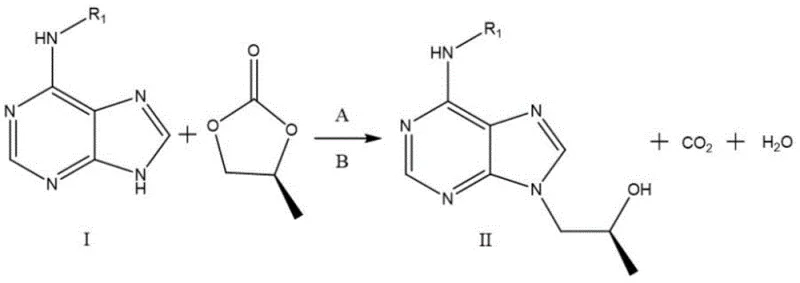
The methodology disclosed in CN111205326A elegantly circumvents these structural inefficiencies by intentionally utilizing S-propylene carbonate as the starting material. The core innovation lies in the acceptance of the S-configuration at the outset and the subsequent application of a Mitsunobu reaction to invert the stereochemistry to the biologically active R-configuration found in Tenofovir. This approach effectively doubles the potential utility of racemic propylene carbonate sources, as manufacturers can now valorize the S-fraction that was previously discarded. The process is streamlined into three distinct operational stages: an initial N-alkylation to attach the chiral side chain, a stereoinversion coupling with a phosphonate ester, and a final hydrolysis to reveal the free phosphonic acid. By shifting the paradigm from 'resolution and discard' to 'utilization and inversion,' this method offers a robust framework for cost reduction in pharmaceutical intermediate manufacturing while adhering to stricter green chemistry principles.
Mechanistic Insights into Mitsunobu-Mediated Configuration Inversion
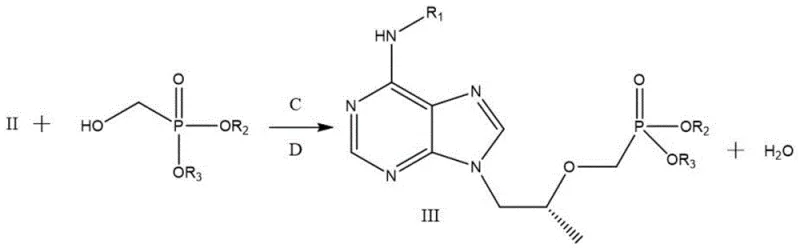
The heart of this synthetic strategy is the second step, which employs the classic Mitsunobu reaction to achieve stereochemical inversion with high fidelity. In this transformation, the secondary hydroxyl group of Intermediate II, which possesses the S-configuration derived from the S-propylene carbonate, acts as the nucleophile precursor. Upon activation by a phosphine (such as triphenylphosphine) and an azodicarboxylate (such as diethyl azodicarboxylate), the hydroxyl group is converted into a good leaving group. Simultaneously, the dialkyl hydroxymethylphosphonate serves as the nucleophile, attacking the chiral center from the backside. This SN2-type mechanism ensures a complete inversion of configuration, converting the S-center into the requisite R-center found in the final Tenofovir molecule. The beauty of this mechanism lies in its predictability and reliability; unlike enzymatic resolutions which can be sensitive to substrate purity, the chemical Mitsunobu inversion proceeds efficiently under mild conditions, typically at room temperature, minimizing thermal degradation of the sensitive purine base.
Impurity control is inherently managed through the choice of reagents and the physical properties of the byproducts. The reaction generates triphenylphosphine oxide and hydrazine dicarboxylate derivatives as stoichiometric byproducts, which are generally solid and can often be removed via filtration or crystallization during the workup phase described in the patent. The patent specifies the use of inorganic weak bases like potassium carbonate in the first step to facilitate the alkylation without promoting excessive side reactions on the adenine ring. In the final hydrolysis step, the use of inorganic strong bases like sodium hydroxide under ice-bath conditions ensures selective cleavage of the ester groups without damaging the glycosidic bond or the purine ring system. This precise control over reaction parameters results in a crude product with high purity, reducing the need for extensive chromatographic purification and facilitating a simpler isolation protocol via pH adjustment and filtration.
How to Synthesize Tenofovir Efficiently
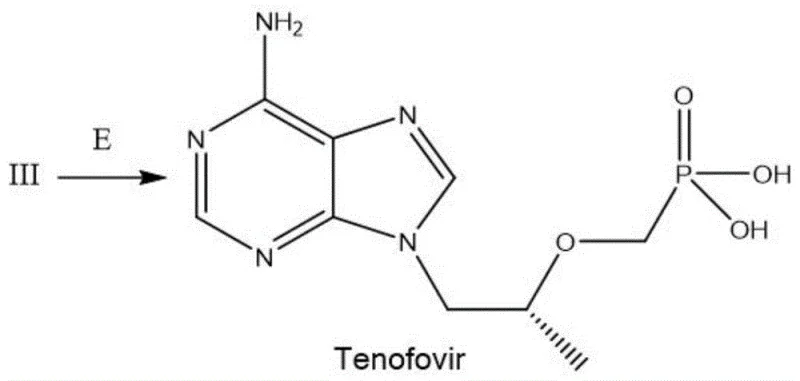
The synthesis of Tenofovir via this green route involves a logical sequence of alkylation, inversion, and deprotection that is amenable to standard reactor setups. The process begins with the dissolution of an adenine derivative (Compound I) and S-propylene carbonate in a polar aprotic solvent such as dimethylformamide or toluene, followed by heating to promote ring opening and N-alkylation. Once Intermediate II is secured, the critical Mitsunobu coupling is performed at ambient temperature to install the phosphonate moiety with the correct stereochemistry. The final step involves a basic hydrolysis to remove the protecting groups and liberate the active phosphonic acid. For detailed operational parameters, stoichiometry, and specific workup instructions, please refer to the standardized synthesis guide below.
- React adenine derivative (Compound I) with S-propylene carbonate and inorganic weak base in organic solvent at 85-120°C to obtain Intermediate II.
- Perform Mitsunobu reaction on Intermediate II with dialkyl hydroxymethylphosphonate, trialkyl phosphine, and azodicarboxylic diester to generate Intermediate III with inverted configuration.
- Hydrolyze Intermediate III using inorganic strong base in an ice bath, adjust pH to 2-3, and isolate pure Tenofovir via filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a multitude of strategic advantages that extend beyond simple yield metrics. The primary value proposition is the decoupling of production costs from the volatile market of single-enantiomer starting materials. By enabling the use of S-propylene carbonate, which is often a lower-cost byproduct of chiral resolution streams, manufacturers can significantly reduce raw material expenditures. Furthermore, the ability to recycle organic solvents like DMF or toluene, as highlighted in the patent disclosure, contributes to substantial cost savings in solvent procurement and waste disposal fees. The process generates wastewater that is predominantly composed of harmless inorganic salts, drastically simplifying the effluent treatment process and lowering the operational expenditure associated with environmental compliance and wastewater management.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolution steps and the utilization of lower-cost S-isomer feedstocks directly impact the bottom line. By avoiding the 50% loss of material inherent in traditional resolution processes, the effective cost per kilogram of the chiral building block is nearly halved. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, further driving down utility costs. The simplicity of the workup, relying on filtration and pH adjustment rather than complex column chromatography, reduces labor hours and consumable costs associated with purification media.
- Enhanced Supply Chain Reliability: Diversifying the source of chiral raw materials enhances supply security. Instead of relying solely on suppliers of R-propylene carbonate, manufacturers can source racemic propylene carbonate or S-isomer streams from a broader range of chemical producers. This flexibility mitigates the risk of supply disruptions caused by capacity constraints at specific chiral synthesis facilities. The robustness of the chemical steps, which do not rely on sensitive biological catalysts, ensures consistent production output regardless of minor fluctuations in raw material quality, providing a more stable supply of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as stirring, heating, filtration, and drying that are standard in multi-ton reactors. The generation of benign inorganic salt waste simplifies the permitting process for new manufacturing lines and reduces the liability associated with hazardous waste storage and transport. As regulatory pressures on pharmaceutical manufacturing emissions increase, adopting a 'green' synthesis route that minimizes organic solvent discharge and maximizes atom economy positions the supplier as a preferred partner for environmentally conscious global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity on process feasibility and product quality. Understanding these nuances is critical for technical teams evaluating the transfer of this technology to commercial scale.
Q: How does this patent solve the waste issue of chiral propylene carbonate?
A: Traditional methods often utilize R-propylene carbonate, discarding the S-isomer during resolution. This patent utilizes the typically wasted S-propylene carbonate as the starting material and employs a Mitsunobu reaction to invert the configuration to the required R-form, thereby doubling the theoretical yield from racemic sources.
Q: What are the environmental advantages of this synthesis route?
A: The process generates wastewater primarily composed of harmless inorganic salt solutions rather than complex organic waste. Additionally, the organic solvents used (such as DMF or toluene) can be recycled, significantly reducing the environmental footprint and treatment costs.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (85-120°C for step 1, room temperature for step 2) and the workup involves simple filtration and pH adjustment, making it highly scalable and safe for industrial manufacturing without requiring complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN111205326A for the global supply of antiviral intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are realized in practical, large-volume manufacturing. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of Tenofovir meets the exacting standards required for FDA and EMA filings. We are committed to leveraging our technical expertise to optimize this inversion-based route, delivering a product that is not only cost-effective but also environmentally sustainable.
We invite forward-thinking pharmaceutical partners to collaborate with us on the commercialization of this advanced synthesis method. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the efficiency and sustainability of your antiviral drug supply chain.
