Advanced Stereoselective Manufacturing of Lamivudine Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antiviral agents, particularly for HIV and Hepatitis B treatments where stereochemical purity is paramount for efficacy and safety. Patent CN101307048A introduces a groundbreaking stereoselective preparation method for Lamivudine, a potent deoxycytidine analogue known chemically as (2R-cis)-4-amino-1-(2-hydroxymethyl-1,3-oxathiolan-5-yl)-1H-pyrimidin-2-one. This innovation addresses the longstanding challenge of controlling the two chiral centers inherent in the oxathiolane ring structure, specifically targeting the biologically active 2R, 5S configuration. By leveraging a chiral auxiliary strategy prior to the glycosylation step, the disclosed technology transforms a complex separation problem into a manageable crystallization process. This shift not only enhances the optical purity of the final active pharmaceutical ingredient (API) but also streamlines the production workflow, making it an attractive solution for reliable API intermediate suppliers aiming to optimize their manufacturing portfolios.
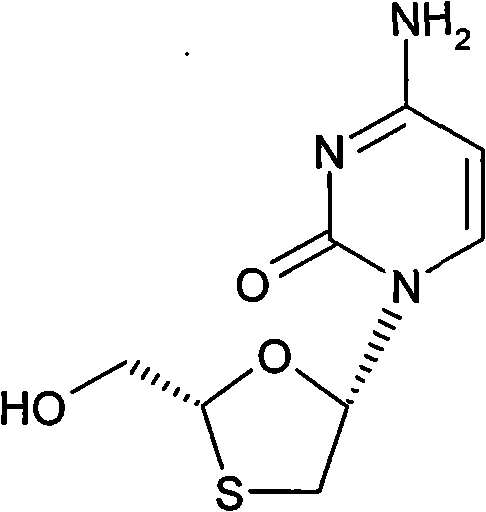
Lamivudine possesses two chiral centers, theoretically allowing for four stereoisomers, yet only the 2R, 5S isomer exhibits the strongest anti-HIV and anti-HBV activity with minimal cytotoxicity. Conventional synthesis routes often struggle to differentiate these isomers effectively, leading to costly purification bottlenecks. The method described in CN101307048A overcomes these hurdles by fixing the configuration at the 2-position of the sugar moiety before coupling with the nucleobase. This strategic foresight ensures that the subsequent glycosylation reaction yields diastereomers rather than enantiomers. Since diastereomers possess different physical properties, they can be separated through standard crystallization techniques rather than expensive and labor-intensive chromatography. This fundamental change in process design represents a significant leap forward in the cost reduction in pharmaceutical intermediates manufacturing, offering a pathway that is both economically superior and operationally simpler for large-scale facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
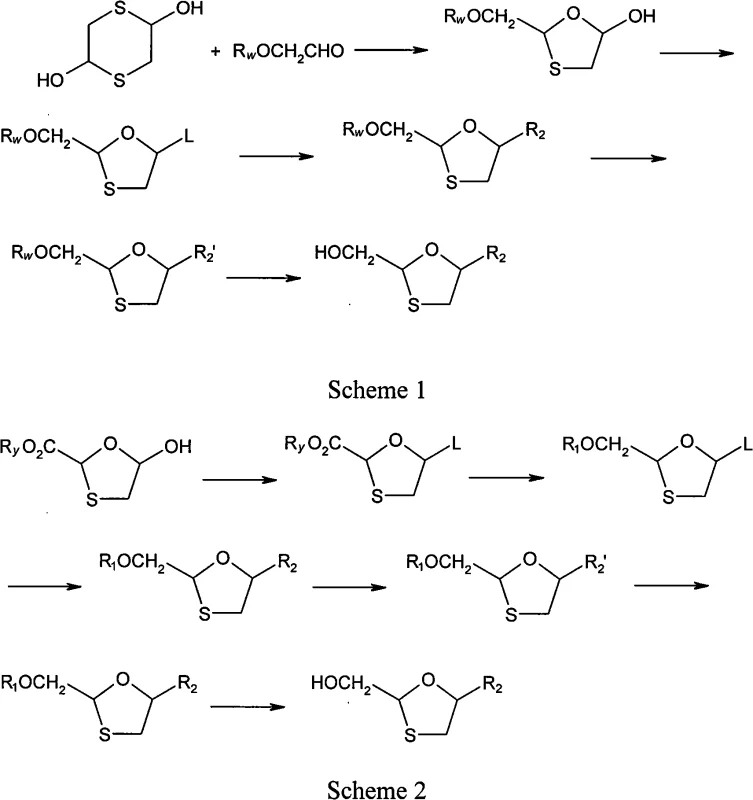
Prior art technologies, such as those referenced in WO94/14802, typically rely on non-stereoselective glycosylation reactions that generate a mixture of all possible stereoisomers. As illustrated in the reaction schemes below, these traditional pathways lack intrinsic chirality control, resulting in a crude product that contains the desired active isomer alongside its inactive or potentially toxic counterparts. To isolate the therapeutically relevant 2R, 5S-Lamivudine from this complex mixture, manufacturers are forced to employ column chromatography. This purification technique is notoriously inefficient for industrial applications due to its high consumption of solvents and silica gel, low throughput, and stringent equipment requirements. Furthermore, the low overall yield associated with recovering a single enantiomer from a racemic or near-racemic mixture drastically inflates production costs and creates significant waste disposal challenges. These operational inefficiencies make conventional routes wayward and unsuitable for the rigorous demands of modern commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to the limitations of the past, the novel approach detailed in this patent introduces a chiral auxiliary at the very beginning of the synthesis sequence. By reacting a glycerol derivative with L-menthol chloroformate, the process establishes a defined stereochemical environment at the C-2 position of the future oxathiolane ring. This pre-determination of chirality means that when the glycosylation reaction eventually occurs with cytosine or protected cytosine, the resulting products are diastereomers. The patent explicitly highlights that separating diastereomers is significantly less difficult than separating enantiomers, often requiring nothing more than a simple crystallization step in an alcoholic solvent like ethanol. This eliminates the need for chromatographic purification entirely. The result is a streamlined process where high-optical-purity intermediates are obtained through crystallization, followed by a straightforward deprotection step to yield the final Lamivudine. This methodology not only improves the purity profile but also drastically simplifies the operational workflow, making it highly convenient to operate and perfectly suited for industrialized production environments.
Mechanistic Insights into Chiral Auxiliary-Controlled Glycosylation
The core of this technological advancement lies in the precise application of chiral auxiliaries to dictate stereochemical outcomes. The preferred embodiment utilizes L-menthol as the chiral source, which is converted into L-menthol chloroformate. This reagent reacts with a protected glycerol derivative to form a chiral carbonate ester. Subsequent steps involve hydrolysis, periodate oxidation to generate an aldehyde, and condensation with 1,4-dihydroxy-2,5-dithiane to form the oxathiolane ring system. Crucially, the chirality established by the menthol group influences the formation of the ring, ensuring that the 2-position maintains the desired R-configuration. When this chiral intermediate is activated (typically by converting the hydroxyl group to a leaving group like chloride or acetate) and reacted with silylated cytosine, the existing chiral center directs the approach of the nucleophile. While the reaction may still produce a mixture of alpha and beta anomers at the 1-position (creating diastereomers), the fixed configuration at the 2-position prevents the formation of the unwanted 2S enantiomers. This mechanistic control is the key to achieving high stereoselectivity without resorting to resolution techniques post-synthesis.
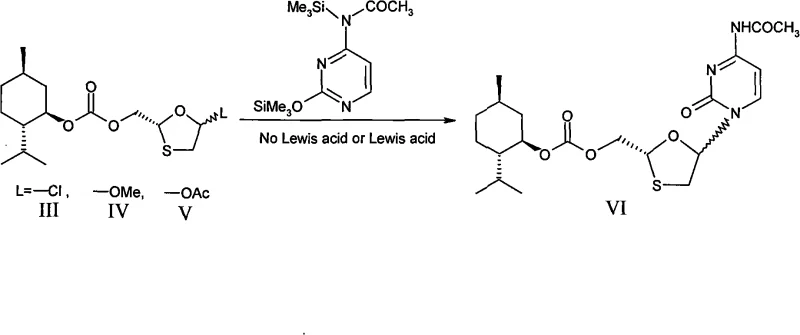
Impurity control in this process is inherently built into the chemical design. Because the unwanted stereoisomers are diastereomers of the desired product, they exhibit distinct solubility profiles in various solvents. The patent describes using ethanol for recrystallization, a step that effectively purges the reaction mixture of the minor diastereomer. For instance, in the preferred examples, the ratio of alpha to beta bodies in the crude mixture might be around 1:1.5, but after recrystallization, the desired isomer is isolated with high purity. Furthermore, the final deprotection step involves mild basic hydrolysis using potassium carbonate, which removes the acetyl protecting group from the cytosine base and the menthol carbonate from the sugar moiety without epimerizing the sensitive chiral centers. The use of p-nitrobenzoic acid for salt formation further aids in purification, as the salt of the desired isomer precipitates selectively from the aqueous layer. This multi-layered approach to impurity management ensures that the final product content can reach above 99%, with enantiomeric impurities kept below 0.2%, meeting the stringent specifications required by pharmacopeias like USP29.
How to Synthesize Lamivudine Efficiently
The synthesis of high-purity Lamivudine via this stereoselective route involves a sequence of well-defined chemical transformations that prioritize yield and optical purity. The process begins with the preparation of the chiral auxiliary reagent, followed by the construction of the oxathiolane ring, glycosylation with a protected cytosine base, and final deprotection. Each step is optimized to minimize side reactions and maximize the recovery of the desired stereoisomer. The detailed standardized synthesis steps provided below outline the specific conditions, reagents, and workup procedures necessary to replicate this high-efficiency pathway in a laboratory or pilot plant setting.
- Prepare the chiral auxiliary reagent by reacting L-menthol with triphosgene to form L-menthol chloroformate, ensuring strict temperature control during the addition of pyridine.
- Synthesize the key oxathiolane intermediate by condensing the chiral carbonate with 1,4-dihydroxy-2,5-dithiane, followed by oxidation and chlorination to introduce the leaving group.
- Perform the glycosylation reaction with silylated cytosine derivatives, separate the resulting diastereomers via crystallization, and finally remove the protecting groups to yield Lamivudine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this stereoselective synthesis method offers transformative benefits that extend far beyond simple chemical yield improvements. The primary advantage lies in the drastic simplification of the purification train. By replacing column chromatography with crystallization, the process eliminates a major bottleneck that typically constrains throughput in API manufacturing. Crystallization is a unit operation that scales linearly and predictably, whereas chromatography often faces diminishing returns and exponential cost increases as volume grows. This shift allows for continuous or large-batch processing, significantly enhancing supply chain reliability and reducing the risk of production delays caused by purification capacity limits. Moreover, the reduction in solvent usage and solid waste (silica gel) aligns with increasingly strict environmental regulations, lowering the total cost of ownership for waste treatment and compliance.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification represents a massive opportunity for cost optimization. Chromatography requires expensive stationary phases, vast quantities of high-purity solvents, and specialized equipment, all of which contribute heavily to the cost of goods sold (COGS). By utilizing a chiral auxiliary that enables crystallization-based separation, the process removes these variable costs entirely. Additionally, the use of L-menthol, a commodity chemical derived from natural sources, ensures that the cost of the chiral controller remains low and stable, avoiding the price volatility often associated with synthetic chiral ligands. The overall result is a manufacturing process that is inherently more economical, allowing for substantial cost savings that can be passed down the supply chain or reinvested into further process optimization.
- Enhanced Supply Chain Reliability: Supply continuity is critical for life-saving medications like Lamivudine. This method enhances reliability by relying on robust, scalable chemistry. The reagents involved, such as triphosgene, L-menthol, and cytosine, are commercially available from multiple global suppliers, reducing the risk of raw material shortages. The simplicity of the operation—primarily involving stirring, heating, and filtration—means that the process can be easily transferred between manufacturing sites or scaled up from pilot to commercial production with minimal technical risk. This flexibility ensures that manufacturers can respond quickly to surges in demand without compromising on quality or lead times, providing a secure and dependable source of high-purity pharmaceutical intermediates for downstream formulation.
- Scalability and Environmental Compliance: Scaling a chemical process often introduces new challenges regarding heat transfer, mixing, and safety. However, the unit operations in this patent (esterification, oxidation, condensation, crystallization) are standard in the fine chemical industry and are well-understood at the multi-ton scale. The absence of chromatography simplifies the equipment footprint, requiring fewer reactors and less floor space. From an environmental perspective, the reduction in organic solvent waste and the avoidance of silica gel disposal significantly lower the ecological impact of the manufacturing process. This aligns with green chemistry principles and facilitates easier regulatory approval in jurisdictions with strict environmental standards. The process is designed for suitability for industrialized production, ensuring that it can meet the rigorous demands of global markets while maintaining a sustainable operational profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the stereoselective synthesis of Lamivudine. These answers are derived directly from the experimental data and technical disclosures within patent CN101307048A, providing clarity on the mechanism, purity, and scalability of the method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial manufacturing.
Q: How does this method improve stereoselectivity compared to conventional routes?
A: Unlike prior art methods that produce racemic mixtures requiring difficult chromatographic separation, this patent utilizes a chiral auxiliary (L-menthol) to pre-determine the configuration at the C-2 position. This results in the formation of diastereomers rather than enantiomers, which can be efficiently separated using simple crystallization techniques, significantly enhancing optical purity.
Q: What are the primary cost drivers eliminated in this manufacturing process?
A: The most significant cost reduction comes from eliminating column chromatography, which is traditionally required to separate enantiomers in non-stereoselective routes. By shifting the purification strategy to crystallization, the process reduces solvent consumption, equipment complexity, and processing time, making it highly suitable for large-scale industrial production.
Q: Is the chiral auxiliary used in this process commercially viable for scale-up?
A: Yes, the process utilizes L-menthol, a readily available and cost-effective natural product from the chiral pool. The patent demonstrates that L-menthol chloroformate provides superior chirality controllability compared to other auxiliaries like Naproxen or Mandelic acid derivatives, ensuring both economic feasibility and high stereochemical fidelity during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lamivudine Supplier
The technological potential of this stereoselective synthesis route is immense, offering a clear path to high-quality, cost-effective Lamivudine production. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemical innovations like this are successfully translated into reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the < 0.2% enantiomeric impurity levels demanded by global pharmacopeias. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process development team works tirelessly to optimize every step of the synthesis, from the initial chiral auxiliary introduction to the final crystallization of the API.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this crystallization-based method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to secure a stable, high-quality supply of Lamivudine intermediates that meets the highest standards of the global pharmaceutical industry.
