Revolutionizing Linezolid Production: A Safe, Scalable Route for Global Pharmaceutical Supply Chains
The global demand for effective antimicrobial agents continues to drive innovation in pharmaceutical manufacturing, particularly for critical antibiotics like Linezolid. Patent CN103391927A introduces a transformative preparation method for Linezolid and its key oxazolidinone intermediates, addressing long-standing challenges in cost, safety, and purity. This technical insight report analyzes the proprietary synthetic route disclosed in the patent, highlighting its potential to redefine supply chain reliability for reliable pharmaceutical intermediate suppliers. By shifting away from hazardous cryogenic chemistry towards a more robust, ambient-temperature process, this methodology offers a compelling value proposition for procurement teams seeking cost reduction in API manufacturing while maintaining stringent quality standards.
The significance of this patent lies in its ability to bypass the limitations of prior art, specifically the reliance on dangerous reagents like n-butyllithium (n-BuLi) which require extreme low-temperature conditions. Instead, the invention leverages a sequence of well-controlled reactions involving R-epichlorohydrin and carbonyl diimidazole (CDI) to construct the chiral oxazolidinone core. For R&D directors and process chemists, this represents a shift towards more sustainable and scalable chemistry, minimizing the formation of difficult-to-remove isomers that plague conventional syntheses. The detailed mechanistic pathway ensures that the final active pharmaceutical ingredient meets the rigorous purity specifications required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Linezolid has been hindered by processes that are commercially unviable due to extreme operational constraints. As illustrated in the prior art schemes, traditional methods often necessitate the use of hazardous chemicals such as n-BuLi at temperatures as low as -78°C to generate essential hydroxy derivatives. Handling such reactive organometallic reagents on a large scale introduces significant safety risks and requires specialized, expensive cryogenic infrastructure. Furthermore, these legacy routes are prone to generating a complex mixture of undesired isomers and unidentified impurities. These contaminants are notoriously difficult to separate during downstream processing, leading to substantial yield losses and compromising the overall purity profile of the final drug substance. The persistence of these impurities not only affects efficacy but also complicates the regulatory approval process for generic manufacturers.
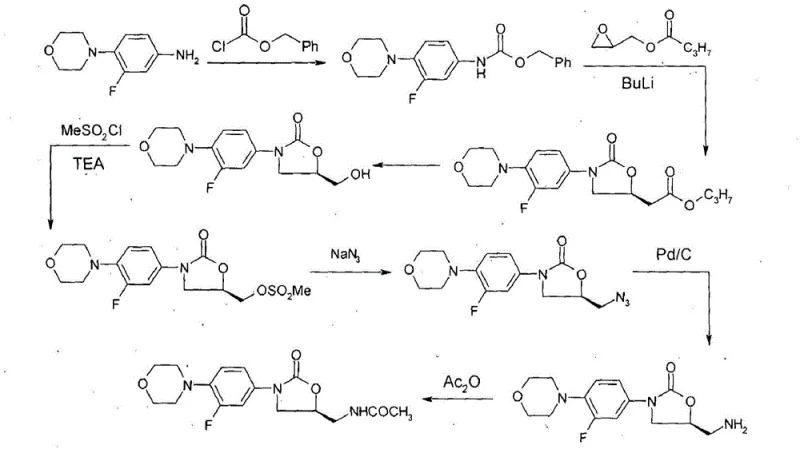
The Novel Approach
In stark contrast, the methodology described in CN103391927A offers a streamlined and safer alternative that eliminates the need for cryogenic conditions. The new route initiates with the nucleophilic substitution of 1,2-difluoro-4-nitrobenzene with morpholine, followed by a catalytic reduction to form 3-fluoro-4-morpholinoaniline. A key innovation is the reaction of this aniline with R-epichlorohydrin in tert-butanol at reflux temperatures, which establishes the chiral center without the need for dangerous lithiation. Subsequent carbonylation using CDI efficiently closes the oxazolidinone ring. This approach not only enhances operator safety but also significantly simplifies the purification workflow. By avoiding the formation of complex impurity profiles associated with low-temperature organometallic chemistry, the new method maximizes yield and ensures a cleaner reaction mass, directly translating to lower production costs and higher throughput for commercial scale-up of complex pharmaceutical intermediates.
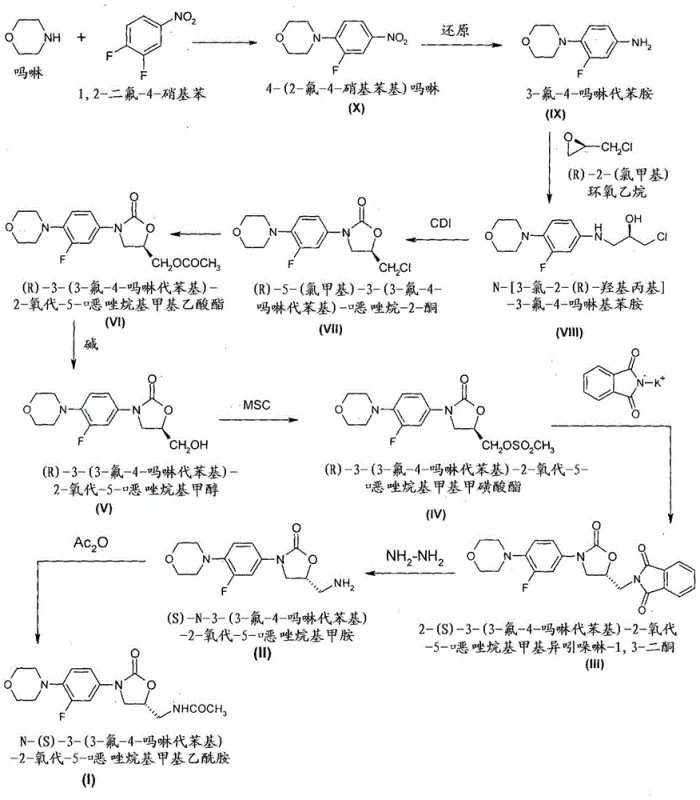
Mechanistic Insights into Chiral Oxazolidinone Construction
The core of this synthetic strategy revolves around the precise control of stereochemistry during the formation of the oxazolidinone ring. The process begins with the reaction of 3-fluoro-4-morpholinoaniline with R-epichlorohydrin. This step is critical as it sets the initial chirality. The subsequent cyclization using carbonyl diimidazole (CDI) proceeds under mild conditions to form the (5R)-5-(chloromethyl) oxazolidin-2-one derivative. The choice of solvent for isolation, specifically n-butyl acetate, is mechanistically significant as it promotes the crystallization of the desired isomer while leaving impurities in the mother liquor. This selective crystallization is a powerful tool for impurity control, ensuring that the intermediate entering the next stage possesses high optical purity. The use of CDI as a carbonyl source is advantageous over phosgene or triphosgene, offering a safer and more manageable reagent profile for large-scale operations.
Further down the synthetic tree, the conversion of the chloromethyl group to the aminomethyl functionality involves a classic Gabriel synthesis modification. The chloromethyl intermediate is first converted to a mesylate or directly displaced by potassium phthalimide. This nucleophilic substitution proceeds with inversion of configuration, converting the (R)-configuration at the side chain to the required (S)-configuration found in the final Linezolid molecule. The use of potassium phthalimide is strategic; it acts as a protected amine equivalent that prevents over-alkylation and allows for clean deprotection later using hydrazine hydrate. This multi-step sequence—acetylation, hydrolysis, mesylation, phthalimide substitution, and deprotection—demonstrates a robust approach to chiral pool synthesis, ensuring that the stereochemical integrity is maintained or inverted predictably at each stage, resulting in a high-purity final amine precursor ready for acetylation.
How to Synthesize Linezolid Efficiently
The synthesis of Linezolid via this patented route involves a logical sequence of ten distinct chemical transformations, starting from readily available commodity chemicals. The process is designed to be modular, allowing for the isolation and quality control of key intermediates such as the chloromethyl oxazolidinone and the phthalimide derivative. Detailed operational parameters, including specific temperature ranges (e.g., 120°C for acetylation, 40-45°C for hydrogenation) and solvent choices (DMF, t-butanol, ethyl acetate), are optimized to maximize yield and minimize byproduct formation. For process engineers looking to implement this technology, the following guide outlines the critical stages of the synthesis, emphasizing the conditions required for successful scale-up.
- Condense 1,2-difluoro-4-nitrobenzene with morpholine followed by catalytic hydrogenation to form 3-fluoro-4-morpholinoaniline.
- React the aniline derivative with R-epichlorohydrin and perform carbonylation using CDI to establish the oxazolidinone ring.
- Execute a sequence of acetylation, hydrolysis, mesylation, and phthalimide substitution to invert stereochemistry and finalize the amine precursor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible benefits that extend beyond simple chemistry. The primary advantage lies in the drastic simplification of the manufacturing infrastructure. By eliminating the requirement for cryogenic reactors capable of maintaining -78°C, facilities can utilize standard glass-lined or stainless steel reactors equipped with conventional heating and cooling systems. This reduction in capital expenditure and operational complexity directly contributes to cost reduction in pharmaceutical manufacturing. Furthermore, the avoidance of pyrophoric reagents like n-BuLi reduces the need for specialized handling protocols and safety containment systems, lowering insurance and compliance costs associated with hazardous material management.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive and hazardous reagents with cost-effective alternatives. The use of R-epichlorohydrin and sodium acetate instead of organolithium compounds significantly lowers raw material costs. Additionally, the improved yield resulting from reduced impurity formation means less waste and higher output per batch. The ability to crystallize intermediates effectively reduces the need for extensive chromatographic purification, which is often a bottleneck in terms of both time and solvent consumption. These factors combine to create a leaner, more cost-efficient production model that enhances margin potential for generic drug manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. 1,2-difluoro-4-nitrobenzene, morpholine, and epichlorohydrin are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—minimizes the risk of batch failures due to equipment malfunction or process deviations. This reliability ensures consistent delivery schedules, a critical factor for pharmaceutical companies managing just-in-time inventory systems. The simplified process flow also shortens the overall cycle time from raw material intake to finished intermediate, reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is superior. The solvents employed, such as ethyl acetate, methanol, and t-butanol, are easier to recover and recycle compared to the exotic solvents often required for cryogenic chemistry. The elimination of heavy metal catalysts (in favor of Raney Nickel which is easily filtered) and toxic reagents simplifies wastewater treatment and waste disposal, aligning with increasingly strict environmental regulations. The process is inherently scalable; the exothermic profiles are manageable, and the unit operations (filtration, distillation, crystallization) are standard in the fine chemical industry. This makes the technology transfer from pilot plant to multi-ton commercial production seamless and predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Linezolid synthesis route. These insights are derived directly from the technical specifications and comparative data provided in the patent documentation, offering clarity on the practical implications of adopting this new methodology.
Q: How does this new process improve safety compared to traditional Linezolid synthesis?
A: Traditional methods often rely on n-BuLi at cryogenic temperatures (-78°C), which poses significant safety risks. This patented route utilizes standard reagents like R-epichlorohydrin and operates at much milder temperatures (e.g., reflux in t-butanol), drastically reducing operational hazards.
Q: What are the key purity advantages of this synthetic route?
A: The process incorporates specific crystallization steps, such as isolating the chloromethyl oxazolidinone from n-butyl acetate, which effectively removes unidentified impurities and isomers that typically persist in older methods, ensuring high-purity intermediates.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the elimination of specialized cryogenic equipment and hazardous organolithium reagents makes this route highly scalable. It uses common solvents like DMF, methanol, and ethyl acetate, facilitating easier technology transfer to multi-ton manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linezolid Supplier
The technological advancements detailed in patent CN103391927A represent a significant leap forward in the production of oxazolidinone antibiotics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory processes into reliable commercial reality. As a premier CDMO partner, we possess the technical expertise and infrastructure to execute this complex synthetic pathway with precision. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel route—safety, purity, and cost-efficiency—are fully realized at an industrial scale. Our stringent purity specifications and rigorous QC labs guarantee that every batch of Linezolid intermediate meets the highest international standards.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only cost-competitive but also resilient and compliant. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Let us provide you with specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your long-term supply goals for this vital antibiotic.
