Advanced Oxidation Protocol for High-Purity Prothioconazole Commercial Scale-Up
Advanced Oxidation Protocol for High-Purity Prothioconazole Commercial Scale-Up
The global demand for high-efficacy fungicides continues to drive innovation in the synthesis of key agrochemical intermediates, with Prothioconazole standing out as a critical molecule for crop protection. Patent CN115448889A introduces a groundbreaking preparation method that addresses long-standing challenges in yield, purity, and environmental impact associated with traditional manufacturing routes. This technical insight report analyzes the proprietary oxidation protocol which leverages phase transfer catalysis to achieve product yields ranging from 90.0% to 96.0% and mass content exceeding 98.0%. By operating under mild thermal conditions between 15°C and 40°C, the process mitigates the thermal instability issues often encountered in triazolethione synthesis, ensuring a robust supply chain for reliable agrochemical intermediate supplier partners seeking consistent quality. The integration of solvent recycling and catalyst regeneration further underscores the commercial viability of this approach for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Prothioconazole has been plagued by complex post-treatment procedures and suboptimal reaction conditions that hinder cost reduction in agrochemical manufacturing. Traditional methods often rely on harsh oxidation environments using ferric chloride in aromatic solvents at elevated temperatures, which frequently lead to the formation of tar and significant product decomposition. For instance, prior art such as CN108689952A requires reaction times as long as 12 hours and results in product discoloration due to entrained iron salts, necessitating additional purification steps that increase waste generation. Furthermore, conventional processes typically involve multiple solvent exchanges, acidification, and alkalization steps, creating substantial volumes of saline wastewater that pose severe environmental compliance challenges. The inability to effectively recycle solvents like toluene and methanol in these older routes results in high raw material consumption and elevated operational expenditures, making the commercial scale-up of complex agrochemical intermediates economically inefficient.
The Novel Approach
The methodology disclosed in CN115448889A represents a paradigm shift by introducing a biphasic system enhanced with quaternary ammonium phase transfer catalysts to streamline the oxidation of the precursor Compound A. This novel approach allows the reaction to proceed efficiently at significantly lower temperatures, specifically within the 15°C to 40°C range, which drastically reduces energy consumption and prevents the thermal degradation of the sensitive triazolethione structure. By utilizing a multifunctional filter for simultaneous filtration, slurry washing, and vacuum drying, the process eliminates the need for multiple vessel transfers, thereby minimizing product loss and exposure to contaminants. The strategic selection of solvents such as o-xylene or chlorobenzene, combined with the ability to directly reuse the organic phase in subsequent batches, creates a closed-loop system that enhances supply chain reliability. This streamlined workflow not only accelerates the production cycle but also ensures that the final product meets stringent purity specifications without the need for extensive recrystallization or chelating agents.
Mechanistic Insights into FeCl3-Catalyzed Oxidation with Phase Transfer
The core chemical transformation involves the oxidative dehydrogenation of 2-(1-chloro-cycloalkane-1-yl)-1-(2-chlorophenyl)-2-hydroxyl-3-(1,2,4-triazolidine-5-sulfur-1-yl)propane to form the stable triazolethione ring system characteristic of Prothioconazole. 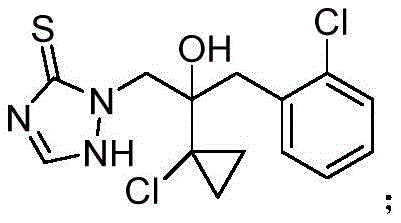 . In this mechanism, the phase transfer catalyst plays a pivotal role by facilitating the transport of the oxidizing species, such as ferric ions or dissolved oxygen, across the interface of the organic and aqueous phases. This interfacial acceleration ensures that the oxidation proceeds rapidly and selectively, minimizing the formation of over-oxidized byproducts or sulfone impurities that could compromise the biological activity of the fungicide. The mild reaction conditions preserve the integrity of the cyclopropyl and chlorophenyl groups, which are essential for the molecule's binding affinity to the target fungal enzymes. Understanding this catalytic cycle is crucial for R&D directors aiming to replicate the high selectivity and yield reported in the patent data.
. In this mechanism, the phase transfer catalyst plays a pivotal role by facilitating the transport of the oxidizing species, such as ferric ions or dissolved oxygen, across the interface of the organic and aqueous phases. This interfacial acceleration ensures that the oxidation proceeds rapidly and selectively, minimizing the formation of over-oxidized byproducts or sulfone impurities that could compromise the biological activity of the fungicide. The mild reaction conditions preserve the integrity of the cyclopropyl and chlorophenyl groups, which are essential for the molecule's binding affinity to the target fungal enzymes. Understanding this catalytic cycle is crucial for R&D directors aiming to replicate the high selectivity and yield reported in the patent data.
Impurity control is inherently built into the reaction design through the precise management of the aqueous phase and the subsequent washing protocols. The use of a multifunctional filter allows for rigorous slurry washing with water, which effectively removes residual iron salts and inorganic byproducts that are common sources of coloration and instability in the final API intermediate. Unlike traditional methods that require chelating agents to sequester metal ions, this process relies on physical separation and the regeneration of the iron catalyst in the aqueous stream, where ferrous salts are re-oxidized to ferric chloride using hydrogen peroxide. This regeneration loop not only reduces the consumption of fresh oxidant but also prevents the accumulation of metal impurities in the product stream. The result is a high-purity Prothioconazole with mass content reaching 98.0%-99.0%, demonstrating superior control over the impurity profile compared to the 95%-97% typical of market standards.
How to Synthesize Prothioconazole Efficiently
The synthesis of Prothioconazole via this optimized route requires careful attention to the stoichiometry of the phase transfer catalyst and the maintenance of the biphasic reaction environment to ensure maximum efficiency. The process begins with the suspension of Compound A in a mixture of organic solvent and water, followed by the addition of the catalyst and oxidant under controlled temperature conditions to initiate the reaction. Detailed standard operating procedures regarding the specific addition rates, stirring speeds, and filtration parameters are critical for maintaining the reproducibility of the 90.0%-96.0% yield range. Operators must ensure that the separation of phases is complete before recycling the organic layer to prevent cross-contamination that could affect subsequent batches. The following guide outlines the standardized synthesis steps derived from the patent examples to facilitate technology transfer and process validation.
- Prepare the reaction mixture by combining Compound A, organic solvent, water, and a quaternary ammonium phase transfer catalyst at 15°C.
- Introduce the oxidizing agent (Ferric Chloride or Air/Oxygen) and maintain the temperature between 15-40°C for 1-5 hours.
- Filter the product using a multifunctional filter, wash with water to remove salts, and recover the organic phase for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthesis route offers substantial cost savings by eliminating the need for expensive chelating agents and reducing the overall solvent consumption through efficient recycling protocols. The ability to reuse the organic phase directly in the next batch significantly lowers the raw material expenditure, while the regeneration of the aqueous iron catalyst solution minimizes the purchase of fresh oxidizing agents. This reduction in consumable inputs translates to a more stable cost structure, protecting margins against volatility in chemical raw material pricing. Furthermore, the simplified post-treatment process reduces the labor and utility costs associated with extended reaction times and complex purification sequences, enhancing the overall economic feasibility of the manufacturing campaign.
- Cost Reduction in Manufacturing: The elimination of multiple solvent exchanges and the removal of chelating agents from the post-treatment workflow drastically simplifies the production process, leading to significant operational cost reductions. By avoiding the use of expensive purification additives and minimizing solvent loss through high-efficiency recovery, the overall cost of goods sold is optimized without compromising product quality. The streamlined process also reduces the energy load required for heating and cooling, as the reaction operates effectively at near-ambient temperatures, further contributing to lower utility expenses. These cumulative efficiencies ensure that the manufacturing process remains competitive in a price-sensitive global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a broad temperature range of 15°C to 40°C, reduces the risk of batch failures due to thermal excursions, thereby ensuring consistent supply continuity. The use of readily available and inexpensive catalysts such as tetrabutylammonium bromide ensures that raw material sourcing is not a bottleneck, supporting reducing lead time for high-purity agrochemical intermediates. Additionally, the high yield and selectivity of the process mean that less starting material is required to produce the same amount of final product, optimizing inventory turnover and warehouse utilization. This reliability is critical for supply chain heads managing just-in-time delivery schedules for downstream formulation plants.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard equipment like multifunctional filters that can be easily scaled from pilot to commercial production volumes. The significant reduction in wastewater generation, achieved through the recycling of aqueous phases and the avoidance of saline byproducts, simplifies compliance with stringent environmental regulations. By minimizing the three wastes (waste gas, waste water, waste residue), the facility reduces its environmental footprint and the associated costs of waste disposal and treatment. This alignment with green chemistry principles enhances the corporate sustainability profile and mitigates regulatory risks associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-content Prothioconazole preparation method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this technology for their own production lines or sourcing strategies. The responses highlight the key differentiators of this method compared to existing industrial standards.
Q: How does the new method improve product purity compared to traditional synthesis?
A: The novel approach utilizes a phase transfer catalyst and mild temperatures (15-40°C) to minimize thermal decomposition and tar formation, achieving purity levels of 98.0%-99.0% compared to the traditional 95%-97%.
Q: What are the environmental benefits of this preparation method?
A: The process allows for the direct recycling of the organic phase and the regeneration of the aqueous iron catalyst solution, significantly reducing wastewater generation and solvent consumption.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method simplifies post-treatment by using a multifunctional filter for filtration, washing, and drying in one unit, making it highly scalable and operationally efficient for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prothioconazole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced synthesis protocols like the one analyzed in this report to deliver exceptional value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Prothioconazole meets the highest international standards for agrochemical applications. Our infrastructure is designed to support the complex requirements of modern fungicide production, including the handling of sensitive intermediates and the implementation of eco-friendly waste management systems.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain needs. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of our manufacturing capabilities. Whether you require immediate stock or a long-term tolling agreement, our dedicated support ensures that your production schedules are met with precision and reliability. Contact us today to discuss how our optimized synthesis routes can enhance your product portfolio and drive operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
