Revolutionizing Adefovir Dipivoxil Manufacturing: A Safer, High-Purity Synthetic Route for Global Pharma
The pharmaceutical industry constantly seeks robust manufacturing processes for critical antiviral agents, and the recent advancements detailed in patent CN109438514B represent a significant leap forward in the production of Adefovir Dipivoxil. This nucleotide analog, widely recognized for its efficacy against Hepatitis B virus (HBV), has traditionally been synthesized through routes plagued by hazardous reagents and complex purification steps. The disclosed innovation introduces a streamlined, three-step synthetic pathway that fundamentally alters the economic and safety landscape of producing this essential active pharmaceutical ingredient (API). By replacing expensive and dangerous catalysts with more benign alternatives, this method not only enhances operator safety but also drastically simplifies the downstream processing required to achieve pharmaceutical-grade purity. As a reliable pharmaceutical intermediates supplier, understanding these technical nuances is crucial for evaluating the long-term viability and cost-efficiency of the supply chain. The structural integrity of the final molecule, as depicted below, relies on precise phosphonate esterification and purine coupling, which this new methodology optimizes for industrial scalability.
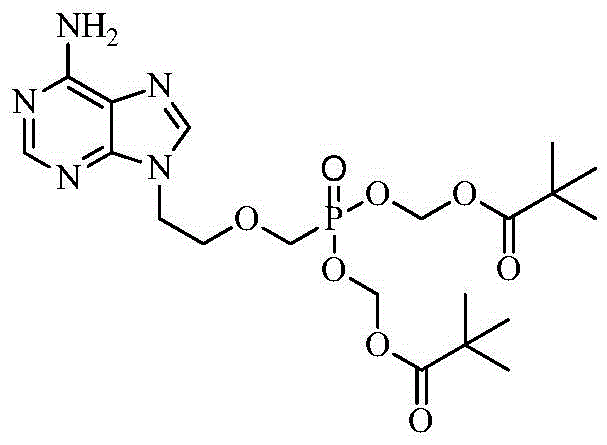
Adefovir Dipivoxil serves as an oral prodrug of adefovir, designed to improve the bioavailability of the active phosphonate species which inhibits HBV DNA polymerase. The chemical architecture involves a phosphonate core linked to an ethoxy side chain and capped with pivaloyloxymethyl groups to facilitate cellular uptake. Achieving this specific configuration with high fidelity is paramount, as impurities can affect both efficacy and toxicity profiles. The patent outlines a preparation method that yields a final product with purity exceeding 99.8%, a benchmark that is exceptionally difficult to reach without resorting to preparative HPLC or extensive column chromatography in conventional syntheses. This level of quality control is essential for meeting the stringent regulatory requirements of global health authorities, ensuring that the high-purity pharmaceutical intermediates delivered to downstream formulators are safe and effective. The ability to produce such high-quality material through a simplified process directly translates to reduced manufacturing costs and a more resilient supply chain for this critical antiviral medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Adefovir Dipivoxil has been hindered by reliance on reagents that are both economically burdensome and operationally hazardous. Traditional routes, such as those described in earlier literature, frequently utilize sodium hydride (NaH) as a condensing agent for the coupling of the phosphonate side chain with the adenine base. Sodium hydride is a pyrophoric solid that requires strict anhydrous conditions and often necessitates cryogenic temperatures (e.g., -50°C) to control exothermic reactions, creating significant safety risks during large-scale production. Furthermore, the hydrolysis of phosphonate esters in prior art typically employs trimethylbromosilane (TMSBr), a reagent that is not only costly but also poses severe challenges regarding transportation, storage, and waste disposal due to its corrosive nature and sensitivity. These conventional pathways often result in complex reaction mixtures that require silica gel column chromatography for purification, a technique that is notoriously difficult to scale industrially and leads to substantial solvent consumption and product loss. The cumulative effect of these factors is a manufacturing process with low total yield, high operational expenditure, and a considerable environmental footprint, making it less attractive for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the innovative process disclosed in the patent data introduces a paradigm shift by substituting these problematic reagents with safer, more cost-effective alternatives without compromising yield or purity. The new methodology replaces the hazardous sodium hydride with anhydrous potassium carbonate (K2CO3), a mild base that allows the coupling reaction to proceed efficiently at moderate temperatures (90-95°C) in dimethylformamide (DMF). This change eliminates the need for cryogenic cooling and significantly reduces the risk of fire or explosion, thereby enhancing the overall safety profile of the manufacturing facility. Additionally, the hydrolysis step is revolutionized by employing a catalytic system comprising trimethylchlorosilane (TMSCl) and potassium iodide (KI) instead of stoichiometric amounts of TMSBr. This catalytic approach not only lowers raw material costs but also simplifies the workup procedure, as the byproducts are easier to manage. Crucially, the entire process is designed to avoid column chromatography entirely, relying instead on optimized crystallization techniques using specific solvent pairs like acetone and isopropyl ether. This strategic modification ensures that the cost reduction in pharmaceutical intermediates manufacturing is realized through both lower input costs and higher throughput, making the process highly suitable for multi-ton production scales.
Mechanistic Insights into TMSCl/KI Catalyzed Hydrolysis and K2CO3 Coupling
The core of this technological breakthrough lies in the mechanistic efficiency of the new catalytic systems employed in the early and middle stages of the synthesis. The hydrolysis of diethyl [(2-chloroethoxy)methyl]phosphonate to the corresponding phosphonic acid is a critical transformation that sets the stage for subsequent esterification. In the novel route, the combination of trimethylchlorosilane and potassium iodide generates reactive silyl iodide species in situ, which act as potent Lewis acids to facilitate the cleavage of the phosphonate ester bonds. This mechanism proceeds smoothly at elevated temperatures (70-75°C), ensuring complete conversion while minimizing the formation of degradation byproducts that often plague acid-catalyzed hydrolyses. The resulting phosphonic acid intermediate is then isolated through a carefully controlled pH adjustment (3.0-3.4), which precipitates the product in high purity, effectively removing inorganic salts and organic impurities before the next step. This precision in pH control is vital for preventing the co-precipitation of impurities that could carry through to the final API, demonstrating a deep understanding of the physicochemical properties of the intermediates involved.
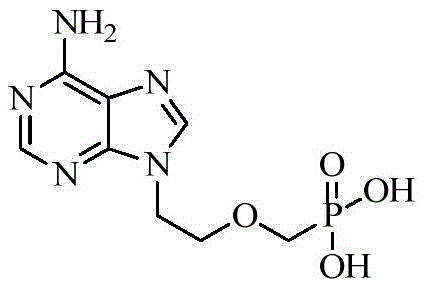
Following the formation of the phosphonic acid, the subsequent esterification and coupling steps are equally optimized for industrial feasibility. The reaction of the phosphonic acid with chloromethyl pivalate is conducted in N-methylpyrrolidone (NMP) using triethylamine as a base, conditions that favor the formation of the bis(pivaloyloxymethyl) ester with high selectivity. The final coupling with adenine utilizes anhydrous potassium carbonate, which acts as a non-nucleophilic base to deprotonate the purine nitrogen, facilitating nucleophilic attack on the chloromethyl group of the phosphonate ester. This reaction avoids the aggressive conditions associated with sodium hydride, thereby preserving the integrity of the sensitive purine ring and preventing side reactions such as alkylation at unwanted positions. The overall reaction pathway, illustrated below, showcases a logical progression where each step is designed to maximize yield and minimize purification complexity. By eliminating the need for transition metal catalysts or hazardous strong bases, the process inherently reduces the burden on the quality control laboratory, as there are fewer heavy metal residues or dangerous byproducts to monitor and remove.
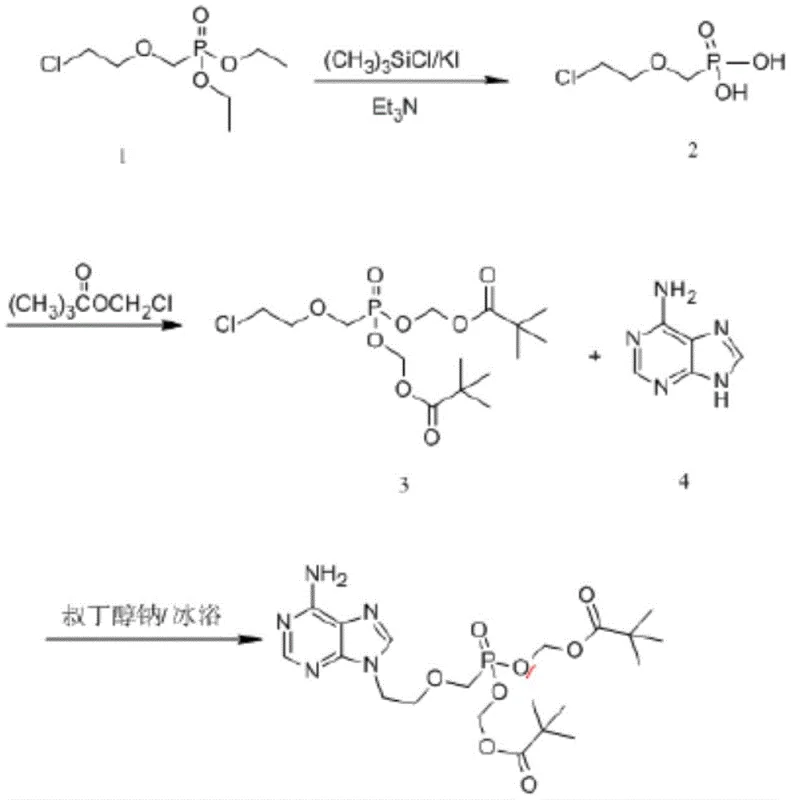
How to Synthesize Adefovir Dipivoxil Efficiently
The synthesis of Adefovir Dipivoxil via this patented route involves a sequence of highly controlled chemical transformations designed for reproducibility and scale. The process begins with the catalytic hydrolysis of the diethyl phosphonate precursor, followed by esterification with chloromethyl pivalate, and concludes with the coupling of the resulting phosphonate ester with adenine. Each stage incorporates specific solvent systems and temperature profiles that are critical for achieving the reported high yields and purity levels. For instance, the final purification utilizes a dual-solvent crystallization strategy involving acetone and isopropyl ether, which effectively strips away residual starting materials and side products. To ensure consistent results in a GMP environment, strict adherence to the molar ratios and reaction times specified in the patent is essential. The detailed standardized synthesis steps, including exact quantities and equipment specifications, are outlined in the guide below for technical reference.
- Hydrolyze diethyl [(2-chloroethoxy)methyl]phosphonate using a trimethylchlorosilane and potassium iodide catalyst system to obtain the phosphonic acid intermediate.
- React the resulting phosphonic acid with chloromethyl pivalate in N-methylpyrrolidone (NMP) using triethylamine to form the bis(pivaloyloxymethyl) ester.
- Couple the ester intermediate with 6-amino-9H-purine (adenine) using anhydrous potassium carbonate in DMF, followed by a dual-solvent crystallization purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling advantages that extend beyond mere technical elegance. The primary benefit is a substantial reduction in the cost of goods sold (COGS), driven by the replacement of expensive reagents like trimethylbromosilane and sodium hydride with commodity chemicals such as trimethylchlorosilane and potassium carbonate. This shift not only lowers the direct material costs but also reduces the overhead associated with the special handling, storage, and disposal of hazardous materials. Furthermore, the elimination of column chromatography significantly shortens the production cycle time, allowing for faster turnover of batches and improved responsiveness to market demand. The simplified post-treatment procedures, which rely on filtration and crystallization rather than complex separations, also reduce the consumption of solvents and energy, contributing to a more sustainable and environmentally compliant manufacturing operation. These factors collectively enhance the reliability of the supply chain, ensuring a steady flow of high-purity pharmaceutical intermediates to meet global healthcare needs.
- Cost Reduction in Manufacturing: The substitution of high-cost reagents with economical alternatives creates a direct positive impact on the bottom line. By utilizing trimethylchlorosilane and potassium iodide instead of trimethylbromosilane, the process avoids the premium pricing associated with bromine-based silylating agents. Additionally, the use of potassium carbonate in place of sodium hydride removes the need for specialized inert atmosphere equipment and cryogenic cooling systems, leading to significant savings in capital expenditure and utility costs. The avoidance of silica gel column chromatography further reduces costs by eliminating the expense of chromatography media and the large volumes of solvents required for elution, resulting in a leaner and more cost-efficient production model.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade raw materials mitigates the risk of supply disruptions that can occur with specialty reagents. Chemicals like potassium carbonate, triethylamine, and chloromethyl pivalate are produced in large volumes globally, ensuring a stable supply even during periods of market volatility. Moreover, the robustness of the reaction conditions, which tolerate moderate temperatures and standard atmospheric pressure, reduces the likelihood of batch failures due to equipment malfunction or operator error. This operational stability translates into predictable lead times and consistent delivery schedules, which are critical metrics for supply chain planners managing the inventory of life-saving antiviral medications.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with unit operations such as crystallization and filtration being easily transferable from pilot plant to commercial production scales. The reduction in hazardous waste generation, particularly the absence of sodium hydride residues and bromine-containing byproducts, simplifies waste treatment and lowers compliance costs associated with environmental regulations. The high atom economy of the coupling reactions and the efficient recovery of solvents like DMF and acetone further minimize the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to eco-conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing facilities or for procurement teams assessing the quality and cost implications of sourcing intermediates produced via this method.
Q: Why is the TMSCl/KI system preferred over traditional TMSBr for hydrolysis?
A: The patent highlights that trimethylbromosilane (TMSBr) is expensive and poses significant transportation and storage hazards. The novel trimethylchlorosilane (TMSCl) and potassium iodide system offers a cost-effective alternative with higher industrial safety profiles while maintaining efficient hydrolysis rates.
Q: How does this method achieve high purity without column chromatography?
A: The process utilizes a optimized pH control (3.0-3.4) during crystallization and a specific dual-solvent refinement system involving acetone, water, and isopropyl ether. This eliminates the need for silica gel column chromatography, which is difficult to scale, allowing for purity levels exceeding 99.8% through simple filtration and washing.
Q: What are the safety advantages of using Potassium Carbonate over Sodium Hydride?
A: Traditional routes often employ sodium hydride (NaH), which requires harsh conditions (e.g., -50°C) and presents severe safety risks during scale-up due to its pyrophoric nature. The new method uses anhydrous potassium carbonate under mild heating (90-95°C), significantly reducing operational risk and simplifying post-treatment procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adefovir Dipivoxil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe manufacturing processes for antiviral APIs like Adefovir Dipivoxil. Our team of expert chemists has thoroughly analyzed the patented route described in CN109438514B and possesses the technical capability to implement these advanced methodologies at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and compliant with international GMP standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Adefovir Dipivoxil meets the highest quality benchmarks required by global regulatory agencies. We are committed to leveraging our expertise in phosphonate chemistry to deliver superior products that support the global fight against Hepatitis B.
We invite potential partners to engage with us to explore how this optimized synthesis route can benefit their specific supply chain requirements. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to this safer, more efficient process. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. By partnering with us, you gain access to a reliable source of high-quality pharmaceutical intermediates backed by a commitment to innovation, safety, and customer success.
