Revolutionizing Methoxyphenamine Production: A Technical Breakdown for Global Pharma Leaders
Introduction to Patent CN102267917B and Technological Breakthroughs
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the production of critical active pharmaceutical ingredients (APIs) and their intermediates. Patent CN102267917B introduces a significant advancement in the synthesis of Methoxyphenamine Hydrochloride, a potent β-adrenergic receptor agonist widely utilized in the management of bronchial asthma. This patent details a robust three-step synthetic route that fundamentally shifts away from the historically cumbersome and waste-intensive methods involving nitro-compounds and stoichiometric metal reductions. By leveraging a novel acylation strategy followed by catalytic hydrogenation, this technology offers a compelling value proposition for reliable pharmaceutical intermediates suppliers aiming to optimize their manufacturing portfolios. The core innovation lies in the substitution of hazardous reagents with safer, more atom-economical alternatives, thereby addressing both economic and ecological concerns simultaneously.
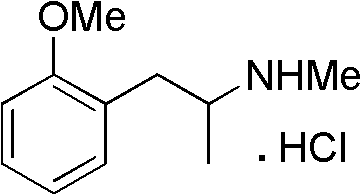
For R&D directors and process chemists, the implications of this patent extend beyond mere regulatory compliance; it represents a tangible opportunity to enhance process safety and product quality. The described methodology ensures high purity profiles, with experimental examples demonstrating HPLC purity levels consistently above 99.5%, which is critical for downstream drug formulation. Furthermore, the operational simplicity of the new route—characterized by mild reaction temperatures and manageable pressure conditions—facilitates easier technology transfer and scale-up. As we delve deeper into the technical specifics, it becomes evident that this synthesis protocol is not just an incremental improvement but a strategic pivot towards greener chemistry that aligns with modern global manufacturing standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Methoxyphenamine Hydrochloride has relied on legacy synthetic routes that are increasingly untenable in the context of modern environmental regulations and cost pressures. The traditional method, often cited in older literature such as J. Am. Chem. Soc. (1953), typically initiates with o-methoxybenzaldehyde undergoing a Henry condensation with nitroethane. This step is fraught with challenges, including the handling of explosive nitro-compounds and the generation of complex reaction mixtures that require rigorous purification. Subsequent reduction steps traditionally employ iron powder and ferric chloride in acidic media, a process notorious for producing vast quantities of iron sludge and acidic wastewater that are expensive and difficult to treat. Additionally, the final reductive amination step often utilizes platinum dioxide (Adams' catalyst), which is not only prohibitively expensive but also difficult to recover efficiently compared to supported catalysts.
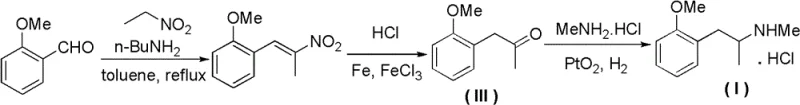
Beyond the environmental liabilities, the conventional approach suffers from significant economic inefficiencies that erode profit margins for manufacturers. The reliance on stoichiometric reducing agents like sodium cyanoborohydride in alternative variations further exacerbates cost issues, as these reagents are pricey and generate toxic cyanide-containing byproducts. Moreover, the separation of isomers in certain Friedel-Crafts type alternatives (using anisole and acetone) results in poor regioselectivity, yielding substantial amounts of meta- and para- isomers that complicate downstream purification and reduce overall yield. These cumulative factors create a supply chain vulnerability, where raw material volatility and waste disposal costs can fluctuate wildly, making long-term planning difficult for procurement managers seeking stability in their API supply chains.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN102267917B presents a streamlined and economically superior alternative that effectively bypasses the pitfalls of the traditional chemistry. The new route commences with the reaction of o-methoxyphenylacetic acid with acetic anhydride in the presence of an organic base, a transformation that cleanly generates the key ketone intermediate, o-methoxypropiophenone. This acylation step avoids the use of nitro compounds entirely, eliminating the associated safety hazards and waste streams. The subsequent conversion of the ketone to the amine is achieved through catalytic hydrogenation using methylamine and a Platinum on Carbon (Pt/C) catalyst. This heterogeneous catalytic system is highly advantageous as it allows for the easy recovery and potential reuse of the precious metal catalyst, drastically reducing the cost per kilogram of the final product.
The operational parameters of this novel approach are designed with industrial scalability in mind, featuring reaction conditions that are both mild and controllable. The hydrogenation step proceeds effectively at moderate temperatures (20-80°C) and hydrogen pressures (0.5-5.0 MPa), which reduces the energy footprint and equipment stress compared to high-pressure alternatives. Furthermore, the final salt formation step utilizes dry HCl gas in common organic solvents, ensuring high crystallinity and ease of isolation. This holistic improvement in the synthetic design translates directly into cost reduction in API manufacturing, as it minimizes raw material consumption, lowers waste treatment expenses, and improves overall throughput. For supply chain stakeholders, this means a more resilient production capability that is less susceptible to regulatory crackdowns on heavy metal waste.
Mechanistic Insights into Acylation and Catalytic Hydrogenation
From a mechanistic perspective, the success of this new synthesis route hinges on the efficiency of the initial acylation and the selectivity of the subsequent reductive amination. In the first step, o-methoxyphenylacetic acid reacts with acetic anhydride, likely proceeding through a mixed anhydride intermediate facilitated by the organic base (such as pyridine or quinoline). This activation enhances the electrophilicity of the carbonyl carbon, promoting an intramolecular or intermolecular acylation that yields the desired ketone with high regioselectivity. The choice of organic base is critical here, as it acts not only as a proton scavenger but also potentially as a nucleophilic catalyst, accelerating the reaction rate without introducing metallic contaminants. This clean mechanism ensures that the crude ketone obtained is of high quality, minimizing the burden on subsequent purification steps and setting the stage for a high-yielding overall process.
The second critical transformation involves the reductive amination of the ketone with methylamine under hydrogen atmosphere. Mechanistically, this involves the initial formation of an imine or iminium ion intermediate between the ketone and the amine, which is then rapidly reduced by hydrogen activated on the surface of the Pt/C catalyst. The use of a supported platinum catalyst provides a large surface area for hydrogen adsorption and activation, facilitating the transfer of hydrogen atoms to the C=N bond with high stereoselectivity and chemoselectivity. Crucially, this catalytic cycle avoids the over-reduction of the aromatic ring or other sensitive functional groups, a common side reaction in less controlled reduction environments. The result is a highly pure amine intermediate, which, upon treatment with HCl, forms the stable hydrochloride salt. This mechanistic clarity allows for precise control over impurity profiles, ensuring that the final high-purity methoxyphenamine hydrochloride meets the rigorous standards required for pharmaceutical applications.
How to Synthesize Methoxyphenamine Hydrochloride Efficiently
Implementing this advanced synthesis protocol requires careful attention to reaction parameters and purification techniques to maximize yield and quality. The process is divided into three distinct stages: the formation of the ketone intermediate, the catalytic hydrogenation to the amine, and the final salt formation. Each step has been optimized in the patent examples to demonstrate robustness across a range of conditions, providing flexibility for process engineers to adapt the method to their specific reactor configurations. The following guide outlines the general workflow derived from the patent data, serving as a foundational reference for technical teams evaluating this route for commercial adoption. Detailed standard operating procedures should be developed based on these principles to ensure safety and consistency.
- React o-methoxyphenylacetic acid with acetic anhydride and an organic base at 40-160°C to form o-methoxypropiophenone.
- Perform catalytic hydrogenation of the ketone intermediate using methylamine and Pt/C catalyst under 0.5-5.0 MPa hydrogen pressure.
- Treat the resulting amine with dry HCl gas in an organic solvent to precipitate and recrystallize the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis method described in Patent CN102267917B offers substantial strategic advantages that go beyond simple unit cost calculations. The primary benefit lies in the drastic simplification of the waste management profile. By eliminating the use of iron powder, ferric chloride, and sodium cyanoborohydride, manufacturers can avoid the generation of heavy metal sludge and toxic cyanide waste. This reduction in hazardous waste significantly lowers the operational costs associated with environmental compliance, wastewater treatment, and disposal fees. In regions with strict environmental regulations, this advantage translates directly into improved margin protection and reduced risk of production shutdowns due to non-compliance, ensuring a more stable and predictable supply of materials.
- Cost Reduction in Manufacturing: The economic benefits of this new route are driven by the substitution of expensive and stoichiometric reagents with catalytic and recyclable alternatives. The replacement of platinum dioxide with Pt/C catalyst allows for the recovery and reuse of the precious metal, significantly lowering the catalyst cost per batch. Furthermore, the use of o-methoxyphenylacetic acid as a starting material is often more cost-effective and stable compared to o-methoxybenzaldehyde and nitroethane, which can be subject to price volatility. The simplified workup procedures, involving standard distillation and crystallization rather than complex extractions to remove metal salts, also reduce labor and utility costs, contributing to a leaner manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable raw materials. Unlike nitroethane, which is regulated and hazardous to transport, acetic anhydride and organic bases are commodity chemicals with robust global supply networks. The robustness of the reaction conditions—operating at moderate temperatures and pressures—also reduces the likelihood of equipment failure or batch failures due to sensitive process windows. This reliability ensures consistent delivery schedules, which is critical for downstream pharmaceutical customers who rely on just-in-time inventory models. The ability to scale this process from pilot to commercial volumes without significant re-engineering further secures the long-term availability of the product.
- Scalability and Environmental Compliance: The pathway described is inherently scalable, making it ideal for the commercial scale-up of complex pharmaceutical intermediates. The heterogeneous nature of the hydrogenation catalyst facilitates easy separation via filtration, a unit operation that scales linearly and efficiently in large reactors. Moreover, the reduction in three wastes (wastewater, waste gas, and solid residue) aligns with green chemistry principles, enhancing the corporate social responsibility profile of the manufacturer. This environmental stewardship is increasingly becoming a prerequisite for qualifying as a vendor for major multinational pharmaceutical companies, thereby opening up new market opportunities and strengthening business relationships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Methoxyphenamine Hydrochloride based on the provided patent data. These insights are derived directly from the experimental examples and background analysis of Patent CN102267917B, aiming to clarify the practical implications of this technology for industry stakeholders. Understanding these details is essential for making informed decisions about process adoption and supplier qualification.
Q: How does the new synthesis method reduce environmental waste compared to traditional routes?
A: The novel process eliminates the use of iron powder and ferric chloride, which traditionally generate massive amounts of acidic sludge and wastewater, significantly lowering environmental compliance costs.
Q: What are the purity specifications achievable with this catalytic hydrogenation route?
A: Experimental data indicates that the final product can achieve HPLC purity levels exceeding 99.5% after standard recrystallization, meeting stringent pharmaceutical grade requirements.
Q: Is the Pt/C catalyst used in this process recoverable for cost efficiency?
A: Yes, the heterogeneous Pt/C catalyst can be recovered via simple suction filtration after the reaction, allowing for potential recycling and reduced catalyst consumption per batch.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methoxyphenamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team has thoroughly analyzed Patent CN102267917B and possesses the expertise to implement this greener, more efficient route for the production of Methoxyphenamine Hydrochloride. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and robust. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of API intermediate we deliver meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners and procurement leaders to collaborate with us to leverage these technological advancements for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can drive down costs, enhance sustainability, and secure a reliable supply of high-quality Methoxyphenamine Hydrochloride for your critical medication formulations.
