Scalable Production of High-Purity Tenofovir Alafenamide Intermediates via Novel Thermal Isomerization
Scalable Production of High-Purity Tenofovir Alafenamide Intermediates via Novel Thermal Isomerization
The global demand for effective antiretroviral therapies continues to drive innovation in the synthesis of nucleotide analogs, particularly for the treatment of HIV and Hepatitis B. Patent CN106946935B introduces a groundbreaking preparation method for a critical diastereoisomeric nucleoside derivative, specifically Compound I, which serves as a key intermediate for Tenofovir Alafenamide Fumarate. This novel approach addresses the longstanding challenges associated with P-chirality in phosphonate prodrugs, offering a robust pathway that bypasses the inefficiencies of traditional chiral separation techniques. By leveraging a thermal isomerization strategy, this technology enables the conversion of a racemic mixture into the therapeutically active diastereomer with exceptional selectivity and yield. For R&D directors and procurement specialists, understanding this shift from complex chromatographic separation to thermodynamic control represents a pivotal opportunity for optimizing supply chains and reducing manufacturing costs in the competitive antiviral market.
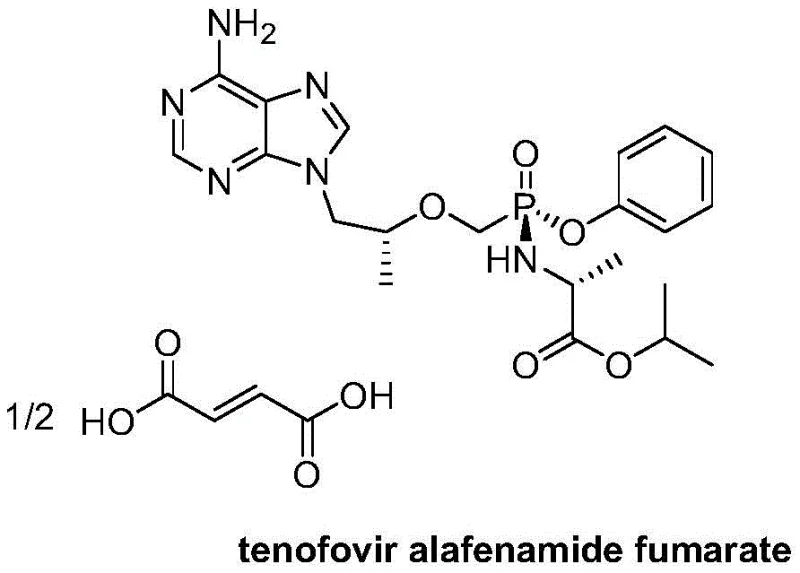
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of P-chiral nucleotide prodrugs like Tenofovir Alafenamide has been plagued by significant technical hurdles regarding stereochemical control. Conventional literature and prior art, such as the methods disclosed in WO2015/40640A2 and related patents, typically rely on the synthesis of a racemic mixture at the phosphorus atom followed by arduous separation processes. These traditional methods often necessitate the use of chiral column chromatography, a technique that is notoriously difficult to scale, time-consuming, and economically inefficient, often resulting in yields of less than 50%. Furthermore, alternative approaches involving chiral induction crystallization have proven operationally difficult, lacking clear in-process control methods and frequently requiring toxic reagents like phenol derivatives. These limitations create substantial bottlenecks in commercial manufacturing, leading to high production costs, extensive waste generation, and unreliable supply continuity for downstream API production.
The Novel Approach
In stark contrast to these legacy methods, the process described in patent CN106946935B utilizes a sophisticated thermal isomerization strategy to achieve high diastereoselectivity. Instead of attempting to separate enantiomers physically, this novel approach involves heating the racemic Compound III in specific organic solvents to induce a configurational conversion, effectively enriching the desired diastereomer Compound I. This method capitalizes on the thermodynamic stability differences between the diastereomers, allowing the system to equilibrate towards the desired product under controlled thermal conditions. The result is a streamlined workflow that replaces complex chromatographic steps with simple crystallization, achieving yields greater than 80% while utilizing cheaper, less toxic solvents like methyl cyclopentyl ether and n-heptane. This paradigm shift not only simplifies the operational complexity but also drastically reduces the environmental footprint of the synthesis.
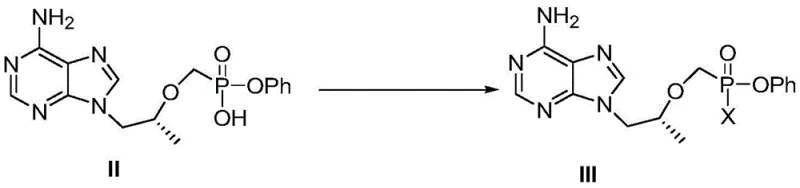
Mechanistic Insights into Thermal Diastereomer Conversion
The core of this technological breakthrough lies in the precise manipulation of the phosphorus chiral center through thermal energy in a carefully selected solvent matrix. The reaction mechanism involves the heating of Compound III, where the phosphorus atom acts as a stereocenter, in solvents such as toluene, n-heptane, or methyl cyclopentyl ether at temperatures ranging from 60°C to 75°C. Under these conditions, the bond dynamics at the phosphorus center allow for epimerization or equilibration, favoring the formation of the thermodynamically more stable diastereomer, Compound I. The choice of solvent plays a critical role in this mechanism; for instance, the use of methyl cyclopentyl ether mixed with n-heptane has been shown to optimize the reaction kinetics, allowing the content of Compound I to exceed 95% within 12 hours. This mechanistic understanding allows process chemists to fine-tune reaction parameters, ensuring consistent quality and minimizing the formation of unwanted isomeric impurities that could complicate downstream purification.
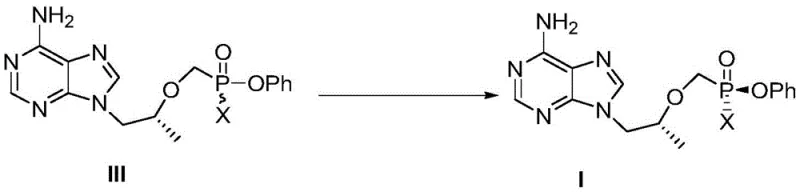
Furthermore, the control of impurities in this process is inherently superior due to the nature of the transformation. Unlike chiral column separation, which merely partitions existing impurities, this thermal conversion actively drives the equilibrium towards the desired product, effectively consuming the unwanted isomer. The subsequent purification step involves a straightforward recrystallization from dichloromethane and n-heptane, which efficiently removes residual starting materials and minor side products. The patent data indicates that this method consistently produces Compound I as a pure white solid powder with high structural integrity, as confirmed by NMR analysis. This high level of purity is essential for meeting the stringent regulatory requirements for pharmaceutical intermediates, ensuring that the final API meets all safety and efficacy standards without the need for excessive reprocessing or refining steps that erode profit margins.
How to Synthesize Compound I Efficiently
The synthesis of Compound I via this patented thermal isomerization route offers a practical and scalable solution for manufacturing teams looking to implement this technology. The process begins with the suspension of the racemic precursor, Compound III, in a green solvent system, followed by controlled heating and monitoring. The simplicity of the workup procedure, involving solvent evaporation and recrystallization, makes it highly attractive for pilot and commercial scale operations. Detailed standard operating procedures and specific stoichiometric ratios for the halogenation of precursor Compound II and the subsequent isomerization are critical for success.
- Suspend Compound III (where X is halogen) in an organic solvent such as methyl cyclopentyl ether, n-heptane, or toluene.
- Heat the reaction mixture to a temperature between 60°C and 75°C while stirring continuously to facilitate configuration conversion.
- Monitor the reaction progress via NMR until the content of Compound I exceeds 95%, then purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route presents a compelling value proposition centered on cost efficiency and operational reliability. The elimination of chiral chromatography, a resource-intensive unit operation, translates directly into significant capital expenditure savings and reduced operational overhead. By shifting to a thermal equilibration process, manufacturers can utilize standard reactor equipment rather than specialized separation columns, thereby enhancing the flexibility and throughput of existing production facilities. Additionally, the use of readily available and less hazardous solvents reduces the logistical burden associated with the storage and handling of toxic chemicals, further contributing to overall cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The transition from low-yield chiral separation (<50%) to high-yield thermal isomerization (>80%) fundamentally alters the cost structure of the intermediate. By nearly doubling the effective yield from the same amount of starting material, the cost per kilogram of the active intermediate is drastically reduced. Furthermore, the avoidance of expensive chiral stationary phases and the reduction in solvent consumption during purification contribute to substantial cost savings. This economic efficiency allows for more competitive pricing strategies in the global market for antiviral drugs, providing a distinct advantage in tender negotiations and long-term supply contracts.
- Enhanced Supply Chain Reliability: The robustness of this thermal process ensures a more stable and predictable supply of critical intermediates. Traditional methods prone to column fouling or inconsistent crystallization can lead to batch failures and delivery delays; however, this new method offers clear reaction endpoints monitored by NMR, ensuring consistent batch-to-batch quality. The reliance on common organic solvents like heptane and toluene, rather than specialized or regulated reagents, mitigates the risk of supply disruptions caused by raw material shortages. This reliability is crucial for maintaining continuous API production schedules and meeting the rigorous demands of global pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with modern green chemistry principles. The reduction in waste generation, evidenced by the simpler workup and higher atom economy, facilitates easier compliance with increasingly strict environmental regulations. The process generates fewer hazardous byproducts compared to prior art methods that utilize toxic phenol derivatives, simplifying waste disposal and lowering environmental compliance costs. This sustainability profile not only enhances the corporate social responsibility standing of the manufacturer but also future-proofs the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diastereomer preparation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, solvent choices, and scalability potential. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of this new preparation method over prior art?
A: The primary advantage is the significantly higher yield (>80%) and simplified purification process. Unlike prior art which relies on difficult chiral column separation with yields under 50%, this method uses thermal isomerization followed by simple crystallization.
Q: Which solvents are preferred for the isomerization of Compound III?
A: The patent identifies methyl cyclopentyl ether, n-heptane, and toluene as preferred organic solvents. Specifically, mixtures of methyl cyclopentyl ether and n-heptane have shown optimal results for yield and reaction rate.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for industrial scale-up. It utilizes cheap and easily obtained raw materials, mild reaction conditions, and generates less environmental pollution compared to methods using toxic reagents like phenol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Alafenamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antiviral medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering compounds with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. By leveraging the advanced thermal isomerization technologies described in recent patents, we can offer our partners a reliable source of Tenofovir Alafenamide intermediates that meet the highest industry standards for quality and consistency.
We invite global pharmaceutical partners to collaborate with us to explore the full potential of this optimized synthesis route. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive edge in the antiviral market through superior chemical manufacturing solutions.
