Scalable One-Pot Synthesis of Flavanone Compounds Using Polyphosphoric Acid Catalysis
The strategic development of efficient synthetic routes for bioactive scaffolds remains a cornerstone of modern pharmaceutical manufacturing. Flavanones, a privileged structural motif found in numerous natural products, exhibit a broad spectrum of biological activities including anticancer, anti-inflammatory, and antioxidant properties, making them highly valuable targets for drug discovery programs. Addressing the critical need for scalable and cost-effective production methods, Patent CN115160280A discloses a groundbreaking one-pot synthesis strategy that utilizes polyphosphoric acid (PPA) as a robust catalyst. This innovation represents a paradigm shift from traditional multi-step protocols, offering a streamlined pathway that combines operational simplicity with high chemical efficiency. By leveraging a ternary solvent system of DMF and methanol, the process achieves rapid cyclization under reflux conditions, delivering a diverse library of flavanone derivatives with exceptional yield stability. This technological advancement not only accelerates the timeline for preclinical material supply but also establishes a foundation for the industrial-scale manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the flavanone core has relied heavily on methodologies that impose significant logistical and economic burdens on the supply chain. Conventional approaches often necessitate the use of specialized and expensive starting materials, such as phenyl alkenyl aryl ethers or complex ketene derivatives, which are not readily available in bulk quantities. Furthermore, many established protocols depend on transition metal catalysis or photochemical conversion processes that require stringent reaction conditions and sophisticated equipment. A notable limitation in prior art involves aniline-mediated cyclization strategies, which, while effective, suffer from prolonged reaction times ranging from 7.5 to 24 hours. These extended durations severely limit reactor turnover rates, creating bottlenecks in production scheduling. Additionally, the reliance on exotic reagents often complicates the purification landscape, leading to lower overall mass balance and increased waste generation, which contradicts the principles of green chemistry and sustainable manufacturing.
The Novel Approach
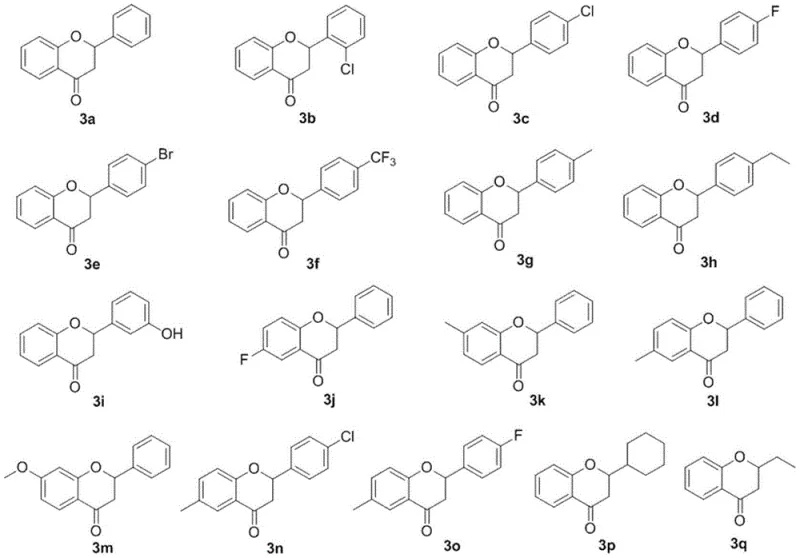
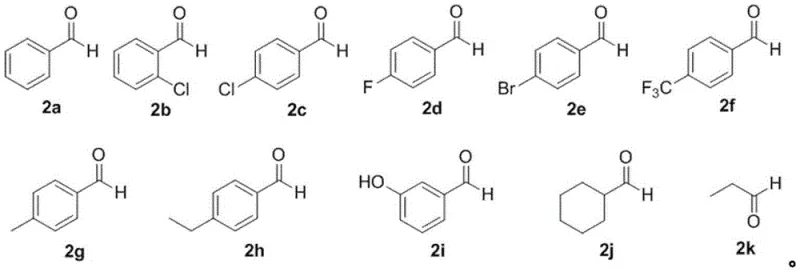
In stark contrast to these legacy methods, the PPA-catalyzed protocol introduced in the patent data offers a remarkably elegant solution that prioritizes atom economy and operational ease. The core of this innovation lies in the direct condensation of commercially abundant 2-hydroxyacetophenones with various aldehydes, eliminating the need for pre-functionalized precursors. As illustrated by the reactant scopes, the method tolerates a wide array of electronic environments on both the ketone and aldehyde components. 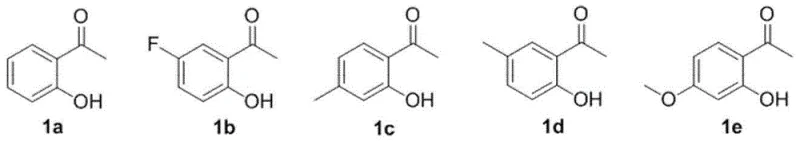
 The use of polyphosphoric acid serves a dual purpose, acting as both a potent Brønsted acid catalyst to drive the aldol condensation and a dehydrating agent to facilitate the subsequent intramolecular cyclization. This one-pot design drastically reduces the number of unit operations, as the reaction proceeds smoothly in a DMF and methanol mixture without the need for inert atmospheres or cryogenic temperatures. The result is a robust process that consistently delivers target flavanones in high yields, with some examples reaching up to 87 percent, thereby validating its potential for immediate adoption in commercial settings.
The use of polyphosphoric acid serves a dual purpose, acting as both a potent Brønsted acid catalyst to drive the aldol condensation and a dehydrating agent to facilitate the subsequent intramolecular cyclization. This one-pot design drastically reduces the number of unit operations, as the reaction proceeds smoothly in a DMF and methanol mixture without the need for inert atmospheres or cryogenic temperatures. The result is a robust process that consistently delivers target flavanones in high yields, with some examples reaching up to 87 percent, thereby validating its potential for immediate adoption in commercial settings.
Mechanistic Insights into PPA-Catalyzed Cyclization
The mechanistic pathway of this transformation is driven by the strong acidic nature of polyphosphoric acid, which effectively activates the carbonyl group of the aldehyde towards nucleophilic attack by the enol form of the 2-hydroxyacetophenone. This initial step generates a chalcone-like intermediate in situ, which subsequently undergoes an intramolecular oxa-Michael addition to close the heterocyclic ring. The unique physicochemical properties of PPA stabilize the transition states involved in this cascade, lowering the activation energy barrier and allowing the reaction to proceed at moderate reflux temperatures. Unlike Lewis acid catalysts that may coordinate irreversibly with oxygenated functional groups, PPA operates through proton transfer mechanisms that are reversible and less prone to causing side reactions such as polymerization or decomposition of sensitive substrates. This gentle yet effective catalytic environment is crucial for maintaining the integrity of functional groups like halogens and trifluoromethyl moieties, ensuring that the final impurity profile is clean and manageable.
From an impurity control perspective, the choice of solvent system plays a pivotal role in suppressing byproduct formation. The combination of DMF and methanol creates a polar medium that solubilizes both the organic substrates and the polyphosphoric acid catalyst, ensuring homogeneous reaction kinetics throughout the vessel. This homogeneity prevents local hot spots or concentration gradients that could lead to over-reaction or the formation of oligomeric impurities. Furthermore, the relatively short reaction window of 7 to 8 hours minimizes the exposure of the product to acidic conditions, reducing the risk of acid-catalyzed degradation or racemization at the chiral center of the flavanone ring. The workup procedure, involving simple aqueous extraction and silica gel chromatography, effectively removes residual PPA and solvent traces, yielding a product that meets stringent purity specifications required for downstream pharmaceutical processing without the need for complex recrystallization sequences.
How to Synthesize Flavanone Compounds Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing pilot and production facilities. The protocol begins with the precise charging of polyphosphoric acid, DMF, and methanol into a standard glass-lined or stainless steel reactor, followed by the sequential addition of the 2-hydroxyacetophenone and aldehyde substrates. The mixture is then heated to reflux, typically maintained for a duration of 7 hours to ensure maximum conversion, as evidenced by the optimization data where extending time beyond this point offered diminishing returns. Following the reaction, the mixture is cooled and subjected to a straightforward extractive workup using ethyl acetate, followed by washing with water and brine to remove inorganic salts and acid residues.
- Prepare the reaction mixture by adding polyphosphoric acid (PPA), N,N-dimethylformamide (DMF), and methanol into a reaction vessel, followed by the addition of 2-hydroxyacetophenone and the selected aldehyde substrate.
- Heat the mixture to reflux conditions and maintain the temperature for a duration of 7 to 8 hours to ensure complete cyclization and conversion.
- Upon completion, perform liquid-liquid extraction using ethyl acetate, wash the organic layer with water and brine, dry over anhydrous sodium sulfate, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this PPA-catalyzed methodology translates into tangible strategic benefits that extend far beyond the laboratory bench. The primary driver of value is the drastic simplification of the raw material portfolio; by utilizing commodity chemicals like 2-hydroxyacetophenone and substituted benzaldehydes, manufacturers can leverage established global supply chains that offer high availability and price stability. This stands in sharp contrast to methods requiring custom-synthesized precursors, which often carry long lead times and volatile pricing structures. Moreover, the elimination of transition metal catalysts removes a significant cost center associated with the procurement of precious metals and the subsequent validation of heavy metal clearance, a critical regulatory hurdle in API manufacturing. The streamlined one-pot nature of the process also implies a reduction in solvent consumption and energy usage per kilogram of product, contributing to a leaner and more sustainable cost structure.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive catalytic systems with polyphosphoric acid is profound, as PPA is a bulk industrial chemical available at a fraction of the cost of palladium or rhodium complexes. This substitution not only lowers the direct material cost but also simplifies the downstream processing requirements, as there is no need for specialized scavenger resins or activated carbon treatments to reduce metal content to ppm levels. The high yields reported, often exceeding 80 percent for a wide range of substrates, further enhance the cost efficiency by maximizing the output from every batch of raw materials charged. Consequently, the overall cost of goods sold (COGS) for these flavanone intermediates can be significantly optimized, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: Reliance on readily available starting materials ensures that production schedules are not held hostage by the supply constraints of niche reagents. The robustness of the reaction conditions, which do not require moisture-free or oxygen-free environments, allows for greater flexibility in manufacturing planning and reduces the risk of batch failures due to environmental excursions. This reliability is crucial for maintaining continuous supply to downstream customers, particularly in the fast-paced pharmaceutical sector where delays in intermediate delivery can stall entire drug development timelines. The scalability of the process from gram to kilogram scale without significant re-optimization further guarantees that supply can be ramped up quickly to meet surging demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with modern green chemistry initiatives by minimizing waste generation and avoiding toxic heavy metals. The simplified workup procedure reduces the volume of aqueous waste streams containing metal ions, easing the burden on wastewater treatment facilities and lowering disposal costs. The use of common solvents like ethyl acetate and methanol facilitates solvent recovery and recycling, further enhancing the environmental footprint of the manufacturing process. These factors collectively make the technology highly attractive for large-scale commercial production, where regulatory compliance and sustainability metrics are increasingly becoming key decision-making criteria for corporate procurement strategies.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this synthesis route for their specific projects, we have compiled answers to common inquiries regarding the process parameters and scope. The following insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a transparent view of the method's capabilities and limitations.
Q: What are the primary advantages of using PPA over traditional transition metal catalysts for flavanone synthesis?
A: Polyphosphoric acid (PPA) offers a significant cost advantage as it is inexpensive compared to precious metal catalysts. Furthermore, it eliminates the need for complex heavy metal removal steps, simplifying the purification process and ensuring higher purity standards suitable for pharmaceutical applications.
Q: Does this synthesis method support a wide range of substrate substituents?
A: Yes, the method demonstrates excellent substrate universality. It successfully accommodates various substituted 2-hydroxyacetophenones and aldehydes, including those with electron-withdrawing groups like halogens and trifluoromethyl groups, as well as electron-donating groups like methyl and methoxy groups.
Q: How does the reaction time compare to previously reported aniline-mediated methods?
A: The PPA-catalyzed method significantly reduces reaction time. While traditional aniline-mediated cyclizations often require 7.5 to 24 hours, this novel approach achieves high yields within a shorter 7 to 8-hour reflux period, enhancing overall production throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flavanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires a partner with deep technical expertise and robust manufacturing infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in small-scale trials are faithfully reproduced at an industrial level. We are committed to delivering high-purity flavanone intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex chemistries, such as the PPA-catalyzed cyclization described here, allows us to offer flexible manufacturing solutions tailored to the unique needs of global pharmaceutical clients.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, enabling you to make informed decisions that accelerate your drug development programs while optimizing your manufacturing budget.
