Advanced Catalytic Bromination for High-Purity 5-Bromo-2-Chlorobenzoic Acid Production
The pharmaceutical industry's relentless pursuit of effective antidiabetic agents has placed SGLT-2 inhibitors at the forefront of modern therapeutic strategies, creating an unprecedented demand for high-quality key intermediates. Patent CN110002989B, published in mid-2021, introduces a groundbreaking preparation method for 5-bromo-2-chlorobenzoic acid, a critical building block in the synthesis of these next-generation diabetes medications. This innovation addresses long-standing challenges in organic synthesis by utilizing a novel catalytic system that dramatically enhances regioselectivity during the bromination of 2-chlorobenzoic acid. By integrating a specific sulfur-containing catalyst into an N-bromosuccinimide (NBS) and sulfuric acid system, the technology effectively suppresses the formation of stubborn isomeric impurities, particularly the 4-bromo derivative, which has historically plagued manufacturers. This technical breakthrough not only simplifies the purification workflow but also ensures the delivery of ultra-high purity materials essential for stringent regulatory compliance in active pharmaceutical ingredient (API) production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-bromo-2-chlorobenzoic acid has been fraught with significant technical and economic hurdles that hinder efficient large-scale production. One prevalent prior art route involves the hydrolysis of 5-bromo-2-chlorotrifluoromethane, a process that not only relies on expensive starting materials but also generates substantial quantities of hazardous fluorine-containing wastewater, imposing heavy environmental treatment costs. Another common approach utilizes salicylic acid as a precursor, undergoing sequential bromination and chlorination; however, this multi-step pathway suffers from prolonged reaction times and inferior yields, making it economically unviable for competitive markets. Furthermore, direct bromination of 2-chlorobenzoic acid using standard NBS systems without specialized additives typically results in poor regioselectivity, yielding a mixture where the desired 5-bromo isomer constitutes only 60-70% of the product, accompanied by approximately 10-12% of the difficult-to-remove 4-bromo-2-chlorobenzoic acid impurity.
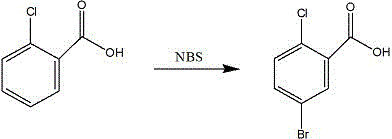
The Novel Approach
The methodology disclosed in CN110002989B represents a paradigm shift by transforming a standard electrophilic substitution into a highly selective catalytic process. By introducing a reducing sulfur-containing salt, such as sodium sulfide, sodium sulfite, or potassium sulfide, directly into the reaction matrix, the electronic environment of the aromatic ring is subtly modulated to favor substitution at the 5-position. This strategic addition effectively blocks the formation of the 4-bromo isomer, reducing its content to virtually zero in the crude product. Consequently, the need for multiple, yield-depleting recrystallization cycles is eliminated, as a single refining step suffices to achieve purity levels exceeding 99.5%. This approach leverages cheap, commercially available raw materials and operates under mild conditions, offering a robust solution that bypasses the complexity and waste generation associated with fluorine-based or multi-step synthetic routes.
Mechanistic Insights into Sulfur-Catalyzed Regioselective Bromination
The core of this technological advancement lies in the intricate interaction between the sulfur-based catalyst and the electrophilic brominating species within the acidic medium. In a conventional NBS/sulfuric acid system, the generation of the bromonium ion or molecular bromine leads to a statistical distribution of products governed primarily by the inherent electronic directing effects of the existing substituents on the benzene ring. However, the introduction of a reducing sulfur species, such as sulfide or sulfite ions, likely acts as a transient modifier of the electrophile or forms a transient complex with the substrate that sterically or electronically disfavors attack at the 4-position. This catalytic influence ensures that the bromination occurs almost exclusively at the 5-position, para to the carboxylic acid group and meta to the chlorine atom, which is the thermodynamically and kinetically favored position for the desired pharmaceutical intermediate. The result is a reaction profile that is not only faster but also inherently cleaner, minimizing the formation of side products that typically complicate downstream processing.
From an impurity control perspective, this mechanism offers a decisive advantage over traditional purification-dependent strategies. In standard processes, the 4-bromo-2-chlorobenzoic acid impurity possesses polarity and solubility characteristics remarkably similar to the target 5-bromo isomer, making separation via crystallization or chromatography extremely inefficient and costly. By preventing the formation of this specific isomer at the molecular level during the reaction phase, the patent circumvents the need for aggressive purification techniques that often sacrifice overall yield. The crude product obtained from this catalytic system already exhibits a significantly improved profile, with the 4-bromo impurity content dropping from typical levels of over 12% to undetectable traces. This intrinsic purity allows for a straightforward workup involving precipitation in ice water followed by a single recrystallization in solvents like methanol, ethanol, or acetic acid, ensuring that the final material meets the rigorous specifications required for GMP manufacturing of SGLT-2 inhibitors.
How to Synthesize 5-Bromo-2-Chlorobenzoic Acid Efficiently
The implementation of this high-selectivity synthesis route is designed for seamless integration into existing chemical manufacturing infrastructure, requiring only standard reactor setups and common reagents. The process begins with the dissolution of 2-chlorobenzoic acid in concentrated sulfuric acid, followed by the precise addition of the sulfur-containing catalyst, such as sodium sulfide, under controlled stirring. Once the solution is homogenous, N-bromosuccinimide is introduced at temperatures ranging from 10°C to 50°C, initiating the selective monobromination. The reaction proceeds rapidly, typically completing within 10 to 120 minutes depending on the specific catalyst loading and temperature, after which the mixture is quenched in an ice-water bath to precipitate the crude product. For detailed operational parameters, stoichiometric ratios, and safety protocols, please refer to the standardized synthesis guide below.
- Dissolve 2-chlorobenzoic acid in concentrated sulfuric acid and add a sulfur-containing catalyst such as sodium sulfide or sodium sulfite.
- Add N-bromosuccinimide (NBS) to the reaction mixture at a controlled temperature between 10°C and 50°C to initiate selective monobromination.
- Quench the reaction in ice water, filter the crude product, and perform a single recrystallization using methanol/water or ethanol to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic bromination technology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The shift from expensive, specialty starting materials like trifluoromethyl derivatives to commodity chemicals like 2-chlorobenzoic acid fundamentally alters the cost structure of the supply chain, insulating production budgets from the volatility of niche raw material markets. Furthermore, the elimination of complex multi-step sequences and the reduction of purification stages significantly shorten the manufacturing cycle time, enhancing the agility of the supply chain to respond to fluctuating market demands for SGLT-2 inhibitor intermediates. The simplicity of the process also reduces the dependency on specialized equipment or hazardous handling procedures, thereby lowering operational risks and ensuring a more consistent and reliable flow of materials to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the synthetic route and the utilization of low-cost, widely available feedstocks. By replacing expensive fluorine-containing precursors with 2-chlorobenzoic acid and eliminating the need for multiple recrystallization steps, the overall consumption of solvents, energy, and labor is substantially decreased. The high selectivity of the reaction means that less material is lost to impurity formation and subsequent purification waste, leading to a significant improvement in mass efficiency. Additionally, the avoidance of hazardous fluorine waste treatment further reduces the environmental compliance costs associated with production, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for pharmaceutical companies, and this method strengthens supply security by relying on a robust and simple chemical transformation. The raw materials, including NBS and sulfuric acid, are commodity chemicals with stable global supply chains, reducing the risk of shortages that can occur with specialized reagents. The process's tolerance for mild reaction conditions and its ability to produce high-purity material in a single batch operation minimize the potential for production delays caused by equipment failures or complex process deviations. This stability ensures that procurement teams can secure long-term contracts with greater confidence, knowing that the manufacturing process is resilient and scalable.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of extreme conditions or sensitive catalytic systems that often fail upon scale-up. The reaction generates minimal hazardous waste compared to fluorine-based routes, aligning with increasingly stringent global environmental regulations and sustainability goals. The use of water and common alcohols for recrystallization simplifies solvent recovery and waste management, reducing the facility's environmental footprint. This eco-friendly profile not only facilitates easier regulatory approval but also enhances the corporate social responsibility standing of the supply chain, making it an attractive option for multinational corporations committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-selectivity bromination technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method outperforms legacy synthesis routes. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their specific manufacturing needs and for procurement specialists assessing the value proposition of suppliers utilizing this advanced methodology.
Q: How does the new catalytic method improve upon traditional NBS bromination?
A: Traditional NBS bromination of 2-chlorobenzoic acid often yields significant amounts of the 4-bromo isomer impurity (around 10-12%), which is difficult to separate. The patented method introduces a sulfur-containing catalyst (e.g., sodium sulfide) that specifically inhibits the formation of the 4-bromo isomer, reducing its content to negligible levels and allowing for >99.5% purity after just one recrystallization.
Q: What are the cost advantages of this synthesis route compared to fluorine-based methods?
A: Unlike routes starting from expensive 5-bromo-2-chlorotrifluoromethane which generate hazardous fluorine-containing wastewater, this method uses cheap and readily available 2-chlorobenzoic acid. It eliminates the need for complex hydrolysis steps and expensive raw materials, significantly lowering the overall production cost and environmental treatment burden.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. It operates at mild temperatures (10-50°C), uses common reagents like sulfuric acid and NBS, and requires only a simple workup involving ice-water precipitation and standard recrystallization. The high yield (approx. 85%) and single-step purification make it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo-2-Chlorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation antidiabetic therapies depends on the availability of intermediates produced with uncompromising quality and consistency. Our technical team has extensively analyzed the catalytic mechanisms described in CN110002989B and possesses the expertise to implement this high-selectivity bromination process on an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to detect and quantify trace isomeric impurities, guaranteeing that every batch of 5-bromo-2-chlorobenzoic acid delivered adheres to the highest standards required for SGLT-2 inhibitor synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that demonstrates how switching to this catalytic route can optimize your overall production economics. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your project timelines. Let us be your strategic partner in delivering high-performance chemical solutions that drive innovation in the global pharmaceutical market.
