Industrial Scale Liquid Phase Synthesis of Semaglutide Side Chain for Global Pharma Supply
The pharmaceutical industry continues to witness unprecedented demand for glucagon-like peptide-1 (GLP-1) analogs, with Semaglutide standing out as a cornerstone therapy for type 2 diabetes and obesity management. The efficacy of this molecule is heavily dependent on its unique fatty diacid side chain, which facilitates albumin binding and extends the drug's half-life in vivo. Patent CN111269137A introduces a robust liquid phase synthesis method for constructing this critical side chain, specifically the Lys(AEEA-AEE-γ-Glu-Octadecanedioic Acid Mono-tert-butylester) moiety. Unlike traditional solid-phase peptide synthesis (SPPS) which often faces limitations in scalability and cost due to resin usage, this liquid phase approach offers a streamlined pathway suitable for multi-kilogram to ton-scale manufacturing. The structural complexity of the target molecule, featuring multiple ether linkages and amide bonds, requires precise control over reaction conditions to ensure high purity and yield.
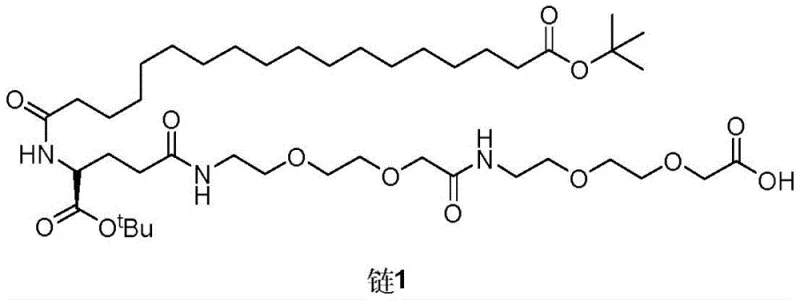
This technical disclosure provides a comprehensive roadmap for producing the side chain intermediate, often referred to as Chain 1, through a series of condensation and deprotection steps. By shifting the synthesis paradigm from solid support to solution phase, manufacturers can leverage standard chemical engineering unit operations, thereby reducing capital expenditure and operational complexity. The method described ensures that the final product meets the stringent quality requirements necessary for subsequent conjugation to the peptide backbone, addressing a critical bottleneck in the supply chain of this high-value active pharmaceutical ingredient (API).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex peptide side chains like those found in Semaglutide has relied heavily on solid-phase peptide synthesis (SPPS). While SPPS offers automation and ease of purification for small research batches, it presents significant challenges when transitioning to commercial scale. The primary limitation lies in the cost and capacity of the resin; large-scale reactors filled with resin have poor heat and mass transfer properties, leading to inconsistent reaction rates and potential hotspots. Furthermore, the excessive use of solvents required to swell the resin and wash away byproducts generates substantial waste, increasing both environmental compliance costs and raw material expenses. The loading capacity of resins also limits the throughput, making it difficult to achieve the economies of scale required for blockbuster drugs with global demand.
The Novel Approach
The liquid phase method detailed in the patent circumvents these issues by utilizing solution-phase chemistry, which allows for better mixing, temperature control, and reaction monitoring. A key innovation in this route is the one-pot synthesis of the PEG-like linker segment, where the amino group of 2-(2-aminoethoxy)ethanol is protected and subsequently elongated via nucleophilic substitution with an alpha-haloester. This strategy minimizes isolation steps and reduces the overall processing time. By employing orthogonal protecting groups such as Boc and benzyl esters, the synthesis achieves high selectivity, ensuring that reactive functional groups are masked until needed. This approach not only improves the overall yield but also simplifies the purification workflow, as intermediates can be crystallized or extracted using standard techniques rather than preparative HPLC.
Mechanistic Insights into Liquid Phase Condensation and Protection Strategies
The core of this synthetic route relies on efficient amide bond formation using carbodiimide-based coupling reagents. Specifically, the condensation reactions utilize EDCI (1-(3-Dimethylaminopropyl)-3-ethylcarbodiimide) in combination with HOBT (1-Hydroxybenzotriazole) or HATU. The mechanism involves the activation of the carboxylic acid by EDCI to form an O-acylisourea intermediate, which is then converted into a more stable active ester by HOBT. This active ester is highly susceptible to nucleophilic attack by the amine component, driving the formation of the amide bond with minimal racemization. The patent specifies optimal molar ratios, such as 1:1 for EDCI and HOBT, and reaction temperatures between 20°C and 40°C, which are critical for suppressing side reactions like N-acylurea formation. Maintaining these parameters ensures that the stereochemical integrity of the chiral centers, particularly in the glutamic acid residue, is preserved throughout the synthesis.
Impurity control is another vital aspect addressed by the mechanistic design of this route. The use of distinct protecting groups allows for sequential deprotection without compromising the rest of the molecule. For instance, the removal of the Boc group is achieved using trifluoroacetic acid (TFA) in dichloromethane, a mild acidic condition that leaves benzyl esters intact. Conversely, the final deprotection of the benzyl ester is performed using inorganic bases like lithium hydroxide in methanol, which cleaves the ester without affecting the amide bonds. This orthogonality prevents the formation of truncated sequences or deletion impurities that are common in less controlled synthesis methods. Additionally, the liquid phase environment facilitates the removal of urea byproducts generated during coupling through aqueous workups, resulting in a cleaner crude product profile before final purification.
How to Synthesize Semaglutide Side Chain Efficiently
The synthesis of the Semaglutide side chain via this liquid phase method involves a logical sequence of protection, chain elongation, and coupling steps that can be executed in standard glass-lined or stainless steel reactors. The process begins with the modification of simple starting materials like 2-(2-aminoethoxy)ethanol and proceeds through several intermediates including Compound 7, Compound 10, and Compound 13 before yielding the final Chain 1. Each step has been optimized for yield and purity, with specific attention paid to solvent selection and reagent stoichiometry. The detailed standardized synthesis steps for implementing this route in a GMP environment are outlined below.
- Protect 2-(2-aminoethoxy)ethanol with Boc and perform nucleophilic substitution with alpha-haloester to form the protected fatty chain.
- Condense the deprotected amine and acid components to form the PEG-like linker structure (Compound 7).
- Couple with Fmoc-Glu-OtBu and subsequently with octadecanedioic acid derivative, followed by final deprotection to yield Chain 1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from solid-phase to liquid-phase synthesis represents a strategic opportunity to optimize cost structures and secure supply continuity. The elimination of expensive polymeric resins directly reduces the bill of materials, while the ability to use commodity solvents like ethanol, methanol, and dichloromethane simplifies sourcing logistics. Furthermore, the higher throughput of liquid phase reactors means that production campaigns can be shorter, allowing for more flexible scheduling and faster response to market demand fluctuations. This method effectively decouples the production of the side chain from the limitations of specialized peptide synthesis equipment, enabling manufacture in versatile multipurpose chemical facilities.
- Cost Reduction in Manufacturing: The liquid phase approach significantly lowers manufacturing costs by removing the dependency on high-cost solid supports and reducing solvent volumes per unit of product. The one-pot synthesis strategy for the linker segment further reduces labor and utility costs by minimizing the number of isolation and purification steps. Additionally, the use of robust coupling reagents like EDCI/HOBT, which are available in bulk quantities at competitive prices, ensures that reagent costs remain stable and predictable. These factors collectively contribute to a more economical production process without compromising the quality of the final intermediate.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials and standard chemical reagents, this synthesis route mitigates the risk of supply disruptions associated with specialized peptide building blocks. The process does not require custom-made resins or proprietary catalysts, meaning that raw materials can be sourced from multiple qualified vendors globally. This diversification of the supply base enhances resilience against geopolitical or logistical shocks. Moreover, the scalability of the liquid phase method ensures that production volumes can be ramped up quickly to meet surging demand for Semaglutide formulations, securing a reliable flow of critical intermediates.
- Scalability and Environmental Compliance: The transition to liquid phase synthesis aligns well with green chemistry principles by reducing waste generation and improving atom economy. The ability to recover and recycle solvents such as dichloromethane and ethyl acetate through distillation further minimizes the environmental footprint of the manufacturing process. From a regulatory perspective, the well-defined reaction parameters and impurity profiles facilitate easier validation and technology transfer to contract manufacturing organizations (CMOs). The process is inherently scalable from pilot plant to commercial tonnage, ensuring that environmental permits and safety protocols can be consistently applied across different production scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the liquid phase synthesis of the Semaglutide side chain. These answers are derived from the specific embodiments and claims found in the patent literature, providing clarity on the feasibility and advantages of this manufacturing route. Understanding these details is crucial for stakeholders evaluating the integration of this technology into their existing supply chains.
Q: What are the primary advantages of liquid phase synthesis over solid phase for this side chain?
A: Liquid phase synthesis eliminates the need for expensive resin supports and reduces solvent consumption per kilogram of product, significantly lowering manufacturing costs and simplifying purification processes compared to traditional SPPS.
Q: Which protecting groups are utilized in this synthetic route?
A: The process employs orthogonal protecting groups such as Boc (tert-butyloxycarbonyl) for amines and benzyl or tert-butyl esters for carboxylic acids, allowing for selective deprotection at different stages without affecting other functional groups.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial application due to lower costs, wider selection of protecting groups, and the ability to handle reactions in standard chemical reactors rather than specialized peptide synthesizers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semaglutide Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful commercialization of complex biopharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Semaglutide side chain meets the highest industry standards. Our facility is equipped to handle the specific solvent systems and reaction conditions required for this liquid phase synthesis, providing a secure and compliant manufacturing environment.
We invite you to collaborate with us to optimize your supply chain for GLP-1 analogs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our expertise in liquid phase peptide synthesis, we can help you reduce lead times and secure a stable supply of this essential intermediate. Please contact us to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
