Advanced Manufacturing of Chiral Oxazolidinone Intermediates for Next-Generation Antibiotics
Advanced Manufacturing of Chiral Oxazolidinone Intermediates for Next-Generation Antibiotics
The pharmaceutical industry's relentless pursuit of potent antibiotics against multidrug-resistant Gram-positive bacteria has placed oxazolidinones, such as linezolid and tedizolid, at the forefront of therapeutic innovation. Central to the commercial viability of these life-saving drugs is the efficient and scalable production of their key chiral intermediates. Patent CN108430999B discloses a groundbreaking preparation method for oxazolidinone intermediates that addresses critical bottlenecks in prior art, specifically regarding yield, purity, and chirality control. This technical insight report analyzes the novel synthetic route which transforms simple starting materials like epichlorohydrin into high-value oxazolidinone cores through a robust sequence of alkylation, ring-opening, and cyclization reactions. By leveraging phase transfer catalysis and mild hydrogenation conditions, this process offers a reliable oxazolidinone intermediate supplier pathway that is uniquely suited for industrial scale-up.
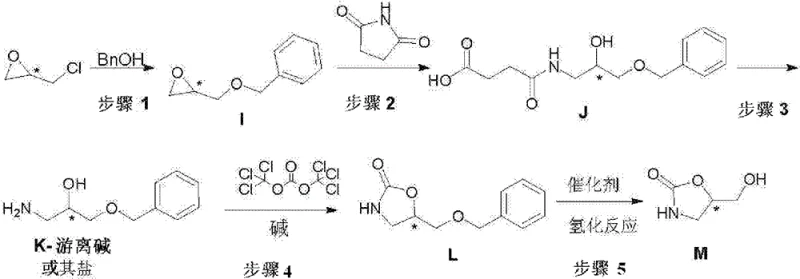
The disclosed technology represents a significant paradigm shift from traditional synthesis strategies that often rely on hazardous reagents or complex purification protocols. The core innovation lies in the strategic construction of the carbon-nitrogen backbone followed by a gentle cyclization step that preserves stereochemical integrity. Unlike older methods that struggle with low conversion rates or difficult-to-remove metal contaminants, this approach utilizes triphosgene for ring closure and palladium-catalyzed hydrogenation for deprotection. This combination not only streamlines the workflow but also ensures that the final product meets the stringent purity specifications required for active pharmaceutical ingredient (API) manufacturing. For procurement and supply chain leaders, understanding this mechanistic advantage is crucial for securing a stable supply of high-purity oxazolidinone intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxazolidinone scaffolds has been plagued by inefficiencies that hinder large-scale production. Early literature describes the cyclization of 3-amino-1,2-propanediol with diethyl carbonate, a method characterized by sluggish reaction kinetics and disappointingly low yields that make it economically unviable for commercial operations. Furthermore, alternative routes involving the reduction of carboxylic acid esters with sodium borohydride or the oxidative deprotection using cerium ammonium nitrate (CAN) present severe drawbacks. The use of CAN, in particular, introduces expensive heavy metal contaminants that are notoriously difficult to remove to trace levels, posing significant risks to patient safety and regulatory compliance. These legacy processes often require multiple isolation steps, excessive solvent usage, and result in products with inconsistent chiral purity, creating substantial barriers for cost reduction in pharmaceutical manufacturing.
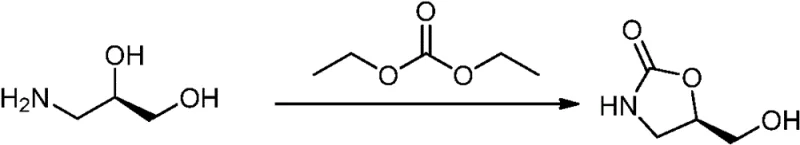
The Novel Approach
In stark contrast, the method detailed in CN108430999B introduces a streamlined, high-efficiency route that circumvents these historical pitfalls. By initiating the synthesis with the alkylation of benzyl alcohol and epichlorohydrin, the process establishes the chiral center early using readily available enantiopure starting materials. The subsequent nucleophilic attack by succinimide and hydrolysis generates a stable amino-alcohol intermediate that serves as a versatile building block. The true brilliance of this approach is the cyclization step using triphosgene under biphasic conditions, which proceeds rapidly with high conversion. Finally, the removal of the benzyl protecting group via catalytic hydrogenation replaces the harsh chemical oxidation of the past with a clean, atom-economical reduction. This modern strategy not only boosts overall yield but also simplifies the impurity profile, making it an ideal solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Triphosgene-Mediated Cyclization and Hydrogenation
The chemical elegance of this synthesis is best understood through the lens of its key transformation steps, particularly the formation of the oxazolidinone ring and the subsequent deprotection. The cyclization of the amino-alcohol intermediate (Compound K) with triphosgene acts as a phosgene equivalent, activating the hydroxyl group to facilitate intramolecular nucleophilic attack by the amine. This reaction is meticulously controlled by maintaining temperatures between 0°C and 20°C in a dichloromethane-water biphasic system buffered by sodium carbonate. This specific condition set is critical for suppressing side reactions such as over-chlorination or polymerization, ensuring that the carbamate linkage forms selectively. The preservation of the chiral center during this step is paramount; the mild basic conditions prevent racemization at the alpha-carbon, thereby locking in the optical purity derived from the initial epichlorohydrin feedstock.
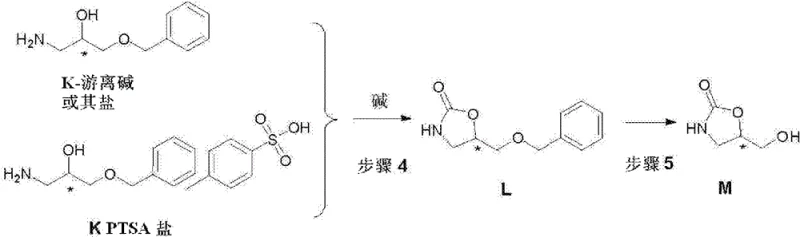
Following cyclization, the removal of the benzyl ether protecting group is achieved through catalytic hydrogenation, a process that exemplifies green chemistry principles in fine chemical synthesis. Utilizing catalysts such as Pd(OH)2/C or Pd/C in solvents like methanol or tetrahydrofuran, the benzyl group is cleaved under hydrogen pressure at moderate temperatures of 40-60°C. This step is mechanistically distinct from oxidative deprotection methods as it avoids the generation of toxic byproducts or acidic waste streams associated with cerium reagents. The heterogeneous nature of the catalyst allows for easy filtration and recovery, further enhancing the process's environmental profile. For R&D directors, this mechanism offers a clear path to controlling the impurity spectrum, as the primary byproduct is toluene, which is easily removed during concentration, resulting in a final product with exceptional chemical and optical purity suitable for downstream coupling reactions.
How to Synthesize (R)-5-(hydroxymethyl)oxazolidin-2-one Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and maintain stereochemical fidelity. The process begins with the phase-transfer catalyzed alkylation of benzyl alcohol, followed by ring-opening with succinimide and hydrolysis to generate the key amino-alcohol precursor. This precursor is then subjected to cyclization with triphosgene in a biphasic system, followed immediately by catalytic hydrogenation to reveal the final hydroxymethyl group. The patent highlights the feasibility of a 'one-pot' strategy where intermediates are not isolated, drastically reducing unit operations. Detailed standardized synthesis steps, including specific reagent ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during technology transfer.
- React epoxy chloropropane with benzyl alcohol using a phase transfer catalyst and alkali to form the epoxide intermediate.
- Perform nucleophilic ring opening with succinimide under alkaline conditions to generate the hydroxy-amide derivative.
- Hydrolyze the amide bond under strong base conditions to release the free amino-alcohol base or its salt.
- Cyclize the amino-alcohol with triphosgene in the presence of base to form the benzyl-protected oxazolidinone.
- Execute catalytic hydrogenation using Pd(OH)2/C to remove the benzyl protecting group and yield the final hydroxymethyl oxazolidinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates directly into tangible operational benefits and risk mitigation. The shift away from expensive and regulated reagents like cerium ammonium nitrate towards commodity chemicals like triphosgene and hydrogen gas fundamentally alters the cost structure of the intermediate. Additionally, the ability to run telescoped reactions without isolating unstable intermediates reduces the footprint of the manufacturing facility and shortens the production cycle time. These factors combine to create a supply chain that is more resilient to raw material fluctuations and regulatory changes, ensuring a consistent flow of critical materials for antibiotic production.
- Cost Reduction in Manufacturing: The elimination of high-cost oxidants and the use of catalytic hydrogenation significantly lower the direct material costs associated with production. By avoiding the need for extensive purification steps to remove heavy metal residues, the process reduces both consumable expenses and waste disposal fees. Furthermore, the high yields reported in the patent examples mean that less starting material is required per kilogram of finished product, driving down the overall cost of goods sold substantially.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as epichlorohydrin, benzyl alcohol, and succinimide mitigates the risk of supply disruptions common with specialty reagents. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry, ensures high batch-to-batch consistency. This reliability is critical for maintaining continuous API production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like liquid-liquid extraction and filtration that are easily adapted from pilot plant to multi-ton reactors. The replacement of stoichiometric oxidants with catalytic hydrogenation aligns with modern environmental, health, and safety (EHS) standards by minimizing hazardous waste generation. This green chemistry approach simplifies regulatory filings and supports the sustainability goals of forward-thinking pharmaceutical manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazolidinone synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on process capabilities and quality attributes. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How is chirality controlled in this oxazolidinone synthesis?
A: Chirality is strictly controlled by the starting material. Using enantiomerically pure (R)-epichlorohydrin ensures the final product maintains the corresponding (R)-configuration throughout the synthesis without racemization.
Q: Can the intermediates be processed without isolation?
A: Yes, the patent describes a 'one-pot' methodology where compounds I, J, or L can be reacted sequentially without isolation, significantly reducing processing time and solvent consumption.
Q: What are the advantages over traditional cerium ammonium nitrate methods?
A: This method avoids the use of expensive and difficult-to-remove cerium ammonium nitrate. Instead, it utilizes catalytic hydrogenation for deprotection, which is more cost-effective, environmentally friendly, and easier to purify.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazolidinone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capacity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in patent examples are replicated reliably at an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of oxazolidinone intermediate meets the highest global standards for chirality and chemical purity.
We invite you to collaborate with us to optimize your supply chain for next-generation antibiotics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and security into your pharmaceutical projects.
