Optimizing Ranolazine Production: A Technical Analysis of Stable Open-Loop Halide Intermediates
Optimizing Ranolazine Production: A Technical Analysis of Stable Open-Loop Halide Intermediates
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, with Ranolazine standing out as a critical antianginal agent that uniquely modulates myocardial metabolism without affecting heart rate or blood pressure. As detailed in patent CN102295622A, a significant technological breakthrough has been achieved in the synthesis of this vital medication by substituting traditional, unstable epoxy compounds with robust open-loop halides. This strategic shift in synthetic design addresses long-standing challenges in impurity control and process safety, offering a pathway to high-purity active pharmaceutical ingredients (APIs). For R&D directors and technical leaders, understanding this transition from epoxide-based routes to halide-mediated condensation is essential for evaluating next-generation manufacturing capabilities. The patent elucidates a method where N-(2,6-dimethylphenyl)-1-piperazineacetamide undergoes condensation with a specific halide intermediate, fundamentally altering the thermodynamic profile of the reaction and simplifying downstream purification.
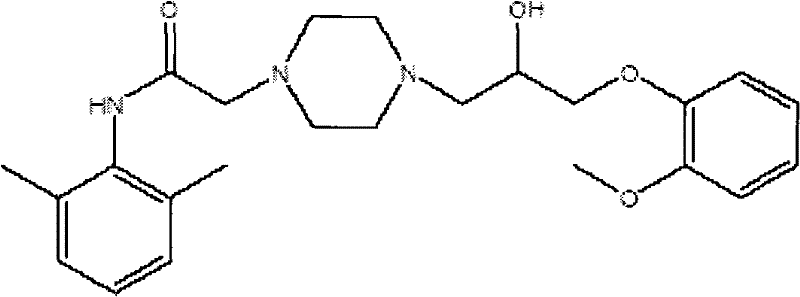
This structural innovation is not merely academic; it represents a tangible solution to the volatility and instability inherent in earlier synthetic pathways. By leveraging the stability of linear halide chains over strained three-membered epoxide rings, manufacturers can achieve superior control over the reaction trajectory. The implications for supply chain reliability are profound, as the new route mitigates the risks associated with difficult separations and thermal degradation. As we delve deeper into the mechanistic and commercial advantages, it becomes clear that this methodology sets a new benchmark for the reliable pharmaceutical intermediate supplier seeking to optimize cost structures while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ranolazine has relied heavily on the reaction between piperazine derivatives and epoxide intermediates, specifically 1-(2-methoxyphenoxy)-2,3-propylene oxide. While conceptually straightforward, this conventional approach is plagued by significant thermodynamic and kinetic hurdles that complicate large-scale production. The primary issue lies in the inherent instability of the oxirane ring under the alkaline conditions required for the condensation reaction. This instability leads to a competitive equilibrium where the epoxide ring opens and subsequently re-closes, often resulting in the formation of complex polymeric byproducts and oligomers. These side reactions not only consume valuable starting materials but also create a nightmare scenario for purification teams, as separating these structurally similar impurities requires extremely harsh conditions.
Furthermore, the isolation of the epoxide intermediate itself is an energy-intensive bottleneck. The patent data highlights that obtaining pure epoxide necessitates fractional distillation at temperatures exceeding 250°C under very low vacuum tightness (approximately 5mmHg). Such extreme operating parameters demand specialized, high-cost equipment and pose significant safety risks regarding thermal runaway and equipment failure. The combination of difficult separation, high energy consumption, and the generation of intractable impurity profiles makes the conventional epoxide route suboptimal for modern, cost-sensitive cost reduction in pharmaceutical intermediates manufacturing. The inability to consistently reach the purity levels required for subsequent reaction steps often leads to batch failures or extensive rework, further eroding profit margins.
The Novel Approach
In a decisive move to overcome these deficiencies, the novel approach described in the patent replaces the problematic epoxide with an open-loop halide, specifically compounds where X is chlorine or bromine. This structural modification fundamentally changes the physical chemistry of the intermediate. Unlike the strained, reactive epoxide ring, the open-chain halide is thermodynamically stable and does not suffer from ring-closing equilibria. This stability translates directly into operational simplicity; the intermediate can be isolated and purified under much milder conditions, eliminating the need for the dangerous high-temperature distillation required by the prior art. The absence of the triatomic ring tension means the molecule is less prone to spontaneous polymerization, resulting in a cleaner reaction profile with fewer unknown impurities.
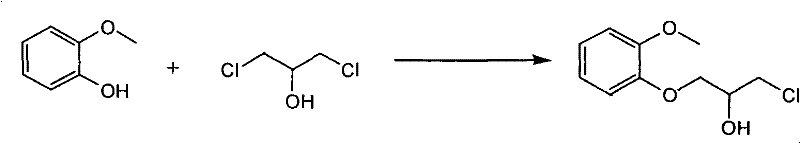
The synthesis of this key intermediate involves the reaction of guaiacol (2-methoxyphenol) with 1,3-dichloro-2-propanol or its dibromo analogue under alkaline conditions. As illustrated in the reaction scheme, this nucleophilic substitution proceeds smoothly to yield the linear halide alcohol. Because the boiling point of these open-loop halides is significantly lower than their epoxide counterparts, they are far easier to separate from unreacted starting materials and solvents. This ease of purification ensures that the material entering the final coupling step is of exceptionally high quality, which is a prerequisite for achieving the stringent purity specifications demanded by regulatory bodies. By decoupling the synthesis from the constraints of epoxide chemistry, this novel approach offers a robust platform for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nucleophilic Substitution and Condensation
The core of this improved synthesis lies in the mechanistic elegance of the final condensation step, where the piperazine nitrogen acts as a nucleophile attacking the electrophilic carbon bearing the halogen. In the conventional epoxide route, the nucleophilic attack is on a strained ring, which, while reactive, is susceptible to competing hydrolysis and polymerization in the presence of base. In contrast, the novel route utilizes a classic SN2-type displacement on a primary or secondary carbon attached to a leaving group (chlorine or bromine). This reaction is performed in a mixed solvent system of alcohols (such as methanol, ethanol, or isopropanol) and toluene, under reflux conditions with a base like potassium carbonate or sodium bicarbonate. The choice of solvent is critical; the alcohol facilitates the dissolution of polar intermediates, while toluene allows for the removal of water via azeotropic distillation, driving the equilibrium towards product formation.
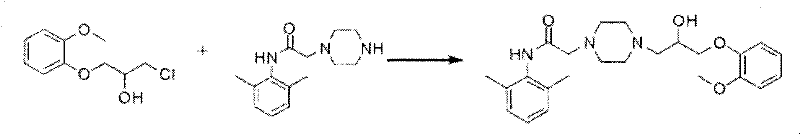
From an impurity control perspective, this mechanism offers distinct advantages. The reaction conditions are sufficiently mild to prevent the degradation of the sensitive amide bond present in the piperazine fragment, yet energetic enough to ensure complete conversion of the halide. The base serves a dual purpose: it deprotonates the piperazine nitrogen to enhance its nucleophilicity and neutralizes the hydrogen halide byproduct generated during the substitution. This prevents the accumulation of acidic species that could catalyze unwanted side reactions. Furthermore, because the starting halide intermediate is free from polymeric contaminants, the final crude product contains a much simpler impurity profile. This simplifies the crystallization process, allowing for the isolation of Ranolazine with purity levels exceeding 99% as demonstrated in the patent examples. The ability to control the stoichiometry precisely (molar ratios of 0.8~1.3:1) further minimizes the presence of unreacted starting materials, streamlining the workup procedure.
How to Synthesize Ranolazine Efficiently
Implementing this synthesis requires a disciplined approach to unit operations, focusing on the preparation of the two key fragments before their final union. The process begins with the acylation of 2,6-xylidine followed by piperazine substitution to form the amide fragment, and parallel to this, the etherification of guaiacol to form the halide fragment. The convergence of these two streams in the final reactor represents the critical control point for quality. Detailed standard operating procedures regarding temperature ramps, addition rates, and quench protocols are essential to replicate the high yields reported in the literature. For a comprehensive breakdown of the specific reagent quantities, reaction times, and workup procedures validated in the patent, please refer to the standardized guide below.
- Synthesize N-(2,6-dimethylphenyl)-1-piperazineacetamide by reacting 2,6-xylidine with chloroacetyl chloride followed by piperazine substitution.
- Prepare the key intermediate 1-halo-3-(2-methoxyphenoxy)-2-propanol by reacting guaiacol with 1,3-dihalo-2-propanol under alkaline conditions.
- Perform the final condensation reaction between the piperazine amide and the open-loop halide intermediate in an alcohol/toluene solvent system with a base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this open-loop halide methodology presents a compelling value proposition centered on risk mitigation and operational efficiency. The elimination of extreme processing conditions directly correlates to reduced capital expenditure and lower operating costs. Facilities no longer need to invest in specialized high-temperature distillation columns capable of withstanding 250°C and deep vacuum, nor do they need to manage the safety protocols associated with such hazardous operations. This simplification of the infrastructure requirements allows for production in standard glass-lined or stainless steel reactors, significantly broadening the pool of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis. Consequently, this enhances supply chain resilience by reducing dependency on niche facilities with limited availability.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the drastic reduction in energy consumption. By avoiding the high-temperature fractionation required for epoxide purification, the process energy load is significantly lowered. Additionally, the higher selectivity of the halide coupling reaction reduces the formation of heavy ends and polymeric waste, leading to improved overall mass balance and yield. The use of common, inexpensive solvents like toluene and ethanol, combined with readily available inorganic bases, further drives down the raw material cost profile. These factors collectively contribute to substantial cost savings without compromising the quality of the final API.
- Enhanced Supply Chain Reliability: The stability of the open-loop halide intermediate is a game-changer for inventory management. Unlike epoxides, which may degrade or polymerize upon storage, the halide intermediates are shelf-stable, allowing manufacturers to produce and stockpile key building blocks in anticipation of demand surges. This decoupling of the synthesis steps provides a buffer against supply disruptions, ensuring continuous production flow. Furthermore, the robustness of the reaction conditions means that batch-to-batch variability is minimized, reducing the likelihood of failed batches that could disrupt delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The avoidance of high-temperature operations reduces the carbon footprint of the manufacturing process. The simpler impurity profile facilitates easier wastewater treatment, as there are fewer complex organic polymers to degrade. The process aligns well with green chemistry principles by improving atom economy and reducing the need for aggressive purification solvents. This compliance with stricter environmental regulations future-proofs the supply chain against evolving regulatory landscapes, making it a sustainable choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Ranolazine synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical benefits for industry stakeholders. Understanding these nuances is critical for making informed sourcing and development decisions.
Q: Why is the open-loop halide intermediate superior to the traditional epoxide route?
A: Traditional epoxide intermediates suffer from ring-opening and re-closing equilibria, leading to polymeric impurities and requiring harsh distillation conditions (>250°C). The open-loop halide is structurally stable, has a lower boiling point, and eliminates the risk of epoxide polymerization, ensuring higher purity and easier isolation.
Q: What are the critical quality parameters for the Ranolazine intermediate?
A: Critical parameters include the absence of polymeric byproducts common in epoxide routes, high chemical purity (demonstrated >99% in patent examples), and strict control over the halogen content to ensure complete conversion in the final coupling step.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method utilizes standard reflux conditions in common solvents like toluene and ethanol, avoiding the extreme vacuum and temperature requirements of the prior art. This makes it highly suitable for large-scale commercial manufacturing with reduced equipment stress.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ranolazine Supplier
The technical advantages outlined in patent CN102295622A demonstrate a clear path toward more efficient and sustainable Ranolazine production. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel halide-based route are fully realized at an industrial scale. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the exacting standards required for cardiovascular drug manufacturing.
We invite you to collaborate with us to optimize your supply chain for Ranolazine and related cardiovascular intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and discuss route feasibility assessments that can drive value and reliability in your pharmaceutical manufacturing operations.
