Advanced Solvent-Free Synthesis of Beta-Enaminones for Scalable Pharmaceutical Manufacturing
Introduction to Green Catalytic Synthesis
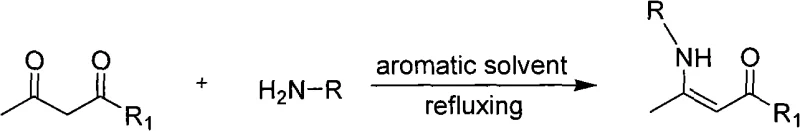
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards greener, more sustainable manufacturing processes, driven by both regulatory pressure and the economic necessity of waste reduction. Patent CN101602681A represents a significant technological breakthrough in this domain, specifically addressing the synthesis of beta-enaminone and ester derivatives, which are critical building blocks in medicinal chemistry. These compounds serve as versatile intermediates for constructing complex heterocyclic systems found in numerous bioactive molecules, including anti-inflammatory, antibacterial, and antitumor agents. The disclosed methodology leverages Nickel(II) acetate as a robust Lewis acid catalyst to facilitate the condensation of amino compounds with 1,3-dicarbonyl compounds. Unlike traditional methods that rely heavily on toxic aromatic solvents and energy-intensive reflux conditions, this innovation operates under solvent-free conditions at mild temperatures ranging from 25°C to 50°C. This approach not only aligns with the principles of green chemistry by eliminating volatile organic compound (VOC) emissions but also streamlines the downstream processing workflow, offering a compelling value proposition for manufacturers seeking to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of beta-enaminones has been plagued by significant operational and environmental inefficiencies that hinder large-scale adoption. Conventional synthetic routes typically necessitate the use of refluxing aromatic solvents, such as toluene or benzene, coupled with Dean-Stark traps to azeotropically remove the water generated during condensation. This reliance on hazardous solvents introduces severe safety risks, including flammability and toxicity concerns, while simultaneously complicating the waste disposal process due to the generation of contaminated solvent streams. Furthermore, many existing catalytic systems require harsh reaction conditions, prolonged heating periods, or the use of expensive and sensitive reagents that degrade upon exposure to atmospheric moisture. These factors collectively contribute to elevated production costs, extended lead times, and a larger environmental footprint, creating substantial bottlenecks for procurement managers aiming to secure reliable sources of complex heterocyclic precursors. The need for specialized equipment to handle high-temperature reflux and solvent recovery further exacerbates the capital expenditure required for setting up these production lines.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN101602681A introduces a streamlined, solvent-free protocol that fundamentally redefines the efficiency of enaminone synthesis. By utilizing Nickel(II) acetate as a catalyst, the reaction proceeds smoothly without the need for any reaction medium, thereby eliminating the costs and hazards associated with solvent purchase, storage, and recycling. The process operates at remarkably mild temperatures between 25°C and 50°C, significantly reducing energy consumption compared to traditional reflux methods. This novel approach ensures high yields and exceptional product purity while drastically shortening reaction times, with some transformations completing in as little as 4 minutes. The simplicity of the work-up procedure, which involves simple filtration to recover the catalyst followed by standard purification, enhances the overall throughput of the manufacturing process. For supply chain leaders, this translates to a more resilient production model that is less susceptible to solvent supply disruptions and regulatory changes regarding VOC emissions.
Mechanistic Insights into Ni(OAc)2-Catalyzed Condensation
The efficacy of this synthesis relies on the unique Lewis acidic properties of the Nickel(II) center within the Ni(OAc)2 complex. Mechanistically, the nickel catalyst coordinates with the carbonyl oxygen atoms of the 1,3-dicarbonyl substrate, thereby increasing the electrophilicity of the carbonyl carbon. This activation lowers the energy barrier for the nucleophilic attack by the amino group of the amine substrate, facilitating the formation of the tetrahedral intermediate. Subsequent proton transfer and dehydration steps lead to the formation of the stable beta-enaminone double bond system. The choice of Nickel(II) acetate is particularly strategic; unlike stronger Lewis acids that might promote side reactions or substrate decomposition, Ni(OAc)2 provides a balanced activation that preserves the integrity of sensitive functional groups often present in pharmaceutical intermediates. Furthermore, the catalyst exhibits remarkable stability towards water and oxygen, allowing the reaction to be conducted without rigorous inert atmosphere techniques, which simplifies the operational requirements for plant operators and reduces the complexity of the reactor setup.
From an impurity control perspective, the mild reaction conditions play a pivotal role in ensuring the high quality of the final product. Traditional high-temperature methods often lead to thermal degradation of the enaminone product or polymerization of the reactive dicarbonyl starting materials, resulting in complex impurity profiles that are difficult to separate. By maintaining the temperature below 50°C, this novel process minimizes thermal stress on the molecules, effectively suppressing the formation of by-products. The solvent-free environment also prevents solvolysis side reactions that can occur when reactive intermediates interact with protic or polar solvents. This inherent selectivity means that the crude product obtained after the reaction is already of high purity, reducing the burden on downstream purification steps such as chromatography or recrystallization. For R&D directors, this level of control over the impurity profile is critical for meeting the stringent specifications required for API intermediates and ensures a more predictable and robust manufacturing process.
How to Synthesize Beta-Enaminone Derivatives Efficiently
The implementation of this solvent-free catalytic system is designed for ease of adoption in both laboratory and pilot-scale settings. The protocol requires minimal equipment modifications, as it eliminates the need for solvent handling infrastructure and complex reflux setups. Operators simply combine the stoichiometric amounts of the 1,3-dicarbonyl compound and the amine in a reaction vessel, add the catalytic amount of Ni(OAc)2, and stir at the specified mild temperature. Reaction progress is easily monitored using thin-layer chromatography (TLC) with standard eluent systems, allowing for precise determination of the endpoint. Once the reaction is complete, the addition of a small volume of ethyl acetate allows for the dissolution of the organic product while leaving the solid catalyst intact for filtration. This straightforward isolation technique not only saves time but also facilitates the immediate recovery and reuse of the valuable nickel catalyst, reinforcing the economic and environmental benefits of the process. Detailed standardized synthesis steps follow below.
- Combine 1,3-dicarbonyl compounds and amino compounds in a reaction vessel with Ni(OAc)2 catalyst (0.05-0.20 molar equivalent) without adding any solvent.
- Stir the mixture at a controlled temperature between 25°C and 50°C for 4 to 420 minutes, monitoring progress via TLC.
- Upon completion, add ethyl acetate to dissolve the product, filter to recover the solid catalyst, and purify the filtrate via flash chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free nickel-catalyzed technology offers transformative advantages that directly impact the bottom line and operational resilience. The most immediate benefit is the drastic reduction in raw material costs associated with the elimination of bulk organic solvents. In traditional manufacturing, solvents often account for a significant portion of the total material cost, not including the expenses related to their recovery, distillation, and disposal. By removing this requirement entirely, the process achieves a leaner cost structure that is less vulnerable to fluctuations in petrochemical prices. Additionally, the ability to recover and reuse the Ni(OAc)2 catalyst multiple times without loss of activity further drives down the cost of goods sold (COGS). This economic efficiency allows suppliers to offer more competitive pricing for high-purity pharmaceutical intermediates, providing a strategic advantage in cost-sensitive markets.
- Cost Reduction in Manufacturing: The elimination of volatile organic solvents fundamentally alters the cost dynamics of production by removing the need for expensive solvent recovery units and waste treatment facilities. Without the burden of managing hazardous solvent waste, facilities can operate with lower overhead costs and reduced regulatory compliance fees. The catalyst's reusability means that the effective cost per kilogram of catalyst consumed is negligible over multiple batches. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to substantial utility savings. These cumulative efficiencies result in a significantly optimized manufacturing budget, allowing for better margin management in the production of complex fine chemicals.
- Enhanced Supply Chain Reliability: Dependence on specific grades of dry, toxic solvents can create supply chain vulnerabilities, especially during global logistics disruptions. This solvent-free method mitigates such risks by relying on readily available, stable solid reagents and catalysts that are easier to store and transport. The robustness of the Ni(OAc)2 catalyst against moisture and air ensures that the process is less prone to batch failures caused by environmental factors, leading to higher first-pass yields and more consistent delivery schedules. For supply chain planners, this reliability translates to reduced safety stock requirements and improved confidence in meeting Just-In-Time delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, particularly those involving large volumes of flammable solvents. This technology inherently addresses these scalability issues by operating in a neat, solvent-free regime, which simplifies heat transfer management and reduces the fire load of the reactor. The absence of solvent emissions aligns perfectly with increasingly stringent environmental regulations regarding VOC limits, future-proofing the manufacturing site against tighter legislation. The simplified waste stream, consisting primarily of solid catalyst residue and minimal organic washings, is easier to treat and dispose of responsibly. This environmental stewardship enhances the corporate sustainability profile, a key metric for modern procurement evaluations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the practical aspects of adopting this technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines or for sourcing partners assessing the quality capabilities of potential suppliers. The answers reflect the balance between high-performance chemistry and practical manufacturing constraints.
Q: What are the primary advantages of using Ni(OAc)2 over traditional acid catalysts?
A: Ni(OAc)2 offers superior stability against moisture and air compared to many Lewis acids, allows for easy recovery via filtration, and can be reused multiple times without significant loss of activity, reducing waste and cost.
Q: Can this solvent-free method be scaled for industrial production?
A: Yes, the absence of volatile organic solvents simplifies safety protocols and waste treatment, making it highly suitable for commercial scale-up while maintaining high yields and purity standards.
Q: What types of substrates are compatible with this synthesis route?
A: The method is versatile, accommodating various 1,3-dicarbonyl compounds (including beta-keto esters and diketones) and a wide range of amines, including aliphatic and aromatic amines with different electronic properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Enaminone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, green synthetic methodologies is essential for the future of the pharmaceutical industry. Our team of expert chemists has extensively evaluated the Ni(OAc)2 catalyzed solvent-free pathway described in CN101602681A and confirmed its potential for delivering high-quality beta-enaminone derivatives. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed at the gram scale are fully realized in multi-ton manufacturing campaigns. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the exacting standards required for API intermediate synthesis. We are committed to leveraging this innovative technology to provide our partners with superior products that drive their own drug development pipelines forward.
We invite you to collaborate with us to explore how this efficient synthesis route can be tailored to your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this solvent-free process for your specific targets. Please contact us to request specific COA data for our catalog of enaminone derivatives or to discuss route feasibility assessments for custom synthesis projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes innovation, sustainability, and unwavering quality assurance.
