Advanced Manufacturing of Adefovir Dipivoxil: Enhancing Yield and Purity for Global Supply Chains
The global demand for effective antiviral therapies continues to drive innovation in the manufacturing of nucleotide analogues, particularly for the treatment of chronic hepatitis B. Patent CN101891767B presents a significant technological advancement in the preparation of Adefovir Dipivoxil, a potent prodrug that offers superior bioavailability compared to its parent compound. This proprietary method addresses critical bottlenecks in traditional synthesis routes by introducing a novel protective strategy that mitigates oxidative degradation during the esterification process. By integrating specific antioxidant agents into the reaction matrix, the process not only enhances the chemical stability of the sensitive amino functionalities but also streamlines the downstream purification workflow. For pharmaceutical manufacturers seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this patented approach is essential for securing a competitive edge in the antiviral market. The structural integrity of the final molecule, as depicted below, relies heavily on the precise control of reaction conditions to prevent the formation of colored impurities that often plague bulk production.
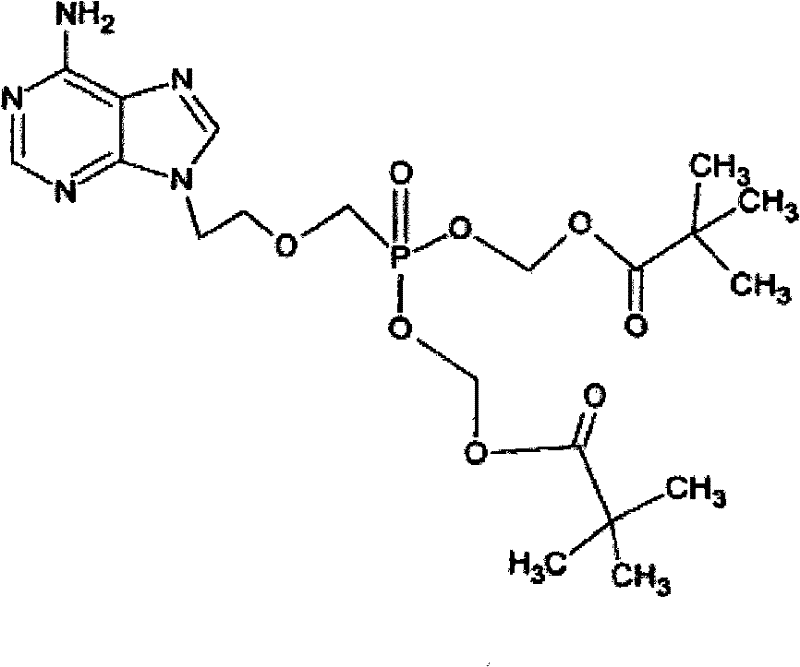
Furthermore, the economic implications of this synthesis route cannot be overstated, as it directly targets the reduction of production costs while simultaneously elevating product quality. The method described leverages readily available reagents and standard unit operations, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates. By minimizing side reactions that typically consume raw materials and complicate waste management, this technology aligns perfectly with the modern industry's push towards greener and more efficient manufacturing practices. As we delve deeper into the technical specifics, it becomes clear that this patent represents a robust solution for companies aiming to optimize their supply chain for high-value antiviral drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for Adefovir Dipivoxil have historically struggled with inherent chemical instabilities associated with the adenine moiety and the phosphonate ester linkage. In standard protocols, the reaction between Adefovir and chloromethyl pivalate is often conducted under conditions that inadvertently promote the oxidation of the primary amino group located on the purine ring. This oxidative degradation manifests visibly as the development of red or yellow hues in the reaction crude, serving as a stark indicator of significant by-product formation. Consequently, these conventional methods suffer from suboptimal reaction conversion rates, typically hovering around 45%, which necessitates extensive and costly purification efforts to meet stringent pharmacopeial standards. The presence of these oxidized impurities not only lowers the overall yield but also complicates the crystallization process, leading to inconsistent batch-to-batch quality and increased material loss during filtration and washing steps.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally alters the reaction environment by introducing a specialized protective agent system that acts as a sacrificial antioxidant. By incorporating compounds such as 2,6-di-tert-butyl-4-methylphenol, sodium sulfite, or vitamin C directly into the reaction mixture, the process effectively scavenges reactive oxygen species before they can attack the sensitive amino groups. This strategic intervention allows the esterification to proceed at elevated temperatures, specifically up to 60°C, without the detrimental side effects observed in prior art. The result is a dramatic improvement in reaction efficiency, with yields climbing to approximately 52%, representing a substantial gain in material throughput. Moreover, the resulting crude product exhibits significantly less coloration, indicating a cleaner reaction profile that simplifies the subsequent decolorization and chromatographic purification stages, thereby reducing the overall processing time and solvent consumption.
Mechanistic Insights into Antioxidant-Protected Esterification
The core chemical mechanism driving this improvement lies in the preservation of the nucleophilic character of the adenine ring during the alkylation of the phosphonate hydroxyl groups. Under normal thermal conditions, the electron-rich amino group is prone to single-electron transfer processes that generate radical species, leading to polymerization or oxidation products that are difficult to separate. The added protective agents function by maintaining a reducing environment within the reaction vessel, effectively suppressing these radical pathways. For instance, when using 2,6-di-tert-butyl-4-methylphenol, the phenolic hydrogen is readily donated to neutralize free radicals, stabilizing the transition state of the esterification reaction. This ensures that the chloromethyl pivalate reacts selectively with the phosphonic acid moieties rather than engaging in non-specific side reactions with the heterocyclic base. The reaction scheme below illustrates the transformation where the protective agent facilitates the formation of the bis-ester without compromising the integrity of the adenine scaffold.
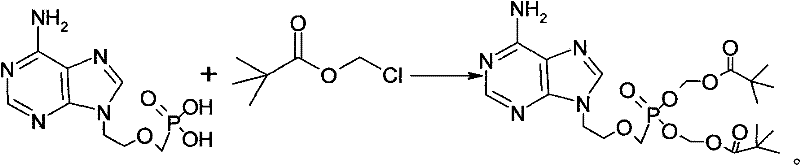
Impurity control is further enhanced through a rigorous two-stage purification protocol that leverages the differential solubility of the target compound versus its precursors and by-products. Following the initial reaction, the mixture is quenched and subjected to a liquid-liquid extraction using isopropyl acetate and water, which removes water-soluble salts and unreacted polar impurities. The organic phase is then concentrated and loaded onto a silica gel column, utilizing a precise gradient of methanol and dichloromethane (1:19 ratio) to elute the product. This specific solvent system is critical for resolving the Adefovir Dipivoxil from the mono-ester intermediate and any remaining starting material. A subsequent decolorization step using activated carbon adsorbs any trace colored organics, followed by a second column chromatography pass to ensure the final active pharmaceutical ingredient meets the highest purity specifications required for clinical applications.
How to Synthesize Adefovir Dipivoxil Efficiently
The operational execution of this synthesis requires strict adherence to the specified parameters regarding temperature control and reagent stoichiometry to maximize the benefits of the protective agent strategy. The process begins with the dissolution of the starting material and antioxidant in a polar aprotic solvent under an inert nitrogen atmosphere to exclude atmospheric oxygen. Precise temperature management is vital, with the initial mixing occurring at low temperatures (0-10°C) to control exotherms before a controlled ramp to 60°C drives the reaction to completion. Detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are outlined in the technical guide below, ensuring reproducibility and safety across different manufacturing sites.
- Under nitrogen protection, dissolve Adefovir and a protective agent (e.g., 2,6-di-tert-butyl-4-methylphenol) in N-methylpyrrolidone, then cool to 0-10°C.
- Add triethylamine and chloromethyl pivalate dropwise, then slowly raise the temperature to 60°C until the reaction is complete.
- Filter the mixture, wash the mother liquor, dry, concentrate, and purify the crude product via silica gel column chromatography and activated carbon decolorization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere chemical yield improvements. The integration of cost-effective antioxidants eliminates the need for expensive protecting group strategies, such as benzyloxycarbonyl protection and subsequent hydrogenation, which are capital-intensive and time-consuming. This simplification of the synthetic route translates directly into a reduction in production costs, estimated to be around 20% according to the patent data, providing a significant margin advantage in a competitive generic drug market. Furthermore, the use of common industrial solvents like N-methylpyrrolidone and isopropyl acetate ensures that raw material sourcing remains stable and unaffected by niche supply constraints, enhancing the overall resilience of the supply chain against market volatility.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection sequences significantly lowers the cost of goods sold by reducing both reagent expenses and processing time. By avoiding the use of precious metal catalysts often required for deprotection steps, the process also removes the financial burden associated with catalyst recovery and heavy metal testing, leading to substantial cost savings in quality control and waste disposal.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as triethylamine and chloromethyl pivalate ensures a robust supply base with multiple qualified vendors globally. This diversification of the raw material portfolio minimizes the risk of production stoppages due to single-source dependencies, allowing for consistent delivery schedules and improved inventory management for downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The streamlined workup procedure, which avoids hazardous reagents and generates less toxic waste, facilitates easier regulatory approval and environmental compliance. The process is inherently scalable, allowing for seamless transition from kilogram-scale development batches to multi-ton commercial production without the need for specialized equipment, thereby reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational advantages and chemical rationale behind the use of protective agents in large-scale manufacturing. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own production lines.
Q: How does the addition of protective agents improve the synthesis of Adefovir Dipivoxil?
A: The amino functional groups in Adefovir and its ester are susceptible to oxidation at high temperatures, leading to colored by-products and reduced yield. Adding antioxidants like 2,6-di-tert-butyl-4-methylphenol protects these groups, increasing yield from 45% to approximately 52%.
Q: What solvents are recommended for this optimized preparation method?
A: The patent specifies the use of polar aprotic solvents such as N-methylpyrrolidone (NMP) or N-formyl dimethylamide to ensure effective dissolution and reaction kinetics during the esterification process.
Q: What purification steps are critical for achieving high purity in the final product?
A: The process involves a dual purification strategy: initial silica gel column chromatography using a methanol and dichloromethane system (1:19), followed by activated carbon decolorization and a second column chromatography step to remove trace impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adefovir Dipivoxil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of process robustness and purity in the production of antiviral intermediates like Adefovir Dipivoxil. Our team of expert chemists has extensively analyzed the methodologies described in patent CN101891767B and possesses the technical capability to implement these advanced protective strategies at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for global pharmaceutical registration.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project needs. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how our efficient synthesis routes can lower your overall procurement costs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and accelerate your time to market.
