Revolutionizing Candesartan Intermediate Production via Safe One-Pot Curtius Rearrangement
Revolutionizing Candesartan Intermediate Production via Safe One-Pot Curtius Rearrangement
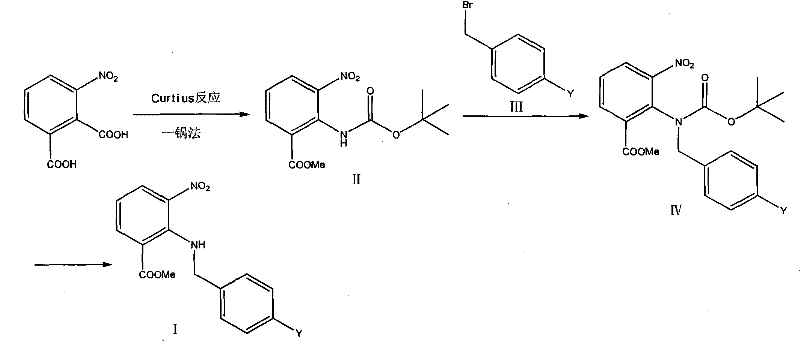
The pharmaceutical industry constantly seeks robust manufacturing pathways for critical antihypertensive agents, and patent CN101880241A presents a transformative approach to synthesizing key Candesartan Cilexetil intermediates. This intellectual property details a highly efficient one-pot methodology for preparing 2-(substituted phenyl) methylamino-3-nitrobenzoic acid methyl esters, specifically targeting the 4-bromo and 2'-cyano-biphenyl variants essential for Angiotensin II receptor antagonist production. By integrating the Curtius rearrangement, alkylation, and deprotection into a continuous sequence, this technology addresses longstanding challenges regarding safety, environmental impact, and operational complexity inherent in legacy synthetic routes. For R&D directors and process chemists, this represents a significant leap forward in minimizing hazardous unit operations while maximizing throughput efficiency in a single reactor vessel.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these vital intermediates has relied on fragmented, multi-step processes that introduce substantial inefficiencies and safety hazards into the supply chain. As illustrated in prior art such as patent CN101068807A, the conventional pathway necessitates the isolation of unstable acyl azide intermediates, requiring dangerous concentration and drying steps that pose severe explosion risks in a commercial setting. Furthermore, these legacy methods often employ toxic solvents like benzene for acylation reactions and involve extensive work-up procedures between each transformation, leading to significant accumulation of solvent waste and increased labor intensity. The cumulative yield losses associated with repeated extraction, purification, and drying stages not only inflate the cost of goods sold but also create bottlenecks that hinder the ability to scale production rapidly to meet global demand for cardiovascular medications.
The Novel Approach
In stark contrast, the methodology disclosed in CN101880241A utilizes a seamless one-pot strategy that maintains the reactive acyl azide species in solution, thereby completely eliminating the need for hazardous isolation and drying protocols. This continuous process begins with the activation of 3-nitro-2-carboxymethyl benzoate using thionyl chloride, followed immediately by azidation and rearrangement in the presence of tert-butanol to generate the Boc-protected amine in situ. By avoiding intermediate isolation, the process drastically reduces solvent consumption and minimizes the exposure of personnel to potentially explosive azide compounds, creating a inherently safer manufacturing environment. The subsequent alkylation and deprotection steps are performed sequentially in the same reaction matrix, streamlining the workflow and ensuring that the valuable intermediate is preserved with minimal degradation or mechanical loss during transfer operations.
Mechanistic Insights into One-Pot Curtius Rearrangement and Alkylation
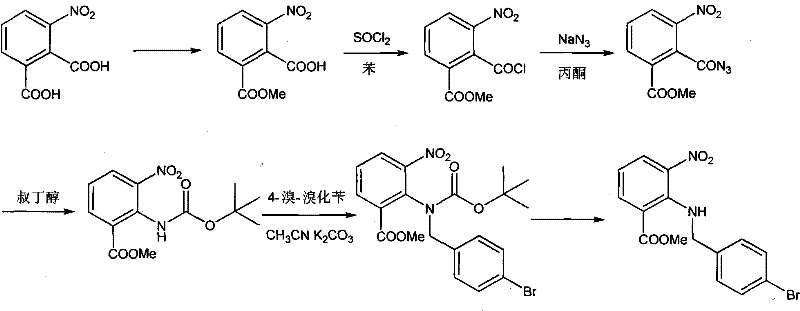
The core chemical innovation lies in the precise orchestration of the Curtius rearrangement within a heterogeneous phase-transfer catalysis system. The reaction initiates with the conversion of the carboxylic acid to an acid chloride using thionyl chloride, activated by a catalytic amount of dimethylformamide (DMF) to enhance electrophilicity. Upon addition of sodium azide aqueous solution and a phase transfer catalyst such as Tetrabutyl ammonium bromide, the acyl azide is generated at the interface and immediately undergoes thermal rearrangement to the isocyanate when treated with tert-butanol at elevated temperatures (65-70°C). This in-situ trapping of the isocyanate by tert-butanol forms the stable tert-butoxycarbonyl (Boc) protected amine, which serves as the pivotal intermediate for the subsequent nucleophilic substitution. The use of phase transfer catalysts is critical here, as they facilitate the transport of the azide anion into the organic phase, accelerating the reaction kinetics and ensuring high conversion rates without the need for excessive heating that could decompose sensitive nitro groups.
Following the formation of the Boc-protected species, the system transitions directly to the alkylation phase where substituted benzyl bromides react with the secondary amine nitrogen under basic conditions. The presence of strong bases like sodium hydroxide or potassium carbonate deprotonates the amine, rendering it nucleophilic enough to displace the bromide leaving group on the benzyl substrate, whether it be 4-bromobenzyl bromide or 4-bromomethyl-2'-cyanobiphenyl. The final step involves the removal of the Boc protecting group using hydrohalic acids such as hydrochloric acid or hydrogen bromide in alcoholic or ester solvents. This acidolysis proceeds cleanly to yield the free amine salt, which is then neutralized to precipitate the final high-purity crystalline product. The entire sequence is designed to maintain the integrity of the nitro group and the ester functionality while efficiently constructing the complex carbon-nitrogen framework required for the final API.
How to Synthesize 2-(Substituted Phenyl) Methylamino-3-Nitrobenzoic Acid Methyl Ester Efficiently
Implementing this one-pot synthesis requires careful control of reaction parameters, particularly temperature and pH, to manage the exothermic nature of the azidation and the sensitivity of the intermediates. The process begins by charging 3-nitro-2-carboxymethyl benzoate into a reactor with DMF and chloroform, followed by the controlled addition of thionyl chloride to generate the acid chloride. Once the activation is complete, the temperature is lowered to below 0°C before introducing the sodium azide solution to prevent runaway reactions, followed by the addition of tert-butanol for the rearrangement step. After the Boc-protection is confirmed, the alkylating agent and phase transfer catalyst are introduced, and the mixture is stirred under basic conditions to effect the coupling.
- React 3-nitro-2-carboxymethyl benzoate with thionyl chloride and DMF to form the acid chloride in situ.
- Perform azidation using sodium azide and phase transfer catalyst, followed immediately by rearrangement with tert-butanol.
- Conduct alkylation with substituted benzyl bromide and subsequent deprotection using hydrohalic acid to yield the final intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this one-pot technology translates into tangible strategic benefits that extend far beyond simple yield metrics. By consolidating multiple reaction steps into a single vessel, manufacturers can significantly reduce the capital expenditure required for reactor trains, as fewer units are needed to achieve the same annual output volume. The elimination of intermediate isolation steps means that solvent recovery systems can be optimized for a smaller variety of waste streams, lowering the operational costs associated with distillation and waste treatment facilities. Furthermore, the reduction in manual handling and transfer operations decreases the likelihood of human error and cross-contamination, leading to more consistent batch-to-bquality and reducing the risk of costly production delays caused by out-of-specification results.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive drying equipment and the energy-intensive operations associated with isolating solid intermediates, resulting in substantial utility savings. By avoiding the use of highly toxic solvents like benzene in favor of safer alternatives like chloroform and esters, the facility reduces its liability and compliance costs related to environmental health and safety regulations. The higher overall yield achieved through the prevention of mechanical losses during filtration and drying directly improves the margin per kilogram of produced intermediate, allowing for more competitive pricing in the global market for Candesartan precursors.
- Enhanced Supply Chain Reliability: The inherent safety of keeping energetic azide compounds in solution removes a major bottleneck often caused by strict safety protocols surrounding the handling of dry azides. This allows for faster campaign turnover times and more flexible scheduling, ensuring that customers receive their orders with greater predictability and shorter lead times. Additionally, the use of readily available and commodity-grade reagents such as thionyl chloride and sodium azide ensures that raw material sourcing remains stable and unaffected by niche supply constraints that might plague more exotic catalysts or reagents used in alternative synthetic routes.
- Scalability and Environmental Compliance: The one-pot nature of the reaction simplifies the scale-up process from pilot plant to commercial production, as the heat transfer and mixing dynamics are easier to manage in a single large reactor compared to a series of smaller vessels. The reduction in total solvent volume and the avoidance of hazardous waste generation align perfectly with modern green chemistry principles, facilitating easier permitting and regulatory approval in jurisdictions with stringent environmental standards. This sustainability profile enhances the brand reputation of the manufacturer as a responsible partner, increasingly important for multinational pharmaceutical companies auditing their supply chains for ESG compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Candesartan intermediates. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of the operational realities and advantages. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this technology for their own manufacturing portfolios or for procurement specialists assessing the quality and reliability of suppliers utilizing this method.
Q: How does the one-pot method improve safety compared to traditional routes?
A: The traditional method requires isolating and drying unstable acyl azides, which poses explosion risks. The patented one-pot method keeps the azide in solution, eliminating the drying step and significantly enhancing operational safety.
Q: What are the key yield improvements observed in this synthesis?
A: Experimental data from the patent indicates yields ranging from 60% to 64% for the final intermediates, achieved through reduced material loss during purification steps that are eliminated in the continuous process.
Q: Which phase transfer catalysts are compatible with this reaction system?
A: The process demonstrates versatility with various catalysts including Tetrabutyl ammonium bromide, benzyltriethylammonium chloride, and crown ethers like 18-crown-6, allowing for flexible optimization based on cost and availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Substituted Phenyl) Methylamino-3-Nitrobenzoic Acid Methyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and safe synthetic pathways in the production of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Candesartan intermediate meets the exacting standards required by global regulatory bodies, providing our partners with absolute confidence in the quality of their supply chain.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized processes can reduce your overall cost of goods. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production timelines, ensuring that your development programs proceed without interruption.
