Optimizing Candesartan Production: A Novel Ethanol-Based Protection Strategy for Commercial Scale
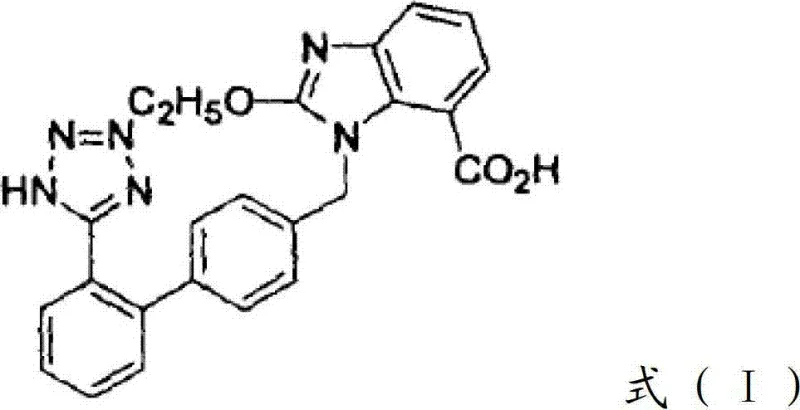
The global demand for angiotensin II receptor blockers (ARBs) continues to surge as hypertension remains a critical public health challenge worldwide. Within this therapeutic class, Candesartan stands out for its long-acting efficacy and superior binding affinity. However, traditional synthetic routes often suffer from complex protection-deprotection sequences that inflate manufacturing costs and extend lead times. A pivotal breakthrough in this domain is documented in patent CN102391254B, which discloses a preparation method of Candesartan that fundamentally reimagines the alkylation of active nitrogen atoms on the benzene ring. By strategically adopting ethanol to replace tert-butyl alcohol, this novel approach enables ethoxycarbonyl groups to function as robust protective groups. This innovation allows the product to undergo nucleophilic substitution and reduction reactions, followed directly by a cyclization reaction to generate the key intermediate 1-[(2'-cyanobiphenyl-4-yl) methyl]-2-ethoxy-1H-benzimidazole-7-ethyl formate. Crucially, this method eliminates the need for removing protective groups via hydrolysis before cyclization and avoids the use of expensive tetraethyl orthoformate. For a reliable API intermediate supplier, understanding this mechanistic shift is vital for optimizing supply chains and delivering high-purity Candesartan to the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthetic route of Candesartan has relied heavily on methodologies that introduce significant inefficiencies into the production lifecycle. A predominant conventional pathway utilizes 3-nitrophthalic acid as a raw material to create intermediates through mono-esterification, azidation, and amidation. However, the critical bottleneck arises during the alkylation phase, where tert-butyl alcohol is typically employed to introduce a protecting group at the nitrogen atom. Following the nucleophilic substitution reaction, manufacturers are forced to execute an additional step to slough off this protecting group before the nitrogen atom can react with orthoformic acid tetra-ethyl ester for ring closure. This introduce-protecting-group-slough-protecting-group-carry-out-ring-closure reaction mode not only prolongs the production cycle substantially but also increases the consumption of raw materials. Furthermore, the reliance on specific deprotection reagents and the subsequent purification steps required to remove byproducts from these extra reactions inevitably reduce the overall productive rate of the end product. For procurement managers, these inefficiencies translate directly into higher operational expenditures and increased vulnerability to supply chain disruptions caused by extended processing times.
The Novel Approach
In order to overcome these defectives, the present invention improves the operational path by introducing a more efficient Candesartan preparation method that streamlines the synthetic sequence. The core innovation lies in the substitution of tert-butyl alcohol with ethanol during the alkylation reaction in step 3 of the process. This simple change allows the 'ethoxycarbonyl' group to act as the protecting group on the active nitrogen atom, which possesses unique stability characteristics. After the nucleophilic substitution reaction of step 4 and the reduction reaction of step 5, the product can directly undergo a cyclization reaction to generate the target intermediate without prior deprotection. Otherwise, the 'ethoxycarbonyl' protecting group on the nitrogen atom can directly serve as the cyclization raw material, meaning there is no need to re-use expensive orthoformic acid tetra-ethyl ester. Therefore, adopting method of the present invention to prepare Candesartan can shorten the production cycle, reduce cost, and improve the productive rate of the end product. This approach represents a significant leap forward in cost reduction in pharmaceutical manufacturing, offering a leaner, more agile production model for complex API intermediates.
Mechanistic Insights into Ethanol-Mediated N-Alkylation and Cyclization
The mechanistic elegance of this patent lies in the dual functionality of the ethoxycarbonyl group introduced via ethanol. In step 3, 3-nitro-2-formyl triazobenzene ethyl formate reacts with ethanol at temperatures between 60°C and 80°C to generate 2-ethoxycarbonyl amino-3-ethyl nitrobenzoate. Unlike bulky tert-butyl groups, the ethoxycarbonyl moiety provides sufficient steric and electronic protection to the nitrogen atom during the subsequent nucleophilic substitution with 4'-bromomethyl-2'-cyanobiphenyl in step 4. This reaction is conducted in a heterogeneous system using phase-transfer catalysts like tetrabutyl ammonium bromide, which enhances the solubility of the basic catalyst and reduces its consumption. Following this, step 5 utilizes a hydrazine hydrate-activated carbon system to reduce the nitro group to an amine at 60°C to 80°C. The critical advantage is that the ethoxycarbonyl group remains intact during this reduction, unlike other protecting groups that might be labile under reducing conditions. This stability allows the molecule to proceed directly to step 6, where glacial acetic acid catalyzes the intramolecular cyclization. The ethoxycarbonyl group participates in the ring closure, effectively becoming part of the benzimidazole core structure's substitution pattern, thereby eliminating the need for a separate deprotection hydrolysis step that would otherwise be required to expose the nitrogen for cyclization.
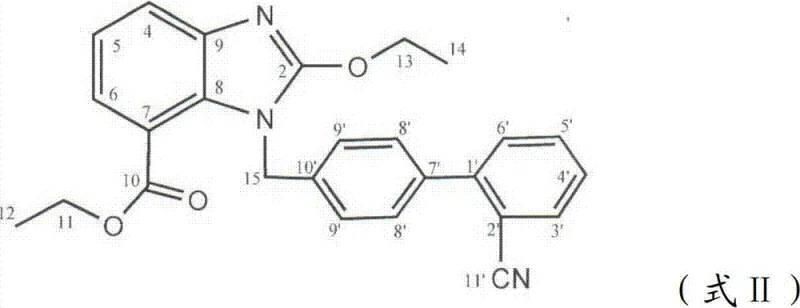
Impurity control is another critical aspect where this novel mechanism offers distinct advantages over traditional routes. In conventional synthesis, the multiple steps of protection and deprotection often generate complex byproduct profiles, including partially deprotected species or over-alkylated impurities that are difficult to separate. By contrast, the streamlined pathway in patent CN102391254B minimizes the number of chemical transformations, thereby inherently reducing the opportunity for side reactions to occur. The use of a heterogeneous reaction system in step 4 further aids in impurity control by facilitating easier phase separation and washing of inorganic salts and catalyst residues. Additionally, the specific reduction conditions using hydrazine hydrate and activated carbon are highly selective for the nitro group, minimizing the risk of reducing other sensitive functionalities such as the nitrile group on the biphenyl ring. The final cyclization step in acetone or ethyl acetate with glacial acetic acid is also highly specific, driving the equilibrium towards the desired benzimidazole formation. For R&D directors focused on purity and impurity profiles, this mechanism ensures a cleaner crude product, which simplifies downstream purification and ensures that the high-purity Candesartan meets stringent regulatory specifications for commercial scale-up of complex API intermediates.
How to Synthesize Candesartan Efficiently
The synthesis of Candesartan via this improved method involves a sequence of eight distinct steps that transform simple starting materials into the complex final API. The process begins with the conversion of 2-carboxyl-3-ethyl nitrobenzoate into its acyl chloride derivative using thionyl chloride, followed by azidation to form the acyl azide. The pivotal step involves the reaction with ethanol to install the ethoxycarbonyl protecting group, which sets the stage for the subsequent coupling with the biphenyl fragment. The detailed standardized synthesis steps involve precise control of temperatures, such as maintaining below 20°C during azidation and heating to 60°C-80°C for reduction and cyclization. Solvent selection is also critical, utilizing dichloromethane, ethanol, and acetone to optimize solubility and reaction kinetics at each stage. The detailed standardized synthesis steps are outlined in the guide below, providing a roadmap for technical teams to replicate this high-efficiency route in a pilot or commercial setting.
- Acyl chloride formation from 2-carboxyl-3-ethyl nitrobenzoate using thionyl chloride.
- Azide reaction followed by ethanol alkylation to form the ethoxycarbonyl protected intermediate.
- Nucleophilic substitution, nitro reduction, and direct cyclization without deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical improvements detailed in this patent translate directly into tangible commercial benefits that enhance the bottom line. The primary advantage is the significant reduction in manufacturing costs driven by the elimination of entire reaction steps. By removing the need for a deprotection hydrolysis step and avoiding the use of expensive cyclization reagents like tetraethyl orthoformate, the overall material cost per kilogram of product is drastically lowered. Furthermore, the shortened production cycle means that manufacturing assets can be turned over more quickly, increasing overall plant capacity without the need for capital expansion. This efficiency is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more rapidly to market demand fluctuations. The use of common, inexpensive solvents and catalysts further insulates the production cost from volatility in the pricing of specialty chemicals, ensuring stable pricing for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of the deprotection step and the replacement of expensive reagents with ethanol result in substantial cost savings. Ethanol is a commodity chemical with a stable supply chain, unlike specialized protecting group reagents. Additionally, the reduction in step count lowers labor costs, energy consumption for heating and cooling, and waste disposal fees associated with processing fewer reaction batches. The heterogeneous system used in the nucleophilic substitution also reduces the consumption of basic catalysts and phase-transfer catalysts, which are often costly inputs. These factors combine to create a highly cost-competitive manufacturing process that offers significant margins for both the producer and the buyer.
- Enhanced Supply Chain Reliability: A shorter synthetic route inherently reduces the risk of supply chain disruptions. With fewer steps, there are fewer points of failure where a reaction might stall or yield poor quality material, ensuring a more consistent flow of product. The raw materials required, such as ethanol, thionyl chloride, and hydrazine hydrate, are widely available globally, reducing the risk of raw material shortages. The robustness of the heterogeneous reaction system also means that the process is less sensitive to minor variations in raw material quality, further enhancing reliability. For supply chain heads, this translates to a dependable source of high-purity Candesartan that can support continuous commercial production without unexpected stoppages.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions and solvents that are easily managed in large-scale reactors. The avoidance of hazardous or difficult-to-handle reagents simplifies the safety profile of the plant, reducing the regulatory burden and insurance costs. Furthermore, the reduction in waste generation due to fewer steps and higher yields contributes to a smaller environmental footprint. The ability to recycle solvents like dichloromethane and acetone further enhances the sustainability of the process. This alignment with green chemistry principles not only meets current environmental compliance standards but also future-proofs the supply chain against tightening regulations, making it a sustainable choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Candesartan synthesis route. These answers are derived directly from the experimental data and technical specifications provided in patent CN102391254B, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this method for commercial production. The insights provided here cover aspects of cost, purity, and scalability, offering a comprehensive overview of the technology's value proposition.
Q: How does the ethanol-based protection method reduce production costs?
A: By using ethanol instead of tert-butyl alcohol, the ethoxycarbonyl group serves as a protecting group that does not require a separate hydrolysis step before cyclization. This eliminates the need for expensive reagents like tetraethyl orthoformate and reduces overall reaction time.
Q: What are the purity specifications for the intermediate Compound 7?
A: According to the patent data, Compound 7 can be achieved with a purity of approximately 97% and a yield of 85% using the described acetone and glacial acetic acid cyclization conditions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes heterogeneous reaction systems and common solvents like ethanol and dichloromethane, which are easily managed on an industrial scale. The reduction in steps significantly enhances scalability and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Candesartan Supplier
The technical potential of the ethanol-based protection strategy for Candesartan synthesis is immense, offering a pathway to more efficient and cost-effective production of this critical antihypertensive agent. NINGBO INNO PHARMCHEM, as a leading CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the global market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of Candesartan or its intermediates meets the highest international standards. We understand the complexities of translating patent chemistry into robust industrial processes and are committed to delivering consistent quality and reliability to our partners.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments for Candesartan and related intermediates. Let us help you secure a stable, high-quality supply of this essential medication while maximizing your operational efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
