Advanced Chemo-Enzymatic Synthesis of 2'-Deoxyuridine for Commercial Scale Production
The pharmaceutical industry constantly seeks robust synthetic routes for critical nucleoside analogues like 2'-deoxyuridine, a pivotal intermediate in the development of potent antitumor and antiviral therapeutics. Patent CN102827902B introduces a groundbreaking chemo-enzymatic strategy that effectively merges the precision of biological catalysis with the scalability of chemical synthesis. This hybrid methodology addresses the longstanding challenges of stereochemical control and substrate availability that have historically plagued traditional manufacturing processes. By leveraging crystallization-induced asymmetric transformation technology, the process ensures the production of single-configuration intermediates without the need for complex separation steps. Furthermore, the subsequent biotransformation step utilizes highly specific uridine phosphorylase to achieve high conversion rates under mild conditions. This report analyzes the technical merits and commercial implications of this innovative approach for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for nucleoside analogues often rely heavily on either purely chemical total synthesis or exclusive biotransformation methods, both of which present significant industrial drawbacks. Chemical total synthesis frequently results in mixtures of alpha and beta anomers, necessitating expensive and wasteful purification steps to isolate the pharmacologically active beta-configuration. Moreover, the glycosylation reactions involved typically require large quantities of toxic and expensive chemical reagents, leading to severe environmental pollution and increased waste disposal costs. On the other hand, purely enzymatic approaches, while environmentally friendly, often suffer from the high cost and limited availability of natural deoxynucleoside substrates. These substrate constraints restrict the scalability of biological methods, making it difficult to meet the high-volume demands of the global pharmaceutical market. Consequently, manufacturers face a dilemma between high purity with high cost or lower cost with compromised stereochemical integrity.
The Novel Approach
The novel chemo-enzymatic route disclosed in the patent overcomes these barriers by integrating crystallization-induced asymmetric transformation with efficient enzymatic catalysis. This hybrid strategy utilizes a chemically synthesized intermediate that is converted into a single alpha-configuration species through thermodynamic phase equilibrium, effectively bypassing the need for chromatographic separation of isomers. The resulting intermediate serves as a stable and abundant substrate for the subsequent enzymatic step, eliminating the reliance on scarce natural nucleosides. By combining the robustness of chemical synthesis for backbone construction with the specificity of uridine phosphorylase for base exchange, the process achieves high yields and exceptional purity. This synergistic approach not only simplifies the overall workflow but also significantly reduces the environmental footprint associated with traditional chemical glycosylation. The result is a manufacturing process that is both economically viable and environmentally sustainable for large-scale production.
Mechanistic Insights into Crystallization-Induced Asymmetric Transformation and Enzymatic Catalysis
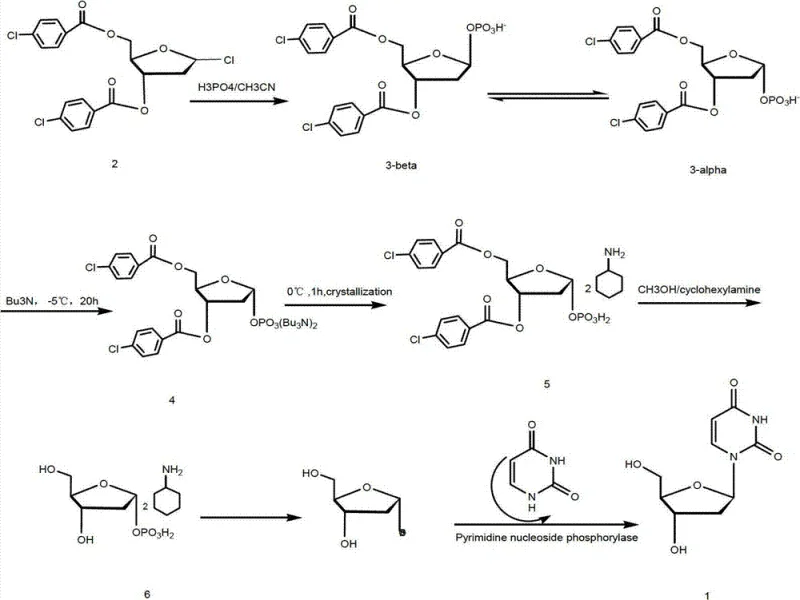
The core of this synthetic innovation lies in the application of crystallization-induced asymmetric transformation (CIAT) to control stereochemistry during the formation of the sugar phosphate intermediate. This technology exploits the thermodynamic phase equilibrium between diastereomers, where the rate of conversion exceeds the rate of crystallization, allowing for the selective precipitation of the most stable isomer. In this specific process, the alpha-configuration of 2-deoxy-alpha-D-ribose-1-phosphate is selectively crystallized from a mixture, driving the equilibrium towards the desired product without the need for chiral resolving agents. This physical-chemical phenomenon ensures that the intermediate obtained possesses a single configuration, which is critical for the specificity of the downstream enzymatic reaction. The ability to control stereochemistry through crystallization rather than complex chemical resolution represents a significant advancement in process chemistry, offering a more direct and practical method for obtaining optically pure intermediates.
Following the chemical preparation of the sugar phosphate, the synthesis proceeds via a biocatalytic transglycosylation reaction mediated by uridine phosphorylase. This enzyme catalyzes the reversible phosphorylation of nucleosides, facilitating the exchange of the uracil base onto the ribose-1-phosphate backbone with high regio- and stereoselectivity. The enzymatic step operates under mild physiological conditions, typically within a pH range of 6 to 8 and temperatures between 45 to 65 degrees Celsius, which preserves the integrity of the sensitive nucleoside structure. The high specificity of the enzyme ensures that only the desired 2'-deoxyuridine is formed, minimizing the formation of by-products and simplifying the final purification process. This combination of thermodynamic control in the chemical step and kinetic control in the biological step creates a highly efficient cascade that maximizes overall process yield.
How to Synthesize 2'-Deoxyuridine Efficiently
Implementing this chemo-enzymatic synthesis requires careful attention to reaction conditions and sequential processing to maximize efficiency and yield. The process begins with the chemical phosphorylation and asymmetric crystallization to generate the key sugar phosphate intermediate, followed by the enzymatic coupling with uracil. Operators must maintain strict control over temperature and water content during the initial chemical phase to ensure proper crystallization of the alpha-anomer. Subsequent deprotection and enzymatic transformation steps require precise pH buffering and temperature regulation to maintain enzyme activity and stability. While the general workflow is outlined here, the specific operational parameters and standardized protocols are critical for successful replication in a manufacturing setting. Detailed standard operating procedures for each stage of the synthesis are provided in the technical guide below to ensure consistency and quality.
- Chemical synthesis of 2-deoxy-alpha-D-ribose-1-phosphate via crystallization-induced asymmetric transformation.
- Deprotection of the intermediate to obtain the free phosphate salt.
- Enzymatic coupling with uracil using uridine phosphorylase to yield 2'-deoxyuridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this chemo-enzymatic technology offers substantial strategic advantages over conventional manufacturing methods. By eliminating the need for expensive chiral resolving agents and reducing the reliance on scarce natural substrates, the process fundamentally alters the cost structure of nucleoside intermediate production. The integration of enzymatic catalysis reduces the number of synthetic steps and minimizes the use of hazardous organic solvents, leading to a cleaner and more sustainable production profile. These improvements translate directly into enhanced supply chain reliability and reduced operational risks associated with regulatory compliance and waste management. The following points detail the specific commercial benefits that this technology brings to industrial-scale manufacturing operations.
- Cost Reduction in Manufacturing: The elimination of toxic chemical reagents and complex purification steps significantly lowers the raw material and processing costs associated with synthesis. By avoiding the use of expensive chiral catalysts and reducing solvent consumption, the overall manufacturing expense is drastically simplified without compromising product quality. This streamlined approach allows for more competitive pricing structures while maintaining healthy profit margins for suppliers. Furthermore, the high conversion rate of the enzymatic step minimizes material loss, ensuring that raw materials are utilized with maximum efficiency throughout the production cycle.
- Enhanced Supply Chain Reliability: The ability to synthesize the key sugar phosphate intermediate chemically ensures a stable and abundant supply of substrates, removing the bottleneck of natural product availability. This independence from fluctuating natural sources enhances the predictability of production schedules and reduces the risk of supply disruptions. The robustness of the chemical step allows for the stockpiling of intermediates, providing a buffer against market volatility and ensuring continuous availability for downstream customers. Consequently, lead times for high-purity nucleoside analogues can be significantly reduced, improving responsiveness to market demands.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous nature of the biotransformation step facilitate easy scale-up from laboratory to industrial production volumes. The process avoids extreme temperatures and pressures, reducing the engineering complexity and capital investment required for manufacturing equipment. Additionally, the environmentally friendly nature of the enzymatic reaction simplifies waste treatment and compliance with environmental regulations, making it easier to obtain necessary permits for expansion. This scalability ensures that the technology can meet growing global demand without significant modifications to the core process design.
Frequently Asked Questions (FAQ)
The following frequently asked questions address common technical and commercial inquiries regarding the implementation of this chemo-enzymatic synthesis route. These answers are derived directly from the patent specifications and technical data to provide accurate guidance for potential adopters. Understanding these key aspects is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows. The responses cover critical topics ranging from stereochemical control to environmental impact and substrate availability.
Q: What is the main advantage of the crystallization-induced asymmetric transformation in this process?
A: It allows for the production of single-configuration intermediates without complex separation, driving equilibrium towards the desired isomer.
Q: How does this method address the substrate limitations of traditional biotransformation?
A: It uses a chemically synthesized sugar phosphate as a substrate, eliminating reliance on scarce natural deoxynucleosides.
Q: What are the environmental benefits of using uridine phosphorylase in this synthesis?
A: The enzymatic step operates under mild conditions with high specificity, reducing toxic waste and energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2'-Deoxyuridine Supplier
Partnering with NINGBO INNO PHARMCHEM provides access to this advanced synthetic technology through our expert CDMO services and extensive manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from development to market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee the quality of every batch produced. By leveraging our expertise in chemo-enzymatic processes, we can help you optimize your supply chain and reduce time-to-market for your critical pharmaceutical intermediates.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how this technology can benefit your product portfolio. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient synthetic route. Our team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us help you engineer a more resilient and cost-effective supply chain for your nucleoside-based therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
