Optimizing Tebipenem Pivoxil Production: A Strategic Analysis of Patent CN103012406A for Commercial Scale-Up
The pharmaceutical landscape for oral carbapenems has been significantly advanced by the development of Tebipenem Pivoxil, also known as L-084, a potent broad-spectrum antibacterial agent. The preparation method disclosed in patent CN103012406A represents a critical evolution in the synthetic strategy for this vital API intermediate, addressing long-standing challenges related to scalability and purity. Unlike previous iterations that relied heavily on cumbersome purification techniques, this novel approach leverages a streamlined three-step sequence that prioritizes industrial feasibility. The core innovation lies in the strategic selection of starting materials and the implementation of robust purification protocols that bypass the need for silica gel chromatography. For technical decision-makers evaluating supply chain resilience, understanding the nuances of this specific pathway is essential for securing a reliable Tebipenem Pivoxil supplier capable of meeting stringent global quality standards.
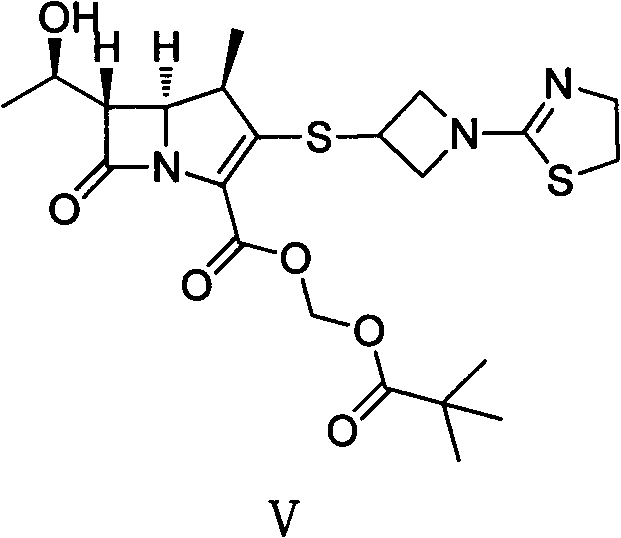
The structural complexity of Tebipenem Pivoxil demands a synthesis route that balances stereochemical integrity with economic efficiency. As illustrated in the molecular diagram, the presence of the beta-lactam ring and the specific thiazoline side chain requires precise control over reaction conditions to prevent degradation. The patent data highlights a method that not only achieves the correct stereochemistry but also optimizes the overall yield through careful manipulation of reaction parameters such as temperature and solvent systems. This level of control is paramount for R&D directors who must ensure that the final active pharmaceutical ingredient meets rigorous impurity profiles without resorting to excessive reprocessing steps that erode profit margins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-084 has been plagued by inefficiencies that hinder large-scale commercial production. Prior art, including patents from major Japanese pharmaceutical entities, often described routes that necessitated the use of silica gel chromatography or macroporous resin purification at multiple stages. These traditional purification methods are inherently batch-oriented, labor-intensive, and difficult to translate into continuous flow or large-batch reactor environments. Furthermore, the reliance on specialized organometallic reagents in some legacy routes introduced significant cost volatility and supply chain risks. The cumulative effect of these limitations was a process with lower total recovery rates and higher generation of chemical waste, making it economically unviable for mass market penetration of this critical oral antibiotic.
The Novel Approach
In stark contrast, the methodology outlined in CN103012406A introduces a paradigm shift by replacing chromatographic purification with crystallization-based isolation. This novel approach utilizes 6-MAP as a foundational building block, reacting it with 3-sulfydryl azetidine hydrochloride under mild alkaline conditions to form the key intermediate. The subsequent deprotection step employs catalytic hydrogenation, a well-established and scalable technology, to remove protecting groups cleanly. Perhaps most ingeniously, the final esterification step generates the reactive iodomethyl pivalate in situ, thereby circumventing the stability issues associated with isolating this reagent. This holistic redesign of the synthetic pathway results in a process that is not only simpler to operate but also inherently safer and more environmentally compliant, offering substantial advantages for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Phase Transfer Catalyzed Esterification
The final step of this synthesis involves a sophisticated esterification reaction that converts the carboxylic acid intermediate into the pivoxil ester prodrug. This transformation is facilitated by the in situ generation of iodomethyl pivalate from chloromethyl pivalate and sodium iodide or potassium iodide in an aprotic polar solvent such as DMF. The reaction mechanism relies heavily on the nucleophilic substitution where the iodide ion displaces the chloride, creating a more reactive alkylating agent that immediately reacts with the carboxylate anion. The presence of a phase transfer catalyst, specifically tetrabutylammonium iodide or triethyl benzyl ammonium iodide, is crucial for enhancing the reaction rate by transporting the ionic species into the organic phase where the reaction occurs. This mechanistic detail ensures high conversion rates even at moderate temperatures, typically ranging from -20°C to 20°C, preserving the sensitive beta-lactam ring from thermal degradation.
Impurity control in this stage is managed through the precise stoichiometry of the reagents and the choice of base, with diisopropylethylamine (DIPEA) being the preferred option to minimize side reactions. The use of recrystallization from ethyl acetate as the final purification step effectively removes unreacted starting materials and byproducts, ensuring the high-purity Tebipenem Pivoxil required for clinical applications. By avoiding the use of unstable isolated intermediates and leveraging the kinetic advantages of phase transfer catalysis, the process minimizes the formation of regioisomers and degradation products. This rigorous control over the reaction environment demonstrates a deep understanding of physical organic chemistry, providing a robust framework for producing consistent quality batches essential for regulatory approval.
How to Synthesize Tebipenem Pivoxil Efficiently
The synthesis of Tebipenem Pivoxil via this patented route involves a logical progression of coupling, deprotection, and esterification steps designed for maximum efficiency. The process begins with the coupling of the carbapenem core with the thiol side chain, followed by hydrogenolytic removal of the protecting group, and concludes with the pivotal esterification. Each step has been optimized to allow for direct telescoping or minimal workup, reducing solvent consumption and processing time.
- Coupling Reaction: React 6-MAP with 3-sulfydryl azetidine hydrochloride in acetonitrile using DIPEA as base at low temperature to form the protected intermediate.
- Deprotection: Perform catalytic hydrogenation using Pd/C in a mixed solvent system to remove the protecting group, followed by recrystallization.
- Esterification: Generate iodomethyl pivalate in situ from chloromethyl pivalate and sodium iodide, then react with the acid intermediate using a phase transfer catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. The elimination of silica gel chromatography translates directly into reduced operational expenditures, as it removes the need for expensive stationary phases and the associated solvent recovery infrastructure. Additionally, the reliance on commercially available and inexpensive starting materials like 6-MAP mitigates the risk of raw material shortages, ensuring a more stable supply chain for high-purity pharmaceutical intermediates. The simplified workflow also reduces the overall production cycle time, allowing for faster response to market demand fluctuations without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of complex purification methods with straightforward crystallization techniques. By eliminating the need for column chromatography, the manufacturer saves significantly on consumables and labor costs associated with fraction collection and analysis. Furthermore, the in situ generation of reactive reagents prevents the financial loss associated with the degradation of unstable intermediates during storage and transport. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings while maintaining high yield standards.
- Enhanced Supply Chain Reliability: The choice of reagents in this synthetic pathway is strategically aligned with global availability, reducing dependency on niche suppliers. The use of common solvents like acetonitrile, ethyl acetate, and DMF ensures that procurement teams can source materials from multiple vendors, fostering competition and price stability. Moreover, the robustness of the catalytic hydrogenation step means that the process is less susceptible to variations in raw material quality, providing a buffer against supply chain disruptions. This reliability is critical for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent usage and the avoidance of silica waste contribute to a greener manufacturing footprint. The process generates fewer three wastes, simplifying effluent treatment and lowering disposal costs. Scalability is inherently supported by the use of standard unit operations such as filtration and crystallization, which are easily adapted from pilot plant to commercial scale reactors. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Tebipenem Pivoxil using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners.
Q: How does this synthesis method improve purity compared to conventional routes?
A: This method eliminates the need for silica gel chromatography, which is difficult to scale and often leads to product loss. Instead, it utilizes recrystallization techniques, specifically using water and acetone mixtures, to achieve high purity suitable for industrial pharmaceutical standards.
Q: What are the key cost-saving factors in this Tebipenem Pivoxil process?
A: The process uses readily available and inexpensive starting materials like 6-MAP. Furthermore, it avoids expensive organometallic reagents used in other routes and simplifies purification, significantly reducing operational costs and waste treatment expenses.
Q: Why is the in situ generation of iodomethyl pivalate advantageous?
A: Iodomethyl pivalate is highly unstable and difficult to store or transport. By generating it in situ from chloromethyl pivalate and sodium iodide immediately before reaction, the process ensures higher safety, better reactivity, and eliminates the need for handling hazardous unstable intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tebipenem Pivoxil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive landscape of antibiotic production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patent CN103012406A are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Tebipenem Pivoxil meets the highest international standards. Our commitment to technological excellence allows us to offer a reliable Tebipenem Pivoxil supplier partnership that is built on transparency, quality, and consistency.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of this method tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most effective and economical manufacturing strategy available.
