Scalable Liquid-Phase Synthesis of Semaglutide Side Chains for Commercial API Production
The pharmaceutical industry is witnessing a paradigm shift in the manufacturing of GLP-1 receptor agonists, driven by the explosive demand for anti-diabetic and weight-loss therapeutics. Central to this evolution is the efficient production of critical intermediates, specifically the lipidated side chains that confer prolonged half-life to molecules like semaglutide. Patent CN115894664A introduces a groundbreaking liquid-phase synthesis method that addresses the longstanding bottlenecks of cost, waste, and scalability inherent in traditional solid-phase approaches. By optimizing carboxyl activation reagents and leveraging specific solubility properties, this technology enables the direct preparation of high-purity semaglutide side chain analogues. For R&D directors and procurement strategists, this represents a vital opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering complex peptide modifiers with superior economic and environmental profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polypeptide side chains has been dominated by solid-phase peptide synthesis (SPPS) strategies, such as the Fmoc protocol. While effective for small-scale laboratory discovery, SPPS suffers from severe limitations when translated to industrial manufacturing. The primary drawback is the reliance on expensive polymeric supports, which account for a disproportionate share of raw material costs and generate massive amounts of solid waste that is difficult to dispose of sustainably. Furthermore, the atom economy of solid-phase reactions is inherently poor due to the excess reagents required to drive reactions on a heterogeneous support. Existing liquid-phase alternatives often fail to resolve these issues completely, as they typically necessitate tedious purification steps like column chromatography after every coupling reaction, leading to significant product loss and excessive solvent consumption.
The Novel Approach
The methodology disclosed in CN115894664A fundamentally reengineers the synthesis pathway by adopting a fully liquid-phase strategy that eliminates the need for polymer resins entirely. Instead of relying on protected amino acid building blocks that require additional deprotection steps, this novel approach utilizes unprotected L-glutamic acid derivatives and AEEA linkers directly. By optimizing the reaction solvent system, the process exploits the differential solubility of the reaction products versus impurities, allowing for purification via simple crystallization or washing rather than complex chromatography. This shift not only drastically simplifies the operational workflow but also aligns with green chemistry principles by reducing the volume of organic waste, thereby offering a robust solution for cost reduction in API manufacturing.
Mechanistic Insights into Liquid-Phase Amide Condensation and Activation
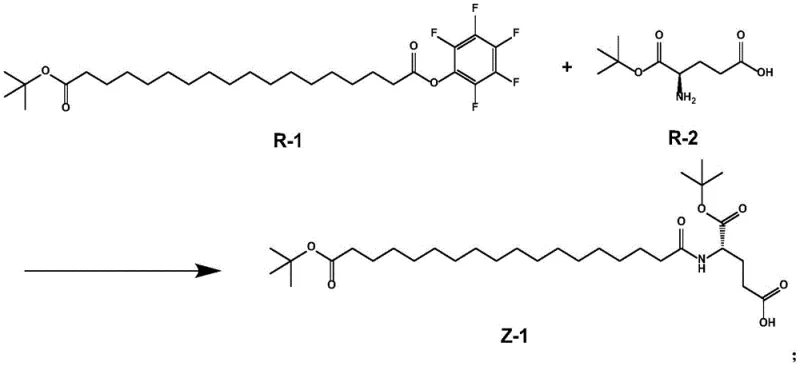
The core of this synthetic innovation lies in a meticulously orchestrated three-step sequence that maximizes reaction efficiency while minimizing side reactions. The first stage involves the amide condensation between octadecanedioic acid mono-tert-butyl ester pentafluorophenol ester (R-1) and L-glutamic acid 1-tert-butyl ester (R-2). Unlike traditional methods that might require harsh conditions, this reaction proceeds smoothly at mild temperatures between 20°C and 35°C in the presence of organic bases like DIEA. The use of a pentafluorophenol ester as the starting electrophile ensures high reactivity towards the nucleophilic amine of the glutamic acid derivative, facilitating rapid bond formation without the need for external coupling agents that could introduce impurities.
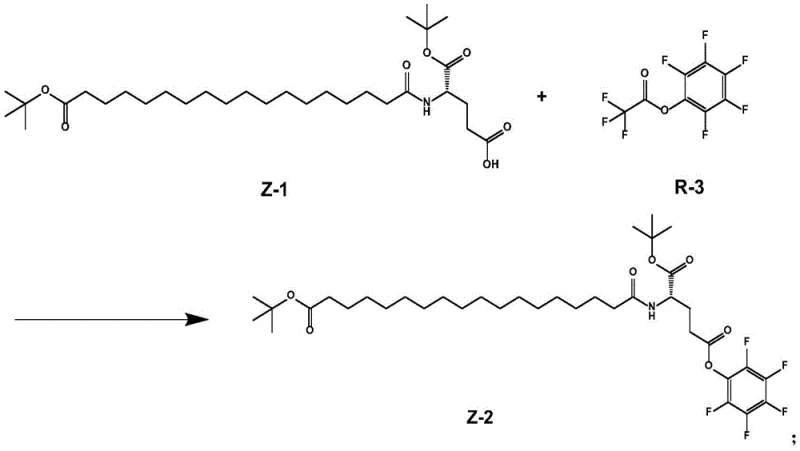
Following the initial coupling, the second critical mechanistic step involves the activation of the free carboxyl group on the intermediate Z-1. This is achieved using pentafluorophenol trifluoroacetate (R-3), a highly efficient activating agent that converts the carboxylic acid into a reactive pentafluorophenyl ester (Z-2). This activation is performed in non-polar solvents like petroleum ether, which promotes the precipitation of the product, further aiding in purification. The choice of R-3 is pivotal; it provides superior stability compared to other active esters while maintaining sufficient electrophilicity for the subsequent coupling step, ensuring that the intermediate remains intact during isolation and storage.
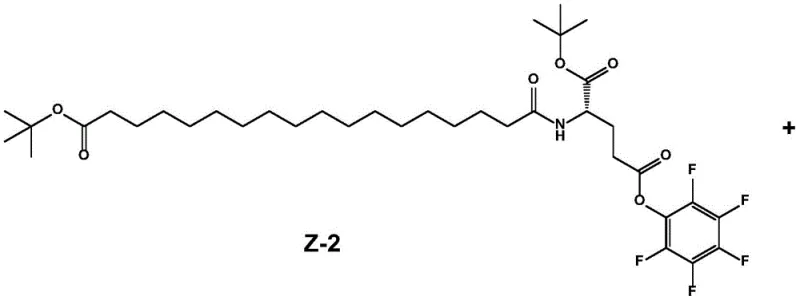
The final coupling step connects the activated lipidated fragment (Z-2) with the hydrophilic linker unit (X), which consists of AEEA (2-(2-aminoethoxy)ethoxy) acetic acid derivatives. A key mechanistic advantage here is the use of unprotected amine linkers (where R is H, phenyl, or ethyl). In conventional syntheses, these amines would require Fmoc protection, adding two extra synthetic steps (protection and deprotection) to the overall process. By bypassing this requirement, the reaction tolerance is significantly enhanced, and the overall yield is preserved. The reaction proceeds in polar aprotic solvents like DMF, where the solubility of both reactants is optimized, leading to near-quantitative conversion rates as evidenced by TLC monitoring.
How to Synthesize Semaglutide Side Chain Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure consistent quality and yield. The process is designed to be operationally simple, avoiding the need for specialized equipment beyond standard reactor vessels capable of temperature control and nitrogen protection. The following guide outlines the standardized procedure derived from the patent examples, demonstrating how to achieve high-purity intermediates and final products through controlled crystallization and extraction techniques. Detailed standardized synthesis steps are provided below.
- Condense octadecanedioic acid mono-tert-butyl ester pentafluorophenol ester (R-1) with L-glutamic acid 1-tert-butyl ester (R-2) using an organic base in ethanol to form intermediate Z-1.
- Activate the carboxyl group of intermediate Z-1 using pentafluorophenol trifluoroacetate (R-3) in petroleum ether to generate the activated ester intermediate Z-2.
- Couple intermediate Z-2 with the AEEA-AEEA derivative (X) in DMF using an organic base, followed by purification to obtain the final semaglutide side chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition from solid-phase to this optimized liquid-phase methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of expensive polymer resins immediately lowers the bill of materials, while the reduction in purification steps translates to shorter cycle times and higher throughput. This process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, as it relies on unit operations like crystallization and filtration that are easily scalable from pilot plants to multi-ton production facilities without the engineering challenges associated with handling large volumes of swollen resin beads.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the removal of Fmoc protection groups from the starting materials. In traditional routes, the cost of protecting reagents and the solvents required for their removal constitutes a major expense. By utilizing unprotected L-glutamic acid and AEEA derivatives directly, the process eliminates these entire cost centers. Furthermore, the ability to purify intermediates via crystallization rather than silica gel column chromatography drastically reduces the consumption of stationary phases and eluents, leading to substantial savings in waste disposal and raw material procurement costs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals and widely available solvents such as ethanol, petroleum ether, and DMF. Unlike specialized resins or exotic coupling reagents that may face supply bottlenecks, the inputs for this process are standard industrial chemicals with robust global supply networks. Additionally, the simplified workflow reduces the dependency on highly skilled labor for complex chromatographic separations, mitigating operational risks and ensuring consistent delivery schedules for high-purity semaglutide side chain batches.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this liquid-phase route is markedly superior. The reduction in solvent usage per kilogram of product lowers the facility's carbon footprint and volatile organic compound (VOC) emissions. The absence of solid polymer waste simplifies effluent treatment and solid waste management, ensuring compliance with increasingly stringent environmental regulations. This sustainability profile not only reduces regulatory risk but also aligns with the corporate social responsibility goals of major pharmaceutical partners seeking greener supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity, scalability, and process robustness for potential partners evaluating this route for their supply chain.
Q: How does this liquid-phase method improve upon traditional solid-phase synthesis?
A: Unlike solid-phase synthesis which relies on expensive polymer supports and generates significant waste, this liquid-phase method utilizes direct coupling of unprotected amino acids, eliminating costly resin materials and simplifying the purification process through crystallization rather than column chromatography.
Q: What are the key advantages of using unprotected L-glutamic acid derivatives in this process?
A: Using unprotected L-glutamic acid 1-tert-butyl ester removes the need for Fmoc protection and subsequent deprotection steps. This significantly shortens the synthetic route, reduces reagent consumption, and minimizes the formation of by-products associated with protection group chemistry.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for scalability. It operates at mild temperatures (20-35°C), uses common organic solvents like ethanol and petroleum ether, and achieves high yields (up to 94% for intermediates) with simplified workup procedures, making it ideal for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semaglutide Side Chain Supplier
As the global demand for GLP-1 analogs continues to surge, securing a stable supply of high-quality side chain intermediates is critical for maintaining competitive advantage in the pharmaceutical market. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced liquid-phase synthesis technologies to deliver exceptional value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of multinational corporations. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of semaglutide side chain meets the exacting standards required for API synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how our innovative synthesis capabilities can support your long-term growth in the diabetes and obesity therapeutic markets.
