Advanced Quinazoline Derivatives: Transforming Oncology Intermediates Manufacturing and Supply
The pharmaceutical landscape is currently witnessing a transformative shift driven by the innovative disclosures within patent CN102452988A, which details a novel class of quinazoline derivatives designed to overcome longstanding limitations in oncology treatment. This specific intellectual property introduces a sophisticated molecular architecture that functions not merely as a passive inhibitor but as an active, multi-targeting therapeutic agent capable of coordinating with noble metals. For industry stakeholders, this represents a significant evolution from traditional cytotoxic chemotherapy towards highly selective molecular targeted therapy that maintains potent efficacy while mitigating severe systemic toxicity. The core innovation lies in the strategic modification of the quinazoline scaffold, a well-known pharmacophore in kinase inhibition, to include specific functional groups that enable coordination with cytotoxic metal complexes such as ruthenium or platinum. This dual-functionality approach addresses the critical bottleneck of drug resistance and poor solubility that has historically plagued the clinical application of many high-potency small molecule inhibitors. By integrating these advanced chemical features, the technology offers a robust pathway for developing next-generation anti-tumor compounds that can simultaneously inhibit protein kinases and interact with DNA, thereby providing a comprehensive therapeutic assault on malignant cells. As a reliable pharmaceutical intermediates supplier, understanding the depth of this chemical innovation is paramount for securing a competitive edge in the global oncology supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the clinical management of cancer has relied heavily on two distinct categories of chemical drugs: non-specific cytotoxic agents and early-generation molecular targeted therapies, both of which suffer from significant drawbacks that limit their therapeutic index and patient quality of life. Conventional cytotoxic drugs, such as cisplatin and other platinum-based complexes, operate by indiscriminately attacking rapidly dividing cells, which results in severe damage to healthy tissues and causes debilitating side effects that often force dose reductions or treatment discontinuation. Furthermore, tumor cells frequently develop innate or acquired resistance to these agents through various mechanisms, including enhanced DNA repair and drug efflux, rendering the treatment ineffective over time. On the other hand, while small molecule kinase inhibitors offered a more targeted approach, many of these compounds exhibit poor water solubility and high toxicity profiles that restrict their bioavailability and clinical utility. The inability to effectively deliver these potent molecules to the tumor site without causing off-target effects has been a persistent challenge in medicinal chemistry. Additionally, the emergence of resistance mutations in the kinase domain often leads to treatment failure, necessitating the continuous development of new analogs. These cumulative limitations highlight the urgent need for a new class of compounds that can bridge the gap between high potency and manageable safety profiles while overcoming the biological barriers of solubility and resistance.
The Novel Approach
The technology disclosed in patent CN102452988A presents a groundbreaking solution by engineering quinazoline derivatives that possess inherent metal-coordinating capabilities, effectively merging the benefits of targeted kinase inhibition with the cytotoxic power of metal complexes. This novel approach allows the quinazoline scaffold to act as a ligand that can replace one or more ligands in cytotoxic metal complexes, creating a single-molecule multi-target system that attacks cancer cells through synergistic mechanisms. By incorporating groups containing atoms capable of coordinating with noble metals, such as nitrogen, oxygen, or sulfur, into the R and R' positions of the quinazoline structure, the invention significantly enhances the water solubility of the resulting complexes. This improvement in physicochemical properties facilitates better pharmacokinetic profiles and ensures that therapeutic concentrations can be achieved at the tumor site with reduced systemic exposure. Moreover, the dual-action mechanism, which involves inhibiting tyrosine protein kinases like EGFR while simultaneously interacting with DNA via the metal center, drastically reduces the likelihood of tumor cells developing resistance. This strategic molecular design not only revitalizes high-activity kinase inhibitors that were previously discarded due to solubility issues but also lowers the required dosage of cytotoxic metal drugs, thereby minimizing their associated toxic side effects and offering a safer, more effective treatment paradigm for patients.
Mechanistic Insights into Quinazoline-Based Coordination Chemistry
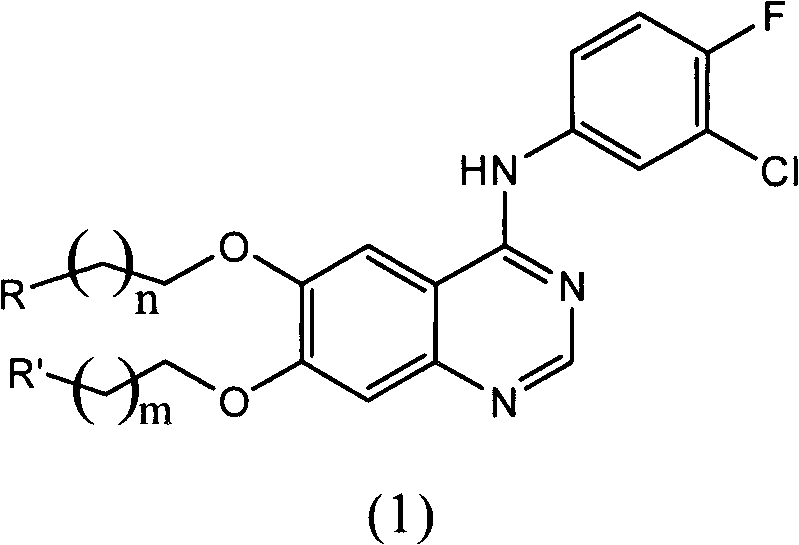
The chemical elegance of this invention lies in the precise structural modifications made to the core quinazoline ring system, which is a privileged structure in the field of tyrosine protein kinase inhibition. The general formula (1) described in the patent features a 4-anilinoquinazoline backbone, similar to known EGFR inhibitors, but is distinctively functionalized at the 6 and 7 positions with alkoxy chains terminating in metal-coordinating groups. These coordinating groups, which can include fused heterocyclic imines, aminoalkylimines, or aromatic heterocycles like pyridine and imidazole, provide the necessary electron donors to form stable coordination bonds with transition metals such as ruthenium (II, III) and platinum. The presence of these donor atoms creates a versatile chelating environment that stabilizes the metal center within the biological milieu, preventing premature decomposition and ensuring that the cytotoxic payload is delivered specifically to the target cells. The mechanism of action involves the quinazoline moiety binding to the ATP-binding pocket of the EGFR kinase domain, thereby blocking downstream signaling pathways that drive cell proliferation. Simultaneously, the coordinated metal complex can intercalate or bind to DNA, inducing strand breaks and triggering apoptosis through a pathway independent of kinase inhibition. This multi-target strategy is particularly effective against heterogeneous tumor populations where subclones may resist one mechanism but remain vulnerable to the other. The ability to fine-tune the length of the alkyl linkers (m and n integers from 0 to 5) allows chemists to optimize the spatial arrangement for optimal binding affinity and solubility, demonstrating a high degree of tunability in the drug design process.
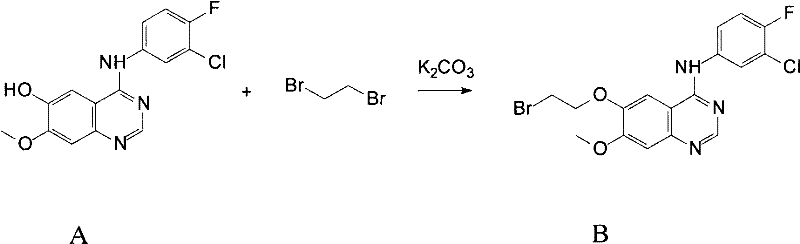
From a process chemistry perspective, the synthesis of these complex derivatives involves a series of well-controlled nucleophilic substitution and condensation reactions that ensure high purity and reproducibility. The preparation method typically begins with a first reactant A, which is a hydroxy-methoxy quinazoline derivative, undergoing alkylation with dihaloalkanes or halo-carboxylates to introduce the necessary linker arms. This step is critical as it establishes the foundation for the subsequent attachment of the metal-coordinating head groups. The reaction conditions are meticulously optimized, utilizing acid-binding agents like potassium carbonate or cesium carbonate in polar aprotic solvents such as DMF or acetonitrile to drive the reaction to completion while minimizing side products. Following the introduction of the linker, the terminal halide or ester functionality is converted into the desired amine or heterocyclic group through reflux conditions with specific organic amines. The use of phase transfer catalysts or specific bases like sodium hydroxide in mixed solvent systems further enhances the efficiency of these transformations. Each step is designed to maintain the integrity of the sensitive quinazoline core while successfully installing the complex side chains required for metal coordination. The final products are subjected to rigorous purification protocols, including column chromatography and recrystallization, to remove any residual starting materials or by-products, ensuring that the resulting intermediates meet the stringent quality standards required for pharmaceutical applications.
How to Synthesize Quinazoline Derivatives Efficiently
The efficient synthesis of these high-value quinazoline derivatives relies on a robust and scalable protocol that balances reaction yield with operational simplicity, making it highly suitable for industrial manufacturing environments. The process generally initiates with the activation of the phenolic hydroxyl group on the quinazoline core through nucleophilic substitution with dihaloalkanes, a reaction that proceeds smoothly under mild heating in the presence of inorganic bases. This step generates a reactive haloalkoxy intermediate that serves as a versatile electrophile for the subsequent introduction of various nitrogen-containing nucleophiles. The choice of solvent and temperature is critical; for instance, using DMF at temperatures between 70-90°C ensures rapid conversion while preventing degradation of the sensitive anilino linkage. Following the isolation of the intermediate, the second stage involves a condensation reaction with specialized organic amines that contain the metal-coordinating motifs, such as ethylenediamine or heterocyclic amines. This step often requires reflux conditions in acetonitrile or ethanol to overcome the activation energy barrier and achieve high conversion rates. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that guarantee consistent batch-to-batch quality.
- Prepare the first reactant A and react with dihaloalkane in the presence of an acid-binding agent to form intermediate B.
- React intermediate B with a first organic amine containing metal-coordinating atoms under reflux conditions to introduce the functional side chain.
- Purify the final condensation product through crystallization or column chromatography to ensure stringent purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits that extend beyond mere technical feasibility, directly impacting cost structures and supply reliability. The manufacturing process utilizes readily available starting materials and common organic solvents, which significantly reduces the dependency on exotic or scarce reagents that often cause supply chain bottlenecks. By eliminating the need for complex transition metal catalysts in the initial synthetic steps and relying instead on stoichiometric base-mediated reactions, the process simplifies the purification workflow and reduces the burden on waste treatment facilities. This streamlined approach translates into significant cost savings in manufacturing, as it lowers the consumption of expensive catalysts and minimizes the number of purification cycles required to achieve pharmaceutical-grade purity. Furthermore, the high solubility of the final derivatives reduces the need for complex formulation strategies downstream, potentially lowering the overall cost of goods for the finished drug product. The robustness of the reaction conditions also implies a lower risk of batch failures, ensuring a more predictable production schedule and enhancing supply chain reliability for long-term contracts.
- Cost Reduction in Manufacturing: The synthesis route avoids the use of expensive noble metal catalysts during the intermediate formation stages, relying instead on cost-effective inorganic bases and common organic solvents that are easily sourced in bulk quantities. This fundamental shift in reagent selection drastically simplifies the raw material procurement process and reduces the overall material costs associated with production. Additionally, the high selectivity of the reactions minimizes the formation of difficult-to-remove impurities, which reduces the consumption of silica gel and solvents during the purification phase. Consequently, the operational expenditure related to waste disposal and solvent recovery is substantially lowered, contributing to a more lean and efficient manufacturing model. The elimination of complex catalytic systems also reduces the capital investment required for specialized reactor linings or handling equipment, further enhancing the economic viability of large-scale production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted quinazolines and simple dihaloalkanes, are commodity chemicals with established global supply networks, ensuring consistent availability even during market fluctuations. This reliance on mature supply chains mitigates the risk of production delays caused by raw material shortages, which is a common concern with highly specialized or custom-synthesized building blocks. The modular nature of the synthesis allows for flexibility in sourcing; if one supplier of a specific amine becomes unavailable, alternative suppliers can be qualified with minimal process re-validation due to the robustness of the reaction conditions. This flexibility strengthens the resilience of the supply chain against geopolitical or logistical disruptions. Moreover, the stability of the intermediates allows for stockpiling if necessary, providing an additional buffer against unexpected demand surges or transportation delays.
- Scalability and Environmental Compliance: The reaction steps involved in this process are inherently scalable, utilizing standard unit operations such as reflux, filtration, and crystallization that are easily transferred from laboratory to pilot and commercial scales. The absence of hazardous reagents like strong Lewis acids or pyrophoric catalysts simplifies the safety profile of the plant, reducing the regulatory burden and insurance costs associated with handling dangerous chemicals. Waste streams generated during the process are primarily aqueous salt solutions and organic solvents that can be efficiently treated or recycled using standard environmental control technologies. This alignment with green chemistry principles not only ensures compliance with increasingly stringent environmental regulations but also enhances the corporate sustainability profile of the manufacturing entity. The ability to scale from 100 kgs to 100 MT annual commercial production without significant process redesign demonstrates the industrial maturity of this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this quinazoline derivative technology, based on the detailed specifications and experimental data provided in the patent documentation. These insights are designed to clarify the practical implications for R&D teams evaluating the compound for drug development and for procurement professionals assessing its supply chain viability. Understanding these nuances is critical for making informed decisions about integrating this advanced intermediate into your product pipeline. The answers reflect a deep analysis of the chemical properties and process capabilities described in the intellectual property.
Q: How does this quinazoline derivative improve upon traditional cytotoxic drugs?
A: This derivative combines tyrosine protein kinase inhibition with metal coordination capabilities, significantly improving water solubility and reducing the toxic side effects associated with conventional cytotoxic agents like cisplatin.
Q: What are the scalability prospects for this synthesis route?
A: The synthesis utilizes standard organic reactions such as nucleophilic substitution and reflux condensation, which are highly amenable to commercial scale-up from 100 kgs to 100 MT annual production.
Q: Does this compound address drug resistance issues in tumor cells?
A: Yes, the single-molecule multi-target mechanism acts on both protein kinases and DNA, which helps reduce the possibility of tumor cells developing acquired resistance to the treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline Derivative Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring these complex quinazoline derivatives from benchtop discovery to global market availability. Our technical team is uniquely equipped to handle the nuanced chemistry involved in synthesizing metal-coordinating intermediates, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that the transition from patent to production involves more than just following a recipe; it requires deep process optimization to maximize yield and minimize cost while maintaining the highest quality. Our facility is designed to accommodate the specific solvent and temperature requirements of this synthesis, with dedicated containment systems for handling potent compounds safely. By leveraging our expertise in process chemistry and scale-up engineering, we can help you navigate the regulatory landscape and accelerate the timeline for your oncology drug candidates. We are committed to being a transparent and responsive partner, providing real-time updates on production status and proactively identifying potential risks before they impact your supply.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project needs. Request a Customized Cost-Saving Analysis to understand how our optimized processes can reduce your overall cost of goods without compromising on quality. We encourage potential partners to contact us for specific COA data and route feasibility assessments that demonstrate our capability to deliver this high-value intermediate reliably. Whether you require small quantities for preclinical studies or metric tons for commercial launch, our flexible production lines and dedicated project management ensure that your supply chain remains uninterrupted. Let us help you unlock the full potential of this innovative technology by providing the manufacturing excellence it deserves.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
