Optimizing Decitabine Manufacturing: A Technical Breakthrough in Chiral Separation and Purification
Optimizing Decitabine Manufacturing: A Technical Breakthrough in Chiral Separation and Purification
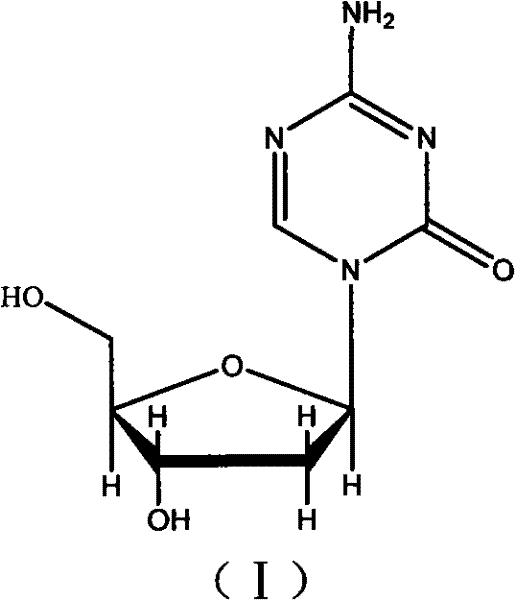
The pharmaceutical industry continuously seeks robust synthetic routes for potent oncology agents, and Decitabine stands out as a critical DNA methyltransferase inhibitor used in the treatment of myelodysplastic syndromes. The patent CN101899079A introduces a refined preparation, separation, and purification method that addresses historical bottlenecks in nucleoside analog synthesis. By utilizing 2'-Deoxy-D-ribose and 5-Azacytosine as foundational building blocks, this technical scheme orchestrates a sequence of methylation, acylation, trimethyl silylation, condensation, and deprotection reactions. The strategic innovation lies not just in the coupling efficiency but in the implementation of a simple yet highly effective chiral separation protocol. This approach ensures the final product achieves a chemical purity above 99.8% and an optical purity exceeding 99.68%, drastically lowering production costs while ensuring suitability for large-scale industrialized production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Decitabine has been plagued by significant technical hurdles that impede commercial viability and supply chain stability. Early literature, such as reports from Nucl. Acid Res. in 1978, described condensation reactions under tin tetrachloride catalysis which often resulted in complex mixtures requiring arduous purification. Furthermore, subsequent methods disclosed in Chinese Journal of Pharmaceuticals utilized methoxyacetic anhydride for hydroxyl protection; however, this specific protecting group creates a formidable challenge during the deprotection phase, rendering the process wayward and difficult to control on a large scale. A critical failure in many prior art methods is the lack of a defined strategy for separating the alpha and beta isomers. Since Decitabine is specifically the beta-configuration, the inability to efficiently split the racemic or anomeric mixture results in low yields of the active pharmaceutical ingredient and necessitates expensive downstream processing that erodes profit margins.
The Novel Approach
The methodology presented in patent CN101899079A represents a paradigm shift by replacing problematic protecting groups with more labile alternatives that facilitate smoother deprotection. Instead of methoxyacetyl groups, this novel route employs stable blocking groups such as p-methoxybenzoyl, p-nitrobenzoyl, or benzoyl moieties, which are easier to remove under mild basic conditions. The synthetic pathway is meticulously designed to improve the yield of the key coupling step by utilizing protected ribodesose acylates reacting with activated 5-azacytosine. This structural modification not only enhances the stereochemical outcome but also integrates a detailed disclosing of separation and purification for the finished product. By overcoming the defects of prior art where fractionation was often omitted or ineffective, this approach guarantees the isolation of the highly purified beta-isomer essential for therapeutic efficacy.
Mechanistic Insights into Protecting Group Strategy and Coupling
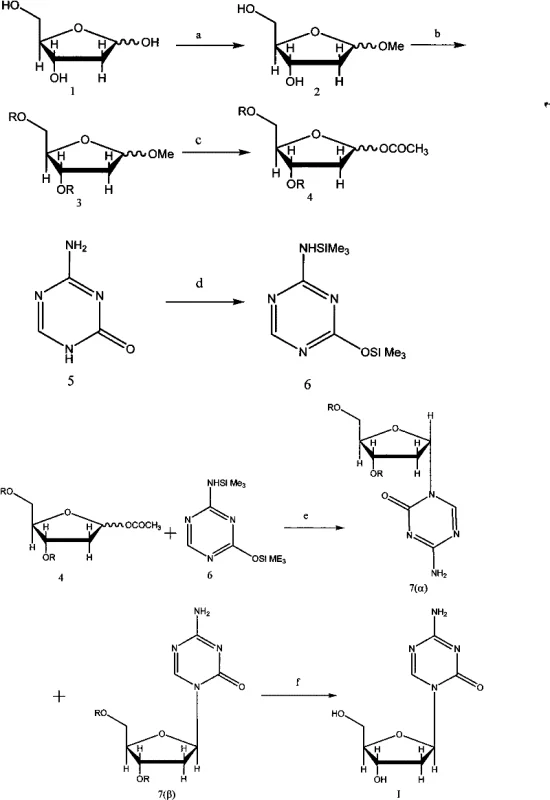
The core of this synthetic success lies in the precise manipulation of the sugar moiety's reactivity through strategic protection. In Step b, the hydroxyl groups of the methylated ribose intermediate are protected using specific acyl chlorides where R can be p-methoxybenzoyl, p-nitrobenzoyl, or benzoyl. This selection is chemically profound because these aromatic acyl groups provide sufficient stability to withstand the subsequent acidic acylation conditions in Step c, where acetic anhydride and sulfuric acid are used to activate the anomeric position. The electronic properties of the R group, particularly the electron-donating methoxy or electron-withdrawing nitro substituents, can be tuned to optimize the solubility and crystallization behavior of the intermediates, which is a critical factor often overlooked in standard nucleoside synthesis protocols.
Furthermore, the activation of the nucleobase in Step d via silylation with hexamethyldisilazane (HMDS) and ammonium sulfate generates a highly nucleophilic silicon ether species. This activated 5-azacytosine is then coupled with the acylated sugar in Step e using anhydrous stannic chloride as a Lewis acid catalyst. The mechanism proceeds through the formation of an oxocarbenium ion intermediate, which is attacked by the silylated base. The use of SnCl4 at controlled temperatures between -20°C and 40°C ensures that the reaction kinetics favor the formation of the desired N-glycosidic bond while minimizing degradation of the sensitive triazine ring. The subsequent deprotection in Step f using sodium methylate in anhydrous methanol cleanly removes the acyl groups without damaging the glycosidic linkage, a testament to the robustness of the chosen protecting group strategy.

How to Synthesize Decitabine Efficiently
The synthesis of Decitabine via this patented route requires strict adherence to anhydrous conditions and precise temperature control to maximize the yield of the beta-anomer. The process begins with the methylation of 2'-deoxy-D-ribose, followed by the critical protection step using the selected acyl chloride. Detailed operational parameters, such as the specific molar ratios of intermediates and the duration of reflux during silylation, are essential for reproducibility. For laboratory and pilot plant chemists aiming to replicate this high-purity output, understanding the nuances of the workup procedures—particularly the extraction and drying steps—is vital. The detailed standardized synthesis steps见下方的指南 provide a comprehensive roadmap for executing this complex transformation safely and effectively.
- Methylation of 2'-Deoxy-D-ribose using HCl in methanol to form the methyl glycoside intermediate.
- Protection of hydroxyl groups using specific acyl chlorides (e.g., p-methoxybenzoyl chloride) followed by acetylation.
- Activation of 5-Azacytosine via silylation with HMDS, followed by Lewis acid-catalyzed coupling and final deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this optimized synthetic route offers tangible benefits that extend beyond mere technical elegance. The primary advantage is the utilization of cheap and readily available chemical products as raw materials, specifically 2'-Deoxy-D-ribose and 5-Azacytosine, which mitigates the risk of supply disruptions associated with exotic or proprietary starting materials. By adopting a process that relies on common reagents like methanol, acetic anhydride, and standard acyl chlorides, manufacturers can secure a more resilient supply chain that is less susceptible to geopolitical fluctuations or single-source vendor bottlenecks. This accessibility of inputs directly translates to a more predictable procurement cycle and enhanced negotiation leverage with upstream chemical suppliers.
- Cost Reduction in Manufacturing: The elimination of difficult deprotection steps associated with methoxyacetyl groups significantly reduces the consumption of specialized reagents and energy. In traditional methods, the stubborn removal of certain protecting groups often requires harsh conditions or multiple cycles, driving up utility costs and waste disposal fees. By switching to benzoyl-type protecting groups that are easier to cleave, the process streamlines the final stages of production. This simplification means fewer unit operations are required, leading to substantial cost savings in labor, solvent usage, and reactor time. Furthermore, the higher overall yield resulting from improved coupling efficiency means less raw material is wasted per kilogram of final API produced.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures consistent batch-to-batch quality, which is paramount for maintaining regulatory compliance and avoiding costly production delays. The patent explicitly details a chiral separation method that reliably produces the active beta-isomer, removing the uncertainty often found in nucleoside synthesis where anomeric mixtures can stall inventory release. Because the reaction conditions are mild and the equipment requirements are simple, the technology is easily transferable between different manufacturing sites. This flexibility allows supply chain heads to diversify their manufacturing footprint, reducing the risk of total supply failure due to localized incidents or maintenance shutdowns at a single facility.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is designed for industrialized production with a focus on minimizing hazardous waste. The use of standard solvents like methanol and chloroform, which have well-established recovery and recycling protocols, facilitates better environmental management compared to processes requiring exotic or highly toxic solvents. The high purity of the crude product (>99.8%) reduces the burden on downstream purification units, thereby lowering the volume of mother liquor and solid waste generated. This efficiency supports sustainable manufacturing goals and helps companies meet increasingly stringent environmental regulations without compromising on output volume or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Decitabine synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing long-term value. The answers reflect the specific improvements in purity and yield that distinguish this method from legacy technologies.
Q: How does this patent address the difficulty of separating alpha and beta isomers in Decitabine synthesis?
A: The patent utilizes a specific chiral separation and purification method involving repeated recrystallization in anhydrous methanol with activated carbon treatment, effectively isolating the biologically active beta-isomer from the alpha-anomer.
Q: What are the advantages of using p-methoxybenzoyl or p-nitrobenzoyl protecting groups over methoxyacetyl groups?
A: Unlike methoxyacetyl groups which create difficult deprotection conditions, the benzoyl-type protecting groups selected in this process are stable during reaction but allow for milder and more efficient removal during the final deprotection step, significantly simplifying the workflow.
Q: What is the achieved chemical and optical purity of the final Decitabine product?
A: The described method consistently yields Decitabine with a chemical purity exceeding 99.8% and an optical purity greater than 99.68%, meeting stringent requirements for oncology drug applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs like Decitabine depends on a partner who can bridge the gap between laboratory innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity specifications demonstrated in patent CN101899079A are maintained at every ton of output. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify both chemical and optical purity, guaranteeing that every batch meets the stringent standards required for global pharmaceutical markets. Our commitment to quality assurance means that you receive a reliable Decitabine supplier partnership backed by data-driven process control.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project's unique requirements. Let us collaborate to enhance your supply chain resilience and deliver high-quality Decitabine to patients who need it most.
